Advanced Thieno Pyridazine Iridium Complexes for High Performance OLED Manufacturing
The landscape of organic electroluminescence (OLED) is undergoing a significant transformation driven by the urgent need for materials that combine high efficiency with robust environmental stability. Patent CN111362986B introduces a groundbreaking class of thieno[2,3-d]pyridazine cyclometalated iridium complexes that address the critical limitations of existing phosphorescent emitters. Traditional iridium complexes often suffer from severe sensitivity to oxygen, which drastically quenches luminescence and limits device lifespan in practical applications. This new technology leverages the unique electronic properties of fusing a thiophene group with a pyridazine ring to create a ligand system that not only enhances hole transport capabilities but also significantly reduces oxygen quenching probabilities. For R&D directors and procurement specialists in the display industry, this represents a pivotal shift towards more reliable and high-performance organic photoelectric materials that can withstand the rigors of commercial manufacturing environments without compromising on brightness or color purity.
![General structural formula of thieno[2,3-d]pyridazine cyclometalated iridium complex showing variable substituents](/insights/img/thieno-pyridazine-iridium-oled-supplier-20260314175613-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of cyclometalated iridium complexes for OLED applications has been hindered by the inherent instability of common ligand structures when exposed to ambient conditions. Conventional pyridazine-based iridium complexes typically exhibit moderate quantum yields ranging between 64% and 76%, which leaves substantial room for improvement in terms of energy conversion efficiency. Furthermore, many existing high-efficiency complexes rely on auxiliary ligands like acetylacetone, which possess weaker coordination abilities and are prone to dissociation under thermal or electrical excitation stress. This dissociation leads to a degradation of device performance over time and complicates the manufacturing process due to the need for stringent inert atmosphere controls. The steric hindrance around the nitrogen atoms coordinated to the iridium center in traditional designs often reduces binding energy, making the complexes susceptible to decomposition and limiting their operational lifetime in commercial display panels.
The Novel Approach
The innovative strategy outlined in this patent overcomes these barriers by utilizing a thieno[2,3-d]pyridazine framework that fundamentally alters the electronic environment of the metal center. By fusing an electron-rich thiophene unit with the pyridazine ring, the new ligand design increases the electron cloud density around the coordination sites, thereby strengthening the bond with the iridium atom and enhancing thermal stability. This structural modification allows for the direct preparation of tricyclometalated iridium complexes using iridium trichloride in a single step, eliminating the need for unstable auxiliary ligands that plague conventional methods. The result is a material that maintains high luminescence efficiency even in air-saturated solutions, offering a level of robustness that simplifies handling and processing requirements for large-scale production lines while delivering superior brightness and color stability.
Mechanistic Insights into Thieno[2,3-d]pyridazine Cyclometalation
The core mechanism driving the superior performance of these complexes lies in the synergistic interaction between the thiophene sulfur atoms and the pyridazine nitrogen atoms within the ligand structure. The thiophene unit acts as an electron donor, effectively improving the hole transport capacity of the luminescent material which is crucial for balancing charge injection in OLED devices. This electron-rich characteristic also facilitates stronger spin-orbit coupling through the heavy atom effect of iridium, promoting efficient intersystem crossing from singlet to triplet excited states. Consequently, the theoretical internal quantum efficiency can approach 100%, utilizing both singlet and triplet excitons for light emission. The specific arrangement of substituents on the aromatic rings further fine-tunes the energy levels, allowing for precise control over emission color while maintaining the structural integrity required for long-term operational stability in high-brightness applications.
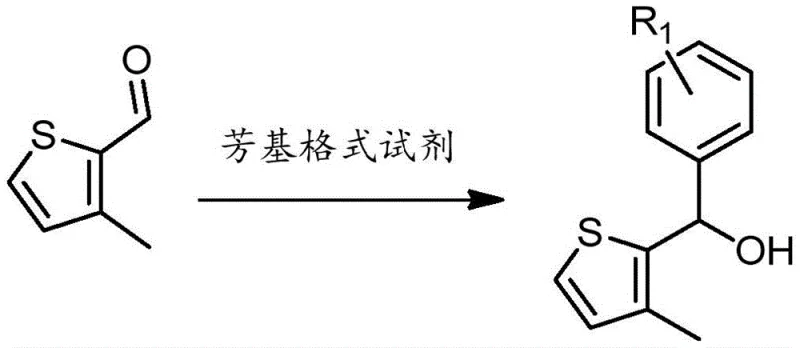
Impurity control is another critical aspect where this mechanistic design offers distinct advantages over prior art. The synthesis pathway avoids the use of difficult-to-separate isomers that often arise from non-selective chlorination reactions in traditional pyridazine synthesis. By starting with 3-methylthiophene-2-carboxaldehyde and proceeding through a controlled four-step sequence involving Grignard addition, oxidation, hydrazine condensation, and chlorination, the process ensures the formation of a single-position monochloro-substituted intermediate with high selectivity. This purity at the intermediate stage translates directly to the final complex, reducing the presence of non-emissive impurities that could act as quenching sites. The robust coordination geometry prevents ligand dissociation during device operation, ensuring that the emission spectrum remains consistent over thousands of hours of use without significant color shift or efficiency roll-off.
