Scalable Palladium-Catalyzed Carbonylation for High-Purity 2,3-Dihydroquinolone Derivatives
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B introduces a significant advancement in the preparation of substituted 2,3-dihydroquinolone compounds, a privileged scaffold found in numerous therapeutic agents. This technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and various olefins as key starting materials. The biological significance of this core structure cannot be overstated, as evidenced by its presence in potent antitumor agents and analgesics shown in the literature. 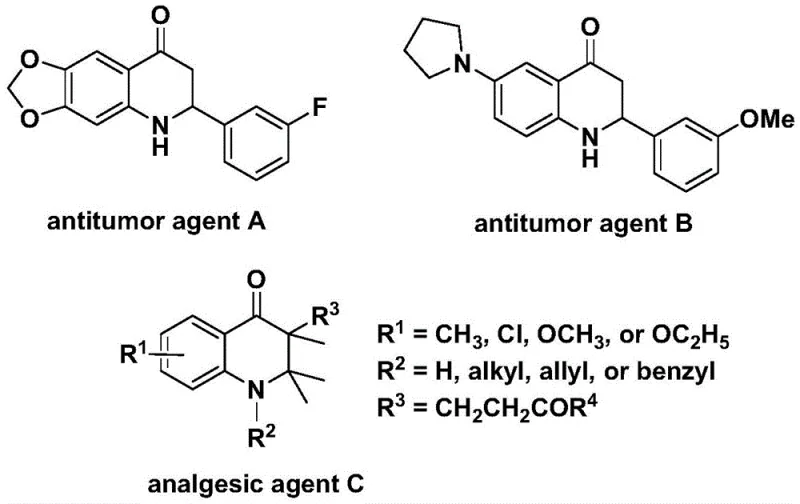 . By replacing toxic carbon monoxide gas with a solid surrogate, this invention addresses critical safety and handling concerns while maintaining high reaction efficiency and broad substrate compatibility, making it an attractive candidate for reliable pharmaceutical intermediate supplier networks.
. By replacing toxic carbon monoxide gas with a solid surrogate, this invention addresses critical safety and handling concerns while maintaining high reaction efficiency and broad substrate compatibility, making it an attractive candidate for reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the 2,3-dihydroquinolone skeleton often rely on direct carbonylation using carbon monoxide gas, which presents severe logistical and safety challenges in a manufacturing environment. Handling high-pressure CO gas requires specialized infrastructure, rigorous safety protocols, and expensive equipment to prevent leakage and exposure, significantly increasing the capital expenditure for production facilities. Furthermore, conventional methods frequently suffer from limited substrate tolerance, where sensitive functional groups on the aromatic ring or the alkene chain may degrade under harsh reaction conditions. Many existing protocols also struggle with regioselectivity issues or require excessive reaction times and temperatures that lead to the formation of complex impurity profiles, complicating downstream purification and reducing overall process economics. These factors collectively hinder the cost reduction in pharmaceutical intermediate manufacturing and limit the ability to rapidly scale up production for clinical trials.
The Novel Approach
The methodology disclosed in CN112239456B revolutionizes this landscape by employing 1,3,5-trimesic acid phenyl ester (TFBen) as a solid carbon monoxide surrogate, effectively decoupling the synthesis from gaseous CO sources. This innovative approach allows the reaction to proceed under relatively mild conditions, typically at 110 °C in 1,4-dioxane, using a palladium catalyst system comprising Pd(acac)2 and dppp ligand. 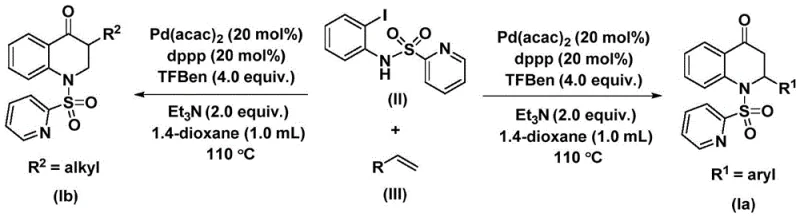 . The process demonstrates exceptional versatility, successfully accommodating a wide range of olefin substrates including aryl, alkyl, and silyl-substituted alkenes to produce both 2-aryl and 3-alkyl substituted derivatives. This flexibility enables the rapid generation of diverse chemical libraries for drug discovery while ensuring that the commercial scale-up of complex pharmaceutical intermediates remains feasible and safe. The use of a solid CO source not only simplifies the operational procedure but also enhances the reproducibility of the reaction across different batch sizes.
. The process demonstrates exceptional versatility, successfully accommodating a wide range of olefin substrates including aryl, alkyl, and silyl-substituted alkenes to produce both 2-aryl and 3-alkyl substituted derivatives. This flexibility enables the rapid generation of diverse chemical libraries for drug discovery while ensuring that the commercial scale-up of complex pharmaceutical intermediates remains feasible and safe. The use of a solid CO source not only simplifies the operational procedure but also enhances the reproducibility of the reaction across different batch sizes.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate.  . Subsequently, carbon monoxide, which is released in situ from the thermal decomposition of the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This step is critical for introducing the carbonyl functionality into the heterocyclic ring. The olefin substrate, represented generally by Formula (III) where R can be aryl or alkyl, then coordinates to the metal center and undergoes migratory insertion into the acyl-palladium bond.
