Scalable Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Applications
Scalable Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Applications
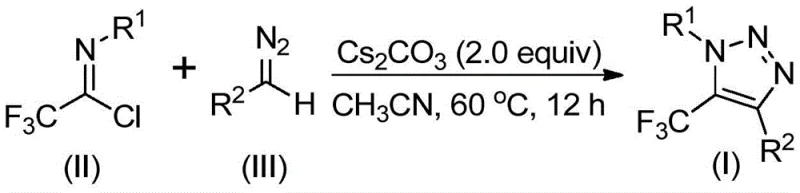
The rapid evolution of medicinal chemistry has placed 1,2,3-triazole scaffolds at the forefront of drug discovery, particularly when functionalized with electron-withdrawing groups like the trifluoromethyl moiety. Patent CN113121462B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical safety and efficiency bottlenecks in current manufacturing paradigms. This technology leverages a unique base-promoted cyclization strategy that bypasses the traditional reliance on hazardous azide chemistry and expensive transition metal catalysts. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a significant leap forward in process safety and cost structure. The method utilizes readily available trifluoroethylimidoyl chlorides and diazo compounds, enabling the construction of complex heterocyclic cores under mild conditions. By shifting away from explosive precursors, this protocol not only enhances laboratory safety but also streamlines the regulatory pathway for commercial production of high-value bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted triazoles has been dominated by two primary methodologies, both of which suffer from severe operational drawbacks that hinder efficient cost reduction in API manufacturing. The first approach involves copper-catalyzed [3+2] cycloaddition between alkynes and organic azides, followed by trifluoromethylation. This route is inherently problematic due to the use of toxic and potentially explosive organic azides, which pose significant safety risks during scale-up. Furthermore, the requirement for copper catalysts introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet pharmaceutical purity standards. The second conventional method employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. While metal-free, this pathway still relies on the dangerous handling of azide reagents and often requires specialized trifluoromethylating agents that drive up raw material costs. These legacy processes create substantial supply chain vulnerabilities and limit the ability to produce high-purity heterocyclic building blocks economically.
The Novel Approach
In stark contrast to these hazardous legacy routes, the technology described in CN113121462B introduces a simple, efficient, and metal-free synthesis strategy. As illustrated in the reaction scheme below, the novel method utilizes trifluoroethylimidoyl chloride and diazo compounds as starting materials, promoted by a mild inorganic base such as cesium carbonate. This approach completely eliminates the need for toxic azides, explosive reagents, and transition metal catalysts, thereby fundamentally altering the safety profile of the manufacturing process. The reaction proceeds smoothly in common aprotic solvents like acetonitrile at moderate temperatures, offering excellent functional group tolerance and high reaction efficiency. This paradigm shift allows manufacturers to achieve commercial scale-up of complex fluorinated intermediates with significantly reduced environmental impact and operational risk. The avoidance of copper not only removes heavy metal removal steps but also simplifies the waste stream, making this a highly attractive option for green chemistry initiatives in the fine chemical sector.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation offers profound implications for impurity control and process robustness, key concerns for any reliable pharmaceutical intermediate supplier. The reaction is hypothesized to initiate via a base-promoted intermolecular nucleophilic addition-elimination process. Specifically, the diazo compound acts as a nucleophile attacking the electrophilic carbon of the trifluoroethylimidoyl chloride, facilitating the formation of a critical carbon-carbon bond. This initial step is followed by an intramolecular 5-endo-dig cyclization, which closes the triazole ring to yield the final 5-trifluoromethyl substituted product. The use of cesium carbonate as a promoter is particularly advantageous; its moderate basicity effectively drives the reaction forward without inducing excessive decomposition of the sensitive diazo species. This controlled reactivity minimizes the formation of polymeric byproducts or decomposition fragments that often plague diazo chemistry, ensuring a cleaner crude reaction profile. Consequently, the downstream purification burden is drastically reduced, allowing for higher overall yields and superior product quality.