How to Synthesize Thieno[2,3-d]pyridazine Efficiently
The synthesis of these high-performance materials follows a logical and scalable pathway designed for industrial feasibility. The process begins with the preparation of the key monochloro-substituted thieno[2,3-d]pyridazine intermediate, which serves as the versatile building block for various derivative complexes. This intermediate is then reacted with a range of active hydrogen-containing compounds such as substituted phenols, thiophenols, or carbazoles to generate the final ligand system. The subsequent coordination with iridium trichloride is straightforward and high-yielding, allowing for the rapid production of tricyclic complexes suitable for immediate device fabrication. Detailed standardized synthesis steps see the guide below.
- Synthesize monochloro-substituted thieno[2,3-d]pyridazine intermediate via Grignard reaction, oxidation, hydrazine condensation, and chlorination.
- React the intermediate with active hydrogen compounds like substituted phenols or anilines to form the thieno[2,3-d]pyridazine ligand.
- Mix the ligand with iridium trichloride and solvent to directly prepare the tricyclometalated iridium complex in one step.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this thieno[2,3-d]pyridazine technology offers substantial strategic benefits that extend beyond mere technical performance. The use of readily available and cost-effective raw materials like 3-methylthiophene-2-carboxaldehyde significantly lowers the entry barrier for production compared to exotic precursors required by competing technologies. The streamlined synthesis route reduces the number of purification steps and minimizes solvent consumption, leading to a drastically simplified manufacturing process that lowers overall operational costs. For supply chain managers, this translates into a more resilient sourcing strategy with reduced dependency on complex global supply networks for specialized reagents, ensuring consistent availability of critical OLED materials even during market fluctuations.
- Cost Reduction in Manufacturing: The ability to directly prepare tricyclometalated iridium complexes in one step using iridium trichloride eliminates the need for expensive auxiliary ligands and multi-step coordination processes. This reduction in synthetic complexity directly correlates with lower labor and energy costs per kilogram of produced material. Furthermore, the high selectivity of the intermediate synthesis minimizes waste generation and reduces the burden on waste treatment facilities, contributing to substantial cost savings in environmental compliance and disposal. The overall process efficiency allows for higher throughput in existing reactor setups without requiring significant capital investment in new infrastructure.
- Enhanced Supply Chain Reliability: The robustness of the chemical structure against oxygen exposure means that storage and transportation requirements are less stringent than for traditional oxygen-sensitive phosphors. This reduces the risk of material degradation during logistics and allows for more flexible inventory management strategies. Suppliers can maintain larger stockpiles without fear of rapid quality deterioration, ensuring that production lines for downstream OLED manufacturers remain uninterrupted. The stability of the intermediates also allows for decentralized production models where key precursors can be synthesized at different locations and assembled locally, further enhancing supply chain resilience against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The synthesis method is inherently scalable, utilizing common organic solvents and reaction conditions that are easily adapted from laboratory to pilot and commercial scales. The avoidance of heavy metal catalysts in the ligand formation steps simplifies the removal of metal residues, ensuring that the final product meets stringent purity specifications required for electronic applications. This ease of purification reduces the environmental footprint of the manufacturing process by minimizing the use of aggressive chelating agents and extensive chromatography. The process aligns well with green chemistry principles, making it easier for manufacturers to meet increasingly strict environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thieno[2,3-d]pyridazine iridium complex technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide accurate guidance for potential adopters. Understanding these details is crucial for making informed decisions about integrating this material into existing product lines or developing new OLED architectures.
Q: How does the thieno[2,3-d]pyridazine ligand improve oxygen stability?
A: The electron-rich thiophene unit increases interaction with electron-deficient groups, making it difficult for oxygen molecules to contact the complex and reducing quenching.
Q: What is the quantum efficiency of these new iridium complexes?
A: The luminescence quantum efficiency exceeds 75% in degassed solutions and maintains over 10% efficiency even in air-saturated solutions.
Q: Can this synthesis be scaled for commercial OLED production?
A: Yes, the method uses cheap raw materials like 3-methylthiophene-2-carboxaldehyde and allows direct one-step tricyclic formation, facilitating scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thieno[2,3-d]pyridazine Iridium Complex Supplier
As a leading CDMO expert in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to bring this advanced iridium complex technology from patent to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of material performs exactly as expected in your OLED devices. Our commitment to quality assurance means that you can rely on us for the high-purity display & optoelectronic materials required to drive the next generation of visual technology.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your manufacturing costs and enhance your product performance. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your operation. We are ready to provide specific COA data and route feasibility assessments to support your R&D and procurement decisions. Let us help you secure a competitive advantage in the rapidly evolving OLED market with our reliable supply of advanced iridium complexes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
