Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial Pharmaceutical Manufacturing
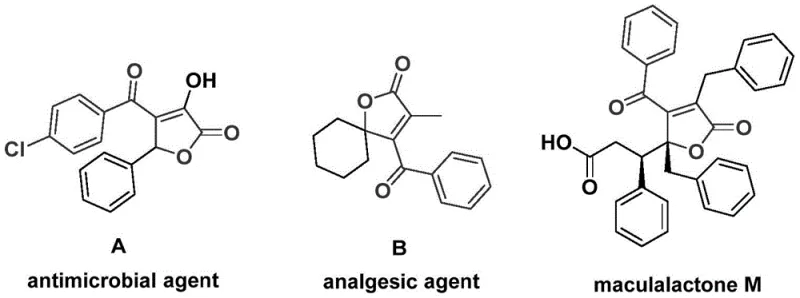
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for safer, more efficient, and scalable methodologies. A significant breakthrough in this domain is detailed in patent CN112239436A, which discloses a robust preparation method for 4-acyl-2(5H)-furanone compounds. These structural motifs are not merely academic curiosities; they serve as critical backbones in numerous natural products and bioactive molecules with profound therapeutic potential. For instance, specific derivatives have demonstrated significant antimicrobial activity, while others exhibit potent analgesic properties or function as antifouling agents in marine ecosystems. The ability to construct these complex scaffolds efficiently is paramount for pharmaceutical developers seeking to access novel chemical space for drug discovery programs.
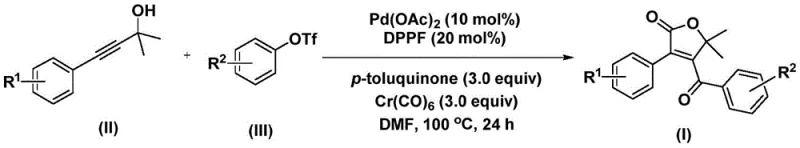
This patent introduces a sophisticated palladium-catalyzed bis-carbonylation strategy that utilizes propiolic alcohols and aryl trifluoromethanesulfonates as key starting materials. Unlike traditional methods that often rely on hazardous gaseous carbon monoxide or multi-step sequences, this innovative approach employs chromium hexacarbonyl as a solid carbon monoxide surrogate. This substitution represents a major leap forward in process safety and operational simplicity. By integrating the carbonylation and cyclization steps into a single pot, the method drastically reduces processing time and waste generation. For R&D directors and process chemists, this translates to a streamlined pathway for generating diverse libraries of furanone derivatives, accelerating the lead optimization phase in drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl-2(5H)-furanone skeletons has been fraught with challenges that hinder their widespread application in industrial settings. Conventional carbonylation reactions typically necessitate the use of high-pressure carbon monoxide gas, which poses severe safety risks regarding toxicity and flammability, requiring specialized high-pressure reactors and rigorous safety protocols. Furthermore, many existing synthetic routes involve multiple steps, including separate protection and deprotection sequences, which inevitably lead to lower overall yields and increased production costs. The reliance on harsh reaction conditions often limits the scope of compatible functional groups, preventing the incorporation of sensitive moieties that are frequently required in modern medicinal chemistry. These limitations create significant bottlenecks for procurement managers and supply chain heads who struggle with the high costs and long lead times associated with sourcing these complex intermediates.
The Novel Approach
The methodology described in CN112239436A effectively dismantles these barriers through a cleverly designed one-pot cascade reaction. By utilizing aryl trifluoromethanesulfonates and propiolic alcohols in the presence of a palladium catalyst and a solid CO source, the process achieves direct construction of the furanone core with remarkable efficiency. The use of chromium hexacarbonyl eliminates the need for handling dangerous CO gas, allowing the reaction to proceed in standard laboratory glassware or conventional stainless steel reactors without high-pressure infrastructure. This shift not only enhances safety but also simplifies the engineering requirements for scale-up. Moreover, the reaction demonstrates exceptional substrate compatibility, tolerating a wide array of substituents including halogens, alkyl groups, and alkoxy groups on both aromatic rings. This versatility ensures that the method can be applied to synthesize a broad spectrum of analogues, making it an invaluable tool for rapid structure-activity relationship (SAR) studies.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring consistent product quality. The catalytic cycle initiates with the oxidative addition of the aryl trifluoromethanesulfonate to the active palladium(0) species, generated in situ from palladium acetate and the DPPF ligand. This step forms a key aryl-palladium intermediate, which is then subjected to the first carbon monoxide insertion. The CO is liberated from the chromium hexacarbonyl additive under the thermal conditions of the reaction, inserting into the palladium-carbon bond to generate an acyl-palladium species. Subsequently, the propiolic alcohol substrate coordinates and inserts into this acyl-palladium bond, forming a five-membered ring palladium intermediate. This cyclization step is critical for establishing the furanone core structure.
Following the initial cyclization, a second molecule of carbon monoxide inserts into the intermediate, expanding the coordination sphere to form a six-membered ring palladium complex. The cycle concludes with a reductive elimination step, which releases the final 4-acyl-2(5H)-furanone product and regenerates the active palladium(0) catalyst to continue the cycle. The presence of p-toluquinone as an additive plays a vital role in facilitating the re-oxidation or stabilization of the catalytic species, ensuring high turnover numbers. From an impurity control perspective, this well-defined mechanism minimizes side reactions such as homocoupling of the alkyne or simple carbonylation without cyclization. The precise stoichiometry and the choice of the bidentate phosphine ligand (DPPF) are instrumental in directing the regioselectivity of the alkyne insertion, thereby ensuring that the acyl group ends up at the desired 4-position of the furanone ring with high fidelity.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent quality and reaction conditions to maximize yield and purity. The protocol is designed to be operationally simple, avoiding the need for exotic equipment or cryogenic conditions. The standard procedure involves combining the palladium catalyst, ligand, solid CO source, oxidant, and substrates in a polar aprotic solvent such as DMF. The mixture is then heated to promote the cascade sequence. While the general concept is straightforward, the specific ratios of reagents and the purification strategy are critical for obtaining pharmaceutical-grade material. The detailed standardized synthesis steps, including precise molar equivalents and workup procedures, are outlined in the guide below to ensure reproducibility across different batches and scales.
