Scaling Green Chemistry: A Novel Glycerol-Based Route for High-Purity Folic Acid Production
The pharmaceutical industry is constantly seeking sustainable alternatives to legacy synthesis routes that burden supply chains with hazardous waste and complex purification steps. Patent CN103755706A introduces a groundbreaking environmentally friendly preparation method for synthesizing folic acid, a critical vitamin essential for DNA synthesis and repair. This innovation fundamentally shifts the paradigm from relying on unstable and impure chlorinated ketones to utilizing glycerol, a abundant and renewable feedstock. By leveraging a composite catalytic system, this method achieves the formation of the key intermediate, 2-amino-4-hydroxy-6-chloromethylpteridine, with superior selectivity. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity actives while adhering to increasingly stringent environmental regulations. The structural integrity of the final product, as depicted below, remains consistent with international pharmacopoeia requirements, ensuring therapeutic efficacy without compromising on safety standards.
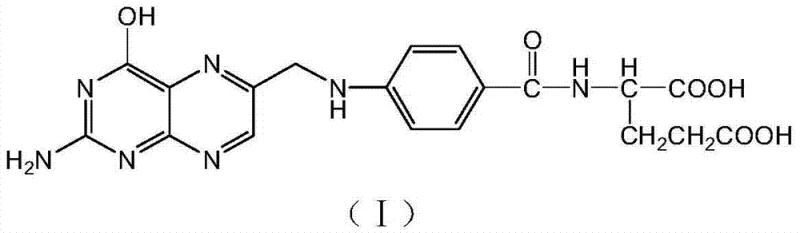
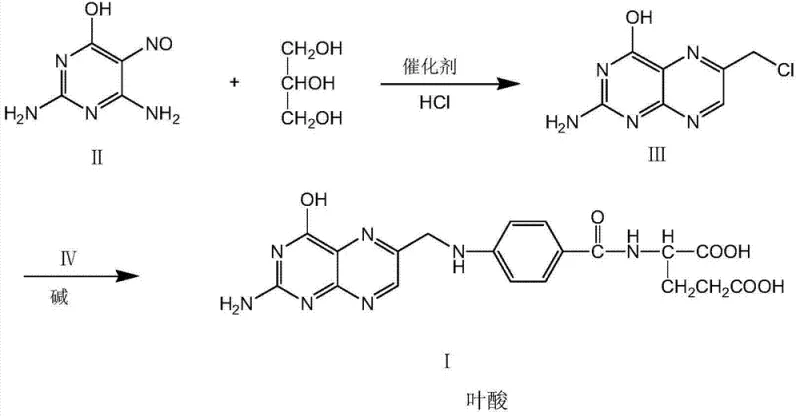
The transition to this novel methodology addresses long-standing inefficiencies inherent in conventional folic acid manufacturing. Historically, the industry has depended heavily on 1,1,3-trichloroacetone as a primary building block. However, obtaining high-purity 1,1,3-trichloroacetone is notoriously difficult; commercial grades often hover around 50% purity, contaminated with various dichloro- and tetrachloro-acetone isomers. These impurities participate in side reactions, generating stubborn byproducts like pteroic acid, which necessitates rigorous and waste-intensive acid and alkali refining processes to meet the European Pharmacopoeia limit of less than 0.6% pteroic acid. In stark contrast, the novel approach described in the patent bypasses these chlorinated ketones entirely. Instead, it employs a direct cyclization reaction between 2,4-diamino-5-nitroso-6-hydroxypyrimidine and glycerol. This strategic substitution not only eliminates the need for hazardous chlorination steps but also drastically simplifies the downstream processing. The reaction pathway, illustrated in the following scheme, demonstrates a streamlined progression from simple starting materials to the complex pteridine core, offering a robust solution for cost reduction in API manufacturing by minimizing raw material variability and waste treatment costs.
