Advanced Metal-Free Synthesis of Indoline Derivatives with C2 Quaternary Centers for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex molecular scaffolds, particularly those found in bioactive natural products. Patent CN109776388B introduces a groundbreaking synthetic methodology for constructing indoline derivatives featuring a C2 quaternary carbon center, a structural motif prevalent in potent alkaloids such as Mitragynine pseudoindoxyl and Trigonoliimine C. These compounds exhibit significant pharmacological activities, ranging from opioid agonism to anti-HIV properties, making their efficient synthesis a high priority for drug discovery programs. The disclosed technology leverages a unique mechanism involving the in situ formation of a Kukhtin-Ramirez adduct, enabling a streamlined, one-pot cyclization that bypasses the limitations of traditional transition-metal catalysis.
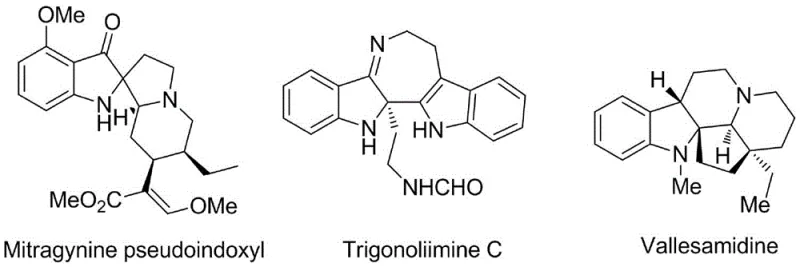
Historically, the construction of indoline cores with quaternary centers has relied heavily on transition-metal catalyzed processes, which often present significant hurdles for industrial application. Conventional strategies, such as those reported by Liang and Hu, typically utilize palladium or rhodium catalysts in conjunction with hazardous diazo compounds. For instance, rhodium-catalyzed decomposition of diazo esters requires precise control and generates toxic byproducts, while palladium-mediated cross-couplings often suffer from limited substrate scope and high catalyst costs. These methods not only increase the financial burden of production but also introduce complex purification challenges to remove trace heavy metals, a critical concern for regulatory compliance in active pharmaceutical ingredient (API) manufacturing.
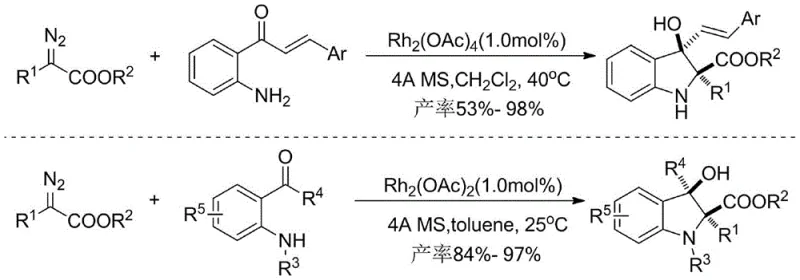
In stark contrast, the novel approach detailed in this patent offers a metal-free alternative that fundamentally reshapes the synthetic landscape for these valuable intermediates. By employing hexamethylphosphoramide as a key mediator alongside a mild base, the reaction proceeds through a nucleophilic insertion mechanism rather than a metal-carbene pathway. This shift eliminates the need for expensive noble metals and dangerous diazo reagents, replacing them with stable, commercially available alpha-keto esters and bifunctional amines. The result is a robust, two-step one-pot protocol that operates under mild conditions, typically starting at low temperatures and finishing at room temperature, thereby significantly enhancing the safety profile and operational simplicity for process chemists aiming for reliable pharmaceutical intermediate supplier status.
Mechanistic Insights into Metal-Free Intramolecular Cyclization
The core innovation of this technology lies in the generation and reactivity of the Kukhtin-Ramirez adduct, a phosphorus-containing intermediate that acts as a potent nucleophile. Upon mixing the alpha-keto ester with hexamethylphosphoramide, a zwitterionic species is formed which subsequently undergoes proton transfer and nucleophilic displacement. This activated intermediate then attacks the polar amino group within the bifunctional substrate, triggering an intramolecular cyclization that constructs the five-membered indoline ring with high stereocontrol. The mechanism avoids the formation of free carbenes, which are often responsible for non-selective side reactions and dimerization in traditional diazo chemistry, thus leading to cleaner reaction profiles and higher purity outputs.