Furthermore, the structural versatility of this mechanism allows for extensive modulation of the R1 and R2 substituents, enabling the synthesis of a diverse library of triazole derivatives. The patent data indicates that various aryl, alkyl, and acyl groups can be tolerated on both the imidoyl chloride and the diazo components without compromising reaction efficiency. This broad substrate scope is essential for medicinal chemists who require rapid access to analog libraries for structure-activity relationship (SAR) studies. The stability of the intermediates formed during the cyclization process ensures that the reaction can be monitored and controlled precisely, reducing the risk of runaway exotherms. By understanding these mechanistic nuances, process engineers can optimize parameters such as temperature and stoichiometry to maximize throughput while maintaining the stringent purity specifications required for clinical-grade materials.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis route in a production environment requires careful attention to reagent quality and reaction parameters to ensure consistent results. The protocol is designed to be operationally simple, utilizing standard laboratory equipment and commercially available solvents. The following guide outlines the generalized procedure derived from the patent examples, demonstrating the ease with which this chemistry can be adopted for pilot and commercial scales. For detailed standard operating procedures and specific batch records, please refer to the technical documentation provided below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this technology offers compelling advantages that directly address the pain points of modern chemical supply chains. By transitioning to this azide-free and metal-free protocol, organizations can achieve substantial cost savings and enhance supply continuity. The elimination of hazardous reagents reduces the need for specialized containment infrastructure and lowers insurance premiums associated with chemical manufacturing. Moreover, the reliance on commodity chemicals like cesium carbonate and acetonitrile ensures that raw material availability remains stable, mitigating the risk of supply disruptions caused by the scarcity of exotic catalysts or dangerous precursors. This stability is crucial for maintaining reducing lead time for high-purity intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the complete removal of transition metal catalysts. In traditional copper-catalyzed routes, the cost of the catalyst itself, combined with the expensive ligands and the subsequent heavy metal scavenging resins required for purification, adds a substantial premium to the final product cost. By utilizing a simple inorganic base promoter, this new method eliminates these line items entirely. Additionally, the simplified workup procedure, which avoids complex extraction or chelation steps, reduces labor hours and solvent consumption. The overall result is a leaner manufacturing process with a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global marketplace without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically trifluoroethylimidoyl chlorides and diazo compounds, are derived from abundant feedstocks and are widely available from multiple global suppliers. This diversification of the supply base reduces dependency on single-source vendors for specialized reagents like organic azides, which often have limited production capacity due to safety regulations. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in raw material quality, further stabilizing the supply chain. For procurement managers, this translates to more predictable delivery schedules and the ability to secure long-term contracts with greater confidence, ensuring uninterrupted production of critical drug substances.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for expansion. The absence of explosive azides removes the most significant barrier to scaling nitrogen-heterocycle synthesis, allowing reactors to be operated at larger volumes without the need for extreme safety mitigation measures. Furthermore, the generation of waste is minimized; the primary byproduct is cesium chloride, which is relatively benign compared to the heavy metal waste streams generated by copper catalysis. This aligns perfectly with increasingly strict environmental regulations and corporate sustainability goals. The ability to run the reaction in green solvents like acetonitrile, which can be efficiently recovered and recycled, further enhances the environmental profile, making it easier to obtain necessary regulatory approvals for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production workflows.
Q: Does this synthesis method require hazardous azide reagents?
A: No, unlike conventional copper-catalyzed methods, this novel protocol utilizes stable diazo compounds and trifluoroethylimidoyl chloride, completely eliminating the need for toxic and explosive organic azides.
Q: What are the advantages regarding heavy metal contamination?
A: This process is metal-free, utilizing cesium carbonate as a promoter instead of copper catalysts. This significantly simplifies downstream purification and ensures the final API intermediates meet stringent heavy metal specifications without expensive scavenging steps.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent demonstrates that the reaction can be easily extended to the gram level with high efficiency. The use of common solvents like acetonitrile and mild temperatures (50-70°C) supports robust commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory to the factory floor. Our state-of-the-art facilities are equipped to handle fluorinated chemistry with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets your exacting requirements. We are committed to delivering high-quality intermediates that accelerate your drug development timelines while optimizing your overall manufacturing costs.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this process can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our triazole portfolio and to discuss route feasibility assessments for your custom synthesis requirements. Let us help you build a more resilient and efficient supply chain for your critical therapeutic candidates.