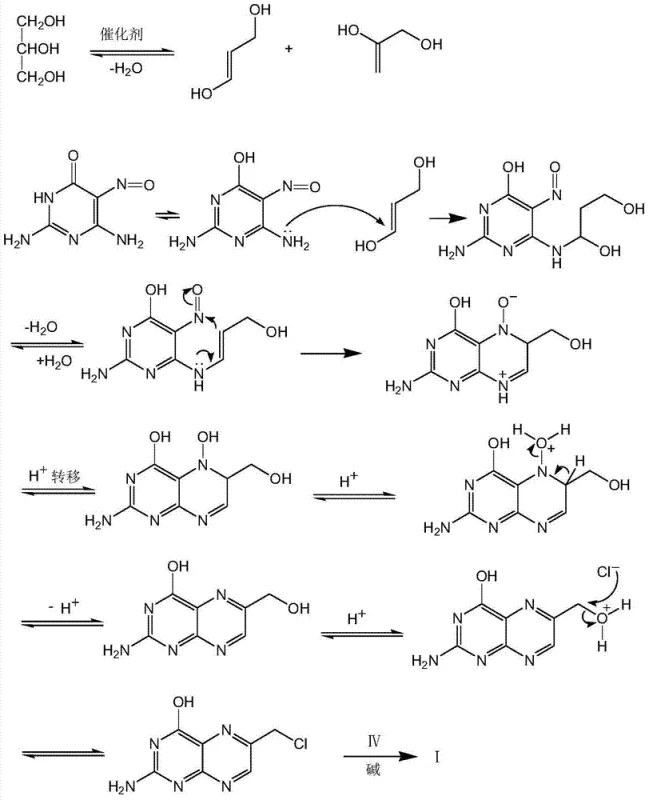
Understanding the mechanistic underpinnings of this glycerol-based cyclization is crucial for optimizing process parameters and ensuring batch-to-batch consistency. The reaction relies on a sophisticated interplay of dehydration and nucleophilic attack facilitated by a composite catalyst. Initially, glycerol undergoes acid-catalyzed dehydration to form reactive enol intermediates. These enols then engage with the amino groups of the pyrimidine derivative. The presence of a Lewis acid, such as cuprous chloride or zinc chloride, alongside a strong inorganic acid and a phase transfer catalyst, creates a highly active microenvironment that promotes ring closure. This multi-component catalytic system ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 6-chloromethylpteridine structure over potential isomers. Furthermore, the mechanism avoids the reduction of the nitroso group, a step often required in other pathways that can introduce additional impurities. The detailed electron movement and intermediate transformations, particularly the dehydration of glycerol and subsequent cyclization, are elucidated in the mechanism diagram below, providing valuable insights for process chemists aiming to replicate or scale this chemistry.
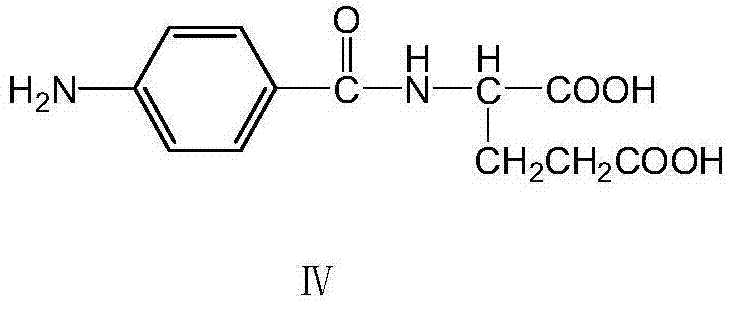
The coupling step involves the reaction of the generated chloromethylpteridine intermediate with p-aminobenzoyl-L-glutamic acid. This specific reactant, shown in the structure below, is critical for attaching the glutamic acid tail which is essential for the biological activity of folic acid. The reaction is conducted under mild alkaline conditions (pH 5-6) at moderate temperatures (40-45°C), which preserves the stereochemical integrity of the L-glutamic acid moiety while facilitating the nucleophilic substitution of the chloride. This gentle condition is a testament to the high reactivity of the intermediate generated by the glycerol route, allowing for efficient coupling without the harsh conditions that might degrade sensitive functional groups. The precise control of pH during this stage is vital to prevent hydrolysis of the intermediate while ensuring complete conversion to the final folate structure.
How to Synthesize Folic Acid Efficiently
The implementation of this green synthesis route requires careful attention to catalyst loading and temperature control to maximize yield and purity. The process begins with the cyclization step in a solvent such as 1,2-dichloroethane or chlorobenzene, where water formed during the reaction is continuously removed to drive the equilibrium forward. Following the formation of the pteridine intermediate, the reaction mixture is carefully neutralized before introducing the glutamic acid derivative. The final isolation involves a simple filtration and a slurry wash with aqueous ethanol, which effectively removes residual salts and organic impurities without the need for complex chromatographic separations. For detailed operational protocols and specific molar ratios optimized for pilot and commercial scales, please refer to the standardized guide below.
