Revolutionizing Alpha-Carbonyl Amide Production: A Deep Dive into Copper-Catalyzed Oxidative Coupling for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access biologically active molecular scaffolds, and patent CN101735095A presents a transformative approach to synthesizing alpha-carbonyl amide compounds. This intellectual property discloses a novel synthetic methodology that leverages copper catalysis to drive the oxidative coupling of aryl alkynes and amines directly into alpha-carbonyl amides, bypassing the need for pre-oxidized precursors. Historically, the construction of this vital pharmacophore has been plagued by inefficient multi-step sequences, but this technology introduces a streamlined, atom-economical route that utilizes molecular oxygen as the terminal oxidant. For R&D directors and process chemists, this represents a significant leap forward in green chemistry, offering a robust platform for generating diverse libraries of bioactive molecules with reduced environmental impact and operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing alpha-carbonyl amides have long been hindered by significant logistical and economic bottlenecks that complicate large-scale manufacturing. As highlighted in the background art of the patent, conventional strategies often rely on the condensation of alpha-keto carboxylic acids with anilines, a process that necessitates the prior synthesis of these specialized acid derivatives through laborious oxidation steps. Alternative methods involving rearrangement reactions or the oxidation of alpha-hydroxy groups further exacerbate the problem by requiring harsh reaction conditions and generating substantial chemical waste. These legacy approaches suffer from poor atom economy and rely on starting materials that are not only expensive but often difficult to source in bulk quantities, creating fragile supply chains that are vulnerable to market fluctuations and delivery delays for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology described in CN101735095A revolutionizes the synthesis landscape by employing a direct oxidative coupling strategy that merges readily available aryl alkynes and amines in a single pot. This innovative route eliminates the need for pre-functionalized alpha-keto precursors, thereby drastically shortening the synthetic timeline and reducing the overall number of unit operations required. By utilizing abundant copper salts as catalysts and molecular oxygen or air as the oxidant, the process achieves high yields, with specific embodiments demonstrating isolated yields reaching up to 90% under optimized stoichiometric ratios. This paradigm shift not only simplifies the chemical transformation but also aligns perfectly with modern principles of sustainable manufacturing, offering a cost reduction in pharmaceutical intermediate manufacturing that is driven by fundamental process intensification rather than marginal tweaks.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The core of this technological breakthrough lies in the intricate catalytic cycle mediated by copper species, which facilitates the activation of the alkyne triple bond and its subsequent coupling with the amine nucleophile under oxidative conditions. The reaction mechanism likely involves the coordination of the copper catalyst to the alkyne, enhancing its electrophilicity and enabling nucleophilic attack by the amine to form an intermediate vinyl-copper species. Subsequent oxidation by molecular oxygen regenerates the active copper catalyst while installing the crucial carbonyl functionality at the alpha position, effectively transforming the alkyne carbon skeleton into the desired amide framework without the need for external stoichiometric oxidants like chromium or manganese reagents. This catalytic efficiency is further bolstered by the presence of additives such as pyridine and water, which act as ligands or proton shuttles to stabilize transition states and accelerate the turnover frequency of the catalytic cycle.
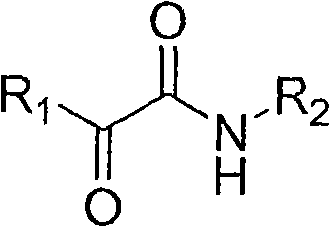
From an impurity control perspective, this mechanism offers distinct advantages over traditional pathways that often generate complex mixtures of over-oxidized by-products or rearrangement artifacts. The specificity of the copper-catalyzed oxidative coupling ensures that the reaction proceeds with high chemoselectivity, tolerating a wide range of functional groups on both the alkyne and amine substrates, including halogens, ethers, and esters. Experimental data from the patent indicates that varying the electronic nature of the substituents, such as using electron-donating methyl groups or electron-withdrawing fluorine atoms, allows for fine-tuning of the reaction kinetics without compromising the structural integrity of the final product. This robustness is critical for producing high-purity alpha-carbonyl amides, as it minimizes the formation of hard-to-remove impurities that could otherwise compromise the safety profile of downstream active pharmaceutical ingredients.
How to Synthesize Alpha-Carbonyl Amides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility, as detailed in the specific embodiments of the patent. The process typically involves charging a reaction vessel with the aryl alkyne and amine substrates alongside a copper catalyst such as copper(II) bromide, followed by the addition of an organic solvent like toluene and auxiliary agents including pyridine and water. The mixture is then subjected to an oxygen atmosphere at a controlled temperature, preferably around 60°C, for a duration of approximately 18 hours to ensure complete conversion. Detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are provided in the guide below to assist technical teams in replicating these results.