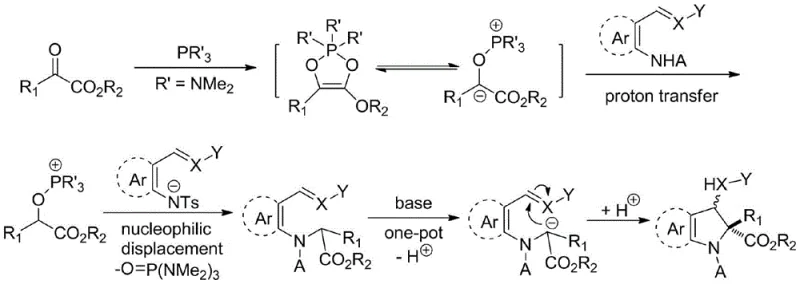
Furthermore, the impurity profile of this reaction is exceptionally manageable due to the mild basic conditions employed in the second step. The use of bases like cesium carbonate or DBU facilitates the final ring closure without promoting extensive decomposition of sensitive functional groups often present in complex drug candidates. The patent data indicates that electronic effects on the aromatic rings play a crucial role in yield optimization; for example, substrates with electron-donating groups at specific positions can achieve yields as high as 92%. This level of predictability allows R&D teams to design synthetic routes with greater confidence, knowing that the reaction tolerates a wide variety of substituents including halogens, alkoxy groups, and trifluoromethyl moieties without compromising the integrity of the C2 quaternary center.
How to Synthesize Indoline Derivatives Efficiently
Executing this synthesis requires careful attention to the order of addition and temperature control to maximize the formation of the key intermediate. The process begins by dissolving the alpha-keto ester and the bifunctional amine substrate in a dry organic solvent, preferably dichloromethane, under an inert atmosphere. Hexamethylphosphoramide is then added dropwise at cryogenic temperatures to ensure controlled adduct formation before warming the mixture to room temperature. Once the initial addition is complete, a stoichiometric amount of base is introduced to drive the cyclization to completion. Detailed standardized synthesis steps see the guide below.
- Dissolve alpha-carbonyl formate and bifunctional compound (e.g., o-aminochalcone) in an organic solvent like dichloromethane under nitrogen protection.
- Add hexamethylphosphoramide dropwise at low temperature (-78°C), then warm to room temperature and stir for 0.5 to 2 hours to form the intermediate adduct.
- Add a base such as cesium carbonate (Cs2CO3) to the reaction mixture and continue stirring at room temperature for 2 to 20 hours to complete the intramolecular cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this metal-free methodology offers transformative advantages over legacy technologies. The elimination of palladium and rhodium catalysts removes a major cost driver and supply chain bottleneck, as these precious metals are subject to significant price volatility and geopolitical sourcing risks. Additionally, the avoidance of diazo compounds, which often require specialized handling and storage due to their explosive nature, drastically reduces the safety infrastructure costs associated with manufacturing. This translates to a more resilient supply chain capable of delivering high-purity pharmaceutical intermediates with consistent quality and reduced lead times, ensuring continuity for downstream drug production.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the use of inexpensive, commodity-grade reagents like hexamethylphosphoramide and cesium carbonate significantly lowers the raw material cost per kilogram. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces solvent consumption and waste disposal costs. This economic efficiency makes the commercial scale-up of complex pharmaceutical intermediates much more viable, allowing manufacturers to offer competitive pricing without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, such as alpha-keto esters and o-aminobenzaldehyde derivatives, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental parameters, ensuring batch-to-b consistency. This reliability is critical for maintaining steady inventory levels and meeting the rigorous delivery schedules demanded by large-scale API manufacturers.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis minimizes the number of unit operations required, simplifying the transition from laboratory benchtop to pilot plant and full commercial production. The absence of heavy metals aligns perfectly with increasingly stringent environmental regulations regarding wastewater discharge and product residual limits. This green chemistry approach not only facilitates regulatory approval but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis platform. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical implementation of this technology for potential partners and clients.
Q: Does this synthesis method require transition metal catalysts?
A: No, this novel method described in patent CN109776388B is completely metal-free. It utilizes hexamethylphosphoramide and a base to mediate the reaction, eliminating the need for expensive palladium or rhodium catalysts and avoiding heavy metal residue issues in the final product.
Q: What is the typical yield range for these indoline derivatives?
A: The patent reports medium to high yields depending on the substrate. For optimized substrates, such as those with specific electron-donating groups, yields can reach as high as 92%. The process demonstrates robust performance across a wide range of substituted starting materials.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It employs a one-pot strategy with readily available raw materials and mild reaction conditions (room temperature cyclization). The absence of dangerous diazo compounds and expensive metals significantly simplifies the safety profile and cost structure for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes that balance efficiency with regulatory compliance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methodologies like this metal-free indoline synthesis can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity pharmaceutical intermediate meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your manufacturing budget.