- Charge a reaction vessel with palladium acetate catalyst, DPPF ligand, chromium hexacarbonyl, p-toluquinone additive, propiolic alcohol substrate, and aryl trifluoromethanesulfonate in DMF solvent.
- Heat the reaction mixture to 100°C under inert atmosphere and stir continuously for 24 hours to facilitate the bis-carbonylation cyclization.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The transition from gaseous carbon monoxide to a solid surrogate like chromium hexacarbonyl fundamentally alters the risk profile and infrastructure costs of the manufacturing process. Facilities no longer need to invest in specialized high-pressure gas handling systems or maintain complex safety protocols for toxic gas storage, leading to substantial capital expenditure savings. Furthermore, the high atom economy and one-pot nature of the reaction reduce the consumption of solvents and auxiliary reagents, aligning with green chemistry principles and lowering waste disposal costs. These factors combine to create a more cost-effective manufacturing process that enhances the overall competitiveness of the final API or intermediate.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of readily available, inexpensive starting materials such as aryl triflates and propiolic alcohols significantly drive down production costs. The high yields reported in the patent data mean that less raw material is wasted, improving the overall material throughput. Additionally, the simplified workup procedure, which involves basic filtration and chromatography, reduces labor hours and processing time compared to multi-step alternatives. This efficiency allows for a more aggressive pricing strategy while maintaining healthy margins, providing a distinct advantage in competitive bidding scenarios for pharmaceutical contracts.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures a stable and continuous supply of critical intermediates. Since the starting materials are commercially available and the reaction conditions are not overly sensitive to minor fluctuations, the risk of batch failure is minimized. This reliability is crucial for maintaining uninterrupted production schedules for downstream drug manufacturing. The ability to source precursors from multiple vendors further mitigates supply chain risks, preventing bottlenecks that could delay project timelines. For supply chain heads, this means greater predictability in inventory management and the ability to respond swiftly to changes in market demand without compromising on quality or delivery commitments.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the use of standard heating and stirring equipment, avoiding the engineering complexities associated with high-pressure gas reactions. The use of DMF as a solvent, while requiring proper recovery systems, is well-established in the industry, and the solid nature of the CO source simplifies dosing and mixing in large reactors. From an environmental standpoint, the reduced waste generation and avoidance of toxic gas emissions contribute to a smaller environmental footprint. This alignment with increasingly stringent environmental regulations ensures long-term operational viability and reduces the regulatory burden on the manufacturing site, making it a sustainable choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial viability of this synthesis method, we have compiled a set of frequently asked questions based on the patent literature. These answers provide clarity on the reaction scope, safety considerations, and potential applications of the resulting furanone compounds. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing pipelines. The following section covers critical aspects ranging from catalyst loading to product purification, ensuring transparency and confidence in the proposed manufacturing route.
Q: What is the primary advantage of using Chromium Hexacarbonyl in this synthesis?
A: Chromium hexacarbonyl serves as a solid, easy-to-handle substitute for toxic gaseous carbon monoxide, significantly enhancing operational safety and simplifying reactor requirements for commercial scale-up.
Q: What is the typical yield range for this palladium-catalyzed furanone synthesis?
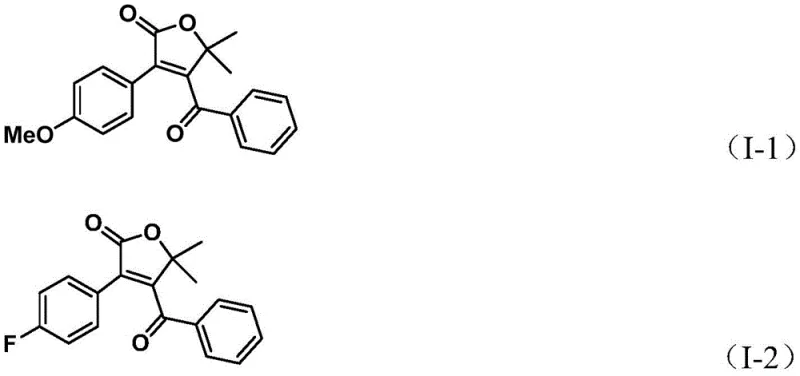
A: According to the patent data, the method demonstrates excellent substrate compatibility with isolated yields ranging from 70% to 95% across various substituted aryl triflates and propiolic alcohols.
Q: Can this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the protocol exhibits broad functional group tolerance, successfully accommodating electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as halogens and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112239436A for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates regardless of volume. Our state-of-the-art facilities are equipped to handle complex organometallic reactions safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We are committed to bridging the gap between innovative academic research and industrial reality, providing a seamless transition from bench-scale optimization to full-scale manufacturing.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthesis technology for their specific projects. Whether you require custom synthesis of novel furanone derivatives or scale-up of existing routes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your unique needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain and deep technical expertise that will empower your R&D efforts and streamline your path to market.