- Cyclization: React 2,4-diamino-5-nitroso-6-hydroxypyrimidine with glycerol in a solvent like 1,2-dichloroethane using a composite catalyst system (inorganic acid, Lewis acid, phase transfer catalyst) at reflux temperatures to form 2-amino-4-hydroxy-6-chloromethylpteridine.
- Condensation: Adjust the reaction mixture pH to 5-6 using alkali, then add p-aminobenzoyl-L-glutamic acid and heat to 40-45°C to facilitate the coupling reaction.
- Purification: Cool the reaction, pour into water, filter the crude product, and slurry wash with 50% ethanol at 50-60°C to obtain high-purity folic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this glycerol-based methodology offers profound strategic advantages beyond mere technical feasibility. The primary benefit lies in the stability and availability of the raw material supply. Glycerol is a ubiquitous commodity chemical, often produced as a byproduct of biodiesel manufacturing, ensuring a steady and cost-effective supply stream that is immune to the volatility associated with specialized chlorinated reagents. This shift significantly mitigates the risk of production delays caused by raw material shortages, thereby enhancing overall supply chain reliability for downstream pharmaceutical manufacturers. Moreover, the elimination of low-purity 1,1,3-trichloroacetone removes a major bottleneck in quality control, as incoming raw material testing becomes less critical and more predictable.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, driven primarily by the simplification of the purification workflow. Traditional methods require extensive acid and alkali refining cycles to remove impurities derived from trichloroacetone, which consumes large volumes of reagents and generates significant wastewater treatment costs. By avoiding these tedious refining steps, the new method drastically reduces utility consumption and waste disposal fees. Additionally, the higher selectivity of the reaction leads to improved crude product quality, which translates directly into higher overall yields and reduced loss of valuable intermediates during purification, contributing to a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like glycerol and standard solvents such as 1,2-dichloroethane ensures that the manufacturing process is not dependent on niche suppliers with limited capacity. This diversification of the supply base allows for greater flexibility in sourcing and reduces the lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and catalyst variations without significant loss of performance, further ensures consistent output even when minor fluctuations in raw material quality occur, securing a stable supply for global markets.
- Scalability and Environmental Compliance: As regulatory pressures mount regarding industrial emissions and waste generation, this process positions manufacturers favorably for future compliance. The reduction in wastewater volume and the avoidance of highly chlorinated organic waste streams simplify the environmental permitting process and lower the carbon footprint of the facility. The straightforward work-up procedure, involving simple filtration and solvent recovery, is inherently easier to scale from kilogram to multi-ton quantities compared to processes requiring complex extraction or crystallization sequences, facilitating rapid commercial scale-up of complex vitamin intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, aimed at clarifying the practical advantages for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production facilities.
Q: How does the glycerol route improve impurity profiles compared to traditional methods?
A: Traditional methods rely on 1,1,3-trichloroacetone which often contains significant impurities like 1,3-dichloroacetone and 1,1,3,3-tetrachloroacetone, leading to difficult-to-remove byproducts like pteroic acid. The glycerol route avoids these chlorinated ketones entirely, resulting in a cleaner reaction profile and easier purification to meet pharmacopoeia standards.
Q: What are the key catalysts used in this novel synthesis?
A: The process utilizes a sophisticated composite catalyst system comprising an inorganic acid (such as sulfuric or phosphoric acid combined with hydrochloric acid), a Lewis acid (like cuprous chloride or zinc chloride), and a phase transfer catalyst (such as tetrabutylammonium chloride) to drive the cyclization efficiently.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of cheap and easily available raw materials like glycerol and simplifies the operational steps by avoiding tedious acid and alkali refining processes required for crude products in older methods, making it highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Folic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry in reshaping the landscape of vitamin production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the glycerol-based folic acid synthesis are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards, providing our clients with the confidence needed to navigate complex regulatory environments.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this environmentally friendly method can enhance your operational efficiency and product quality.
