Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN112239456B introduces a robust methodology for the preparation of substituted 2,3-dihydroquinolone compounds, a structural motif prevalent in numerous bioactive molecules including antitumor agents and analgesics. This innovation leverages a transition metal palladium-catalyzed carbonylation strategy, utilizing N-pyridine sulfonyl-o-iodoaniline and various olefins as key starting materials. By replacing traditional gaseous carbon monoxide sources with a solid surrogate, the process addresses critical safety and operational challenges inherent in industrial organic synthesis. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant advancement in accessing complex heterocyclic libraries with improved safety and scalability.
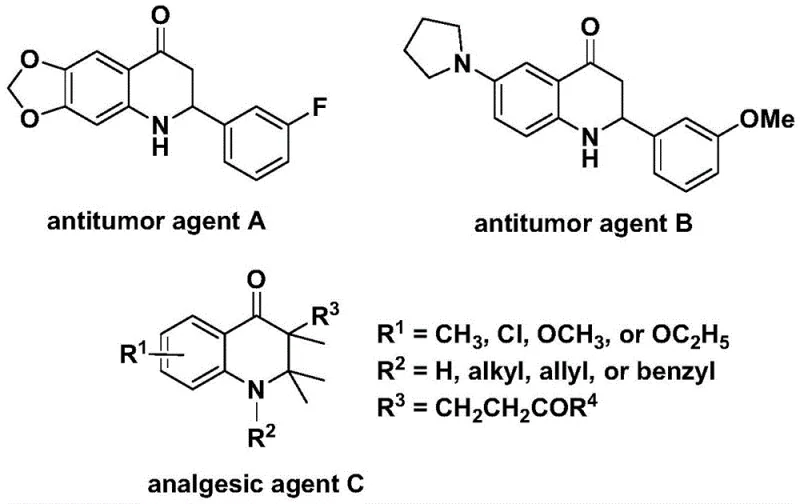
The 2,3-dihydroquinolone skeleton is a privileged structure in drug discovery, underpinning the efficacy of various therapeutic agents. As illustrated in the background art, compounds such as antitumor agent A and B demonstrate significant activity against human cancer cells, while analgesic agent C exhibits potent pain-relieving properties. The widespread presence of this core in pharmacologically active molecules underscores the urgent need for versatile and efficient synthetic methods. Traditional approaches to constructing this ring system often suffer from limitations such as harsh reaction conditions, limited substrate scope, or the requirement for hazardous reagents. The disclosed invention overcomes these barriers by employing a carbonylation reaction that is not only operationally simple but also highly adaptable to diverse functional groups, thereby widening the practical applicability of this valuable chemical scaffold in the development of new drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has relied heavily on direct carbonylation using carbon monoxide gas. While effective in principle, the use of CO gas presents severe logistical and safety hurdles for large-scale manufacturing. Handling high-pressure cylinders of toxic carbon monoxide requires specialized infrastructure, rigorous safety protocols, and expensive containment systems, which drastically increases capital expenditure and operational complexity. Furthermore, conventional methods often struggle with substrate compatibility, particularly when sensitive functional groups are present on the aromatic ring or the olefin partner. Many existing protocols necessitate extreme temperatures or stoichiometric amounts of toxic heavy metals, leading to difficult purification processes and substantial waste generation. These factors collectively hinder the rapid scale-up of promising drug candidates, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
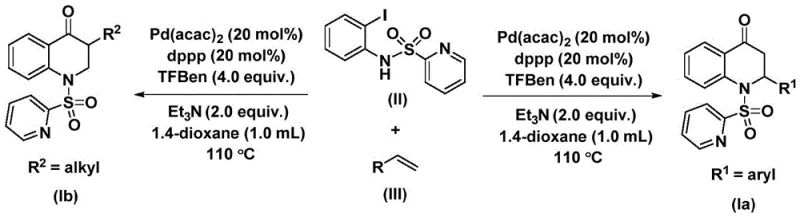
The methodology described in patent CN112239456B offers a transformative solution by utilizing 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide substitute. This strategic substitution eliminates the need for gaseous CO, allowing the reaction to proceed under standard atmospheric pressure in a closed vessel, thereby significantly enhancing operational safety. The reaction employs a palladium catalyst system, specifically bis(acetylacetone)palladium coordinated with 1,3-bis(diphenylphosphino)propane (dppp), which facilitates the efficient insertion of the carbonyl group into the carbon-nitrogen bond framework. This novel approach enables the synthesis of both 2-aryl and 3-alkyl substituted 2,3-dihydroquinolones through simple substrate design. The process operates at a moderate temperature of 110 °C in 1,4-dioxane, demonstrating excellent functional group tolerance and high reaction efficiency, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial for activating the inert aryl halide towards subsequent transformations. Following this activation, the carbon monoxide molecule, released in situ from the decomposition of the solid 1,3,5-trimesic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This insertion step effectively builds the carbonyl functionality directly into the growing molecular framework without the need for external gas feeds. The coordination and subsequent migratory insertion of the olefin substrate into the acyl-palladium bond then generates a palladium-alkyl intermediate. Finally, an intramolecular reductive elimination occurs, closing the six-membered ring and releasing the substituted 2,3-dihydroquinolone product while regenerating the active palladium catalyst for the next turnover.