- Prepare the reaction mixture by combining aryl alkyne compounds, amine compounds, a copper catalyst (such as CuBr2), pyridine, and an organic solvent like toluene in a reaction vessel.
- Introduce an oxidizing agent, preferably oxygen gas at 1 atm pressure, and optionally add auxiliaries such as water or TEMPO to enhance yield and selectivity.
- Heat the mixture to a controlled temperature of approximately 60°C for 18 hours, followed by solvent evaporation and purification via silica gel column chromatography to isolate the pure alpha-carbonyl amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The primary driver of value is the substitution of exotic, multi-step precursors with commodity chemicals; aryl alkynes and anilines are produced on a massive global scale, ensuring a stable and competitive supply base that mitigates the risk of raw material shortages. Furthermore, the elimination of expensive precious metal catalysts in favor of abundant copper salts significantly lowers the bill of materials, while the use of oxygen as the oxidant removes the cost and safety hazards associated with handling hazardous stoichiometric oxidizing agents. These factors combine to create a manufacturing process that is not only more economical but also inherently safer and more resilient to supply chain disruptions.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, as it fundamentally alters the cost structure of alpha-carbonyl amide production by removing expensive intermediate isolation steps. By performing the synthesis in a direct one-pot fashion, manufacturers can realize substantial cost savings through reduced solvent consumption, lower energy usage due to mild reaction temperatures, and minimized waste disposal fees. The avoidance of heavy metal contaminants often associated with traditional oxidation methods also reduces the downstream burden of purification, leading to a more efficient overall process that enhances profit margins without sacrificing quality.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as phenylacetylene and substituted anilines ensures that production schedules are not held hostage by the lead times of niche chemical suppliers. The robustness of the reaction conditions, which tolerate variations in substrate quality and do not require stringent anhydrous environments, further contributes to operational stability. This reliability is crucial for maintaining continuous production flows in the fast-paced pharmaceutical sector, where reducing lead time for high-purity pharmaceutical intermediates can be the difference between meeting a market deadline and facing costly delays.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, requiring only standard pressure vessels and heating mantles, makes this process exceptionally easy to scale from kilogram to multi-ton quantities. The use of oxygen or air as the oxidant generates water as the primary by-product, aligning with increasingly strict environmental regulations regarding waste discharge and carbon footprint. This green chemistry profile not only facilitates regulatory approval but also enhances the corporate sustainability credentials of the manufacturer, making it an attractive partner for multinational corporations committed to responsible sourcing and eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative coupling technology, drawing directly from the experimental data and claims within the patent documentation. These insights are designed to clarify the operational feasibility and strategic value of adopting this novel synthetic route for your specific application needs. Understanding these nuances is essential for making informed decisions about process development and vendor selection in the competitive landscape of fine chemical manufacturing.
Q: What are the primary advantages of this copper-catalyzed method over traditional alpha-keto acid routes?
A: Traditional methods often rely on alpha-keto carboxylic acids or complex rearrangement reactions which require multi-step synthesis of difficult-to-obtain raw materials. This novel approach utilizes readily available aryl alkynes and amines directly, significantly simplifying the supply chain and reducing raw material costs while maintaining high yields up to 90% under optimized conditions.
Q: Is the copper catalyst system scalable for industrial production?
A: Yes, the process is highly amenable to scale-up. It operates at mild temperatures (preferably 60°C) and atmospheric pressure (1 atm oxygen), utilizing common solvents like toluene. The simplicity of the reaction equipment and the use of abundant copper salts rather than precious metals make it economically viable for commercial scale-up of complex pharmaceutical intermediates.
Q: How does the impurity profile compare to conventional synthesis routes?
A: The direct oxidative coupling mechanism minimizes the formation of by-products associated with multi-step precursor synthesis. By avoiding the isolation of unstable intermediates often found in rearrangement or hydroxyl oxidation methods, this route offers a cleaner reaction profile, facilitating easier purification and ensuring high-purity alpha-carbonyl amides suitable for sensitive downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed oxidative coupling technology described in CN101735095A and possess the technical expertise to bring this innovation to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in an industrial setting. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of alpha-carbonyl amide meets the exacting standards required for pharmaceutical applications, providing our partners with the confidence they need to integrate these intermediates into their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your project, along with specific COA data and route feasibility assessments that demonstrate our commitment to transparency and technical excellence. Let us partner with you to optimize your supply chain and accelerate your time to market with high-quality, cost-effective chemical solutions.