. Subsequently, carbon monoxide, which is released in situ from the thermal decomposition of the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This step is critical for introducing the carbonyl functionality into the heterocyclic ring. The olefin substrate, represented generally by Formula (III) where R can be aryl or alkyl, then coordinates to the metal center and undergoes migratory insertion into the acyl-palladium bond.  . This sequence creates a new carbon-carbon bond and extends the molecular framework. Finally, a reductive elimination step releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst, allowing the cycle to continue. This well-defined mechanism ensures high atom economy and minimizes the formation of side products associated with uncontrolled radical pathways.
. This sequence creates a new carbon-carbon bond and extends the molecular framework. Finally, a reductive elimination step releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst, allowing the cycle to continue. This well-defined mechanism ensures high atom economy and minimizes the formation of side products associated with uncontrolled radical pathways.
Impurity control in this system is largely governed by the stability of the palladium intermediates and the purity of the starting materials. The use of the pyridine sulfonyl protecting group on the aniline nitrogen plays a dual role: it activates the ortho-iodo position for oxidative addition and prevents unwanted N-alkylation or polymerization side reactions during the olefin insertion phase. The choice of triethylamine as an additive helps to neutralize any acidic byproducts generated during the reaction, maintaining a neutral pH that preserves the integrity of acid-sensitive functional groups on the substrate. Furthermore, the specific ligand environment provided by dppp stabilizes the palladium center against aggregation into inactive palladium black, ensuring consistent catalytic turnover throughout the 48-hour reaction period. This mechanistic robustness translates directly to a cleaner crude reaction mixture, reducing the burden on purification teams and improving the final yield of high-purity pharmaceutical intermediates.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and temperature controls to maximize yield and minimize waste. The standard protocol involves charging a reaction vessel with the iodinated aniline derivative, the chosen olefin, the palladium catalyst, the phosphine ligand, and the solid CO source in an anhydrous organic solvent.
- Combine bis(acetylacetone)palladium, dppp ligand, triethylamine, TFBen (CO surrogate), N-pyridine sulfonyl-o-iodoaniline, and olefin in 1,4-dioxane solvent.
- Heat the reaction mixture to 110 °C and maintain stirring for 48 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify the crude product via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers distinct strategic advantages over traditional carbonylation methods. The shift from gaseous carbon monoxide to a solid surrogate fundamentally alters the risk profile of the manufacturing process, eliminating the need for specialized gas storage facilities and reducing insurance premiums associated with hazardous material handling. This transition facilitates a more agile supply chain, as the solid reagents can be stored safely in standard warehouses and transported without the regulatory burdens associated with compressed toxic gases. Consequently, this leads to significant cost reduction in pharmaceutical intermediate manufacturing by lowering overhead costs related to safety compliance and infrastructure maintenance. The ability to source stable, shelf-stable reagents ensures continuity of supply even during global logistics disruptions, providing a buffer against market volatility.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas equipment and the use of commercially available, inexpensive catalysts like Pd(acac)2 drastically lower the capital and operational expenditures required for production. By avoiding the complexities of gas handling, facilities can repurpose existing reactor trains for this chemistry without major retrofitting, accelerating time-to-market for new drug candidates. Additionally, the high conversion rates observed with substrates like 4-methylstyrene and 4-chlorostyrene mean less raw material is wasted, directly improving the cost-of-goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on widely available organic building blocks, such as substituted styrenes and simple alkenes, reduces dependency on single-source suppliers for exotic reagents. The robustness of the reaction conditions, which tolerate a variety of functional groups including halogens and ethers, allows for the use of grade-appropriate starting materials that are easier to procure in bulk quantities. This flexibility ensures that production schedules remain uninterrupted even if specific grades of raw materials face temporary shortages, thereby securing the supply chain for critical drug substances.
- Scalability and Environmental Compliance: The process has been demonstrated to work efficiently at the gram level with clear pathways for scaling to kilogram and tonne scales, supported by the use of standard organic solvents like 1,4-dioxane which are easily recovered and recycled. The absence of toxic gas emissions aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. The straightforward post-treatment involving filtration and column chromatography allows for the efficient isolation of products with high purity, minimizing solvent usage and waste generation compared to multi-step alternative syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this technology into their existing pipelines.
Q: What carbon monoxide source is used in this synthesis?
A: The process utilizes 1,3,5-trimethoxybenzene phenyl ester (TFBen) as a solid carbon monoxide surrogate, eliminating the need for hazardous CO gas cylinders.
Q: What yields can be expected from this palladium-catalyzed method?
A: Experimental data indicates isolated yields ranging from 59% to 88%, depending on the specific aryl or alkyl substituents on the olefin substrate.
Q: Is this method suitable for large-scale production?
A: Yes, the patent explicitly states the method is operable at the gram level and possesses the simplicity and robustness required for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced palladium-catalyzed technology for the commercial production of high-value heterocyclic intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2,3-dihydroquinolone derivative meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance means that clients can rely on us for consistent supply of materials that are ready for immediate use in downstream API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this novel carbonylation method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this safer, more efficient route can optimize your overall manufacturing budget. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate route feasibility assessments for your proprietary targets, ensuring your supply chain remains competitive and resilient in a rapidly evolving market.