Understanding the mechanistic nuances is vital for controlling the impurity profile of the final product. The use of the pyridine sulfonyl group on the aniline nitrogen serves a dual purpose: it acts as a directing group to facilitate the initial palladium insertion and subsequently participates in the cyclization event to form the lactam ring. The choice of ligand, specifically dppp, is optimized to balance the steric and electronic properties of the palladium center, ensuring rapid turnover while minimizing side reactions such as homocoupling of the aryl iodide or polymerization of the olefin. The reaction conditions, including the use of triethylamine as a base and the specific solvent system, are tuned to stabilize the key intermediates and drive the equilibrium towards the desired cyclic product. This precise control over the reaction pathway ensures high chemoselectivity, which is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can be detrimental.


How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The experimental procedure outlined in the patent provides a clear roadmap for executing this transformation with high reproducibility. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an organic solvent, followed by heating to promote the carbonylative cyclization. The detailed standardized synthesis steps below describe the specific molar ratios and workup procedures required to achieve optimal yields ranging from 59% to 88% across various substrates.
- Charge a reaction vessel with bis(acetylacetone)palladium, dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and the selected olefin substrate in 1,4-dioxane.
- Heat the reaction mixture to 110 °C and maintain stirring for 48 hours to ensure complete conversion of the starting materials into the dihydroquinolone scaffold.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the high-purity substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The shift from gaseous to solid reagents fundamentally alters the risk profile and cost structure of the manufacturing process. By removing the dependency on hazardous gas infrastructure, facilities can reduce their insurance premiums and safety compliance costs, leading to substantial cost savings in the long term. Furthermore, the use of commercially available starting materials, such as the olefins and the palladium catalyst, ensures a stable and continuous supply chain, reducing the risk of production delays caused by raw material shortages. The robustness of the reaction conditions also implies a wider operating window, which enhances process reliability and reduces the likelihood of batch failures.
- Cost Reduction in Manufacturing: The elimination of specialized high-pressure equipment for carbon monoxide handling translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX). Additionally, the high atom economy of the carbonylation reaction and the use of a solid CO source minimize waste disposal costs associated with gas scrubbing systems. The ability to run the reaction at moderate temperatures (110 °C) rather than extreme conditions further reduces energy consumption, contributing to a more sustainable and cost-effective manufacturing process that aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: The starting materials, including N-pyridine sulfonyl-o-iodoaniline and various olefins, are either commercially available or easily synthesized from commodity chemicals like o-iodoaniline. This accessibility ensures that the supply chain is not vulnerable to the bottlenecks often associated with exotic or highly regulated reagents. The stability of the solid carbon monoxide substitute also simplifies logistics and storage, as it does not require the same stringent safety measures as compressed gas cylinders, thereby streamlining inventory management and ensuring consistent production schedules.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable, with the potential for expansion from gram-level laboratory synthesis to industrial-scale production. The simplified post-treatment procedure, involving filtration and standard column chromatography, facilitates easy isolation of the product. Moreover, the avoidance of toxic CO gas significantly reduces the environmental footprint of the process, making it easier to meet increasingly stringent environmental regulations. This compliance advantage is critical for maintaining uninterrupted operations in regions with strict emissions controls.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on substrate scope, reaction conditions, and product quality.
Q: What is the primary advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: Using 1,3,5-trimesic acid phenol ester serves as a solid carbon monoxide substitute, eliminating the need for handling toxic and hazardous CO gas cylinders, thereby significantly improving laboratory and plant safety profiles.
Q: What types of olefin substrates are compatible with this palladium-catalyzed protocol?
A: The method demonstrates broad substrate compatibility, successfully accommodating both aryl-substituted olefins (such as styrenes with methyl, chloro, or fluoro groups) and alkyl-substituted olefins (including linear, branched, and silyl-containing alkenes).
Q: How does this method impact the purity profile of the final pharmaceutical intermediate?
A: The reaction proceeds with high chemoselectivity and functional group tolerance, minimizing side reactions and facilitating easier downstream purification to meet stringent purity specifications required for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results can be seamlessly translated into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle complex organometallic reactions safely and efficiently, leveraging technologies like the one described in CN112239456B to provide our partners with a competitive edge.
We invite you to collaborate with us to explore the full potential of this innovative synthetic route for your specific project needs. Whether you require custom synthesis services or bulk supply of key intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our capabilities can support your supply chain goals and drive your projects forward.
