Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which significantly enhance metabolic stability and lipophilicity. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, utilizing a transition metal palladium-catalyzed carbonylation cascade reaction. This technology represents a significant leap forward for manufacturers aiming to produce high-purity pharmaceutical intermediates efficiently. By leveraging cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts, this process addresses critical bottlenecks in traditional heterocycle synthesis. The method operates under remarkably mild conditions, specifically at 30°C, ensuring safety and energy efficiency while maintaining high reaction yields up to 97% in optimized examples. For R&D directors and procurement managers, this patent offers a viable pathway to scale complex molecular architectures without the prohibitive costs associated with exotic reagents or extreme processing parameters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted nitrogen heterocycles has relied heavily on the use of specialized synthons like trifluorodiazoethane or direct fluorination techniques that often suffer from poor atom economy and safety hazards. Conventional routes frequently require harsh reaction conditions, including high temperatures and pressures, which can degrade sensitive functional groups and lead to complex impurity profiles that are difficult to separate. Furthermore, many traditional methods exhibit limited substrate scope, failing to accommodate diverse aryl substitutions necessary for modern drug discovery programs. The reliance on gaseous carbon monoxide from external cylinders in older carbonylation protocols introduces significant logistical and safety challenges for large-scale manufacturing facilities. These limitations collectively increase the cost of goods sold (COGS) and extend development timelines, creating a pressing need for more adaptable and safer synthetic strategies in the production of API intermediates.
The Novel Approach
The methodology disclosed in CN111423381A overcomes these hurdles through an elegant palladium-catalyzed cascade that constructs the imidazole core in a single pot. As illustrated in the general reaction scheme below, the process integrates trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts seamlessly.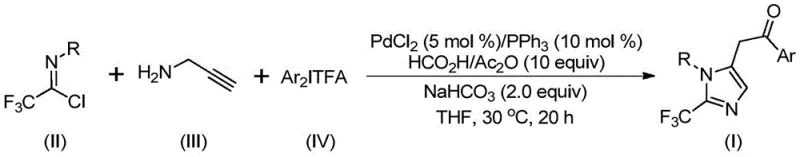
This novel approach utilizes an in situ generation of carbon monoxide from formic acid and acetic anhydride, effectively removing the need for handling toxic CO gas directly. The reaction proceeds with exceptional efficiency at a low temperature of 30°C over 16 to 24 hours, demonstrating remarkable functional group tolerance. The system accommodates various substituents on both the imidoyl chloride and the diaryliodonium salt components, including electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups such as nitro and halogens. This versatility allows chemists to access a wide library of diversified imidazole derivatives, such as compounds I-1 through I-5 shown below, which are crucial for structure-activity relationship (SAR) studies.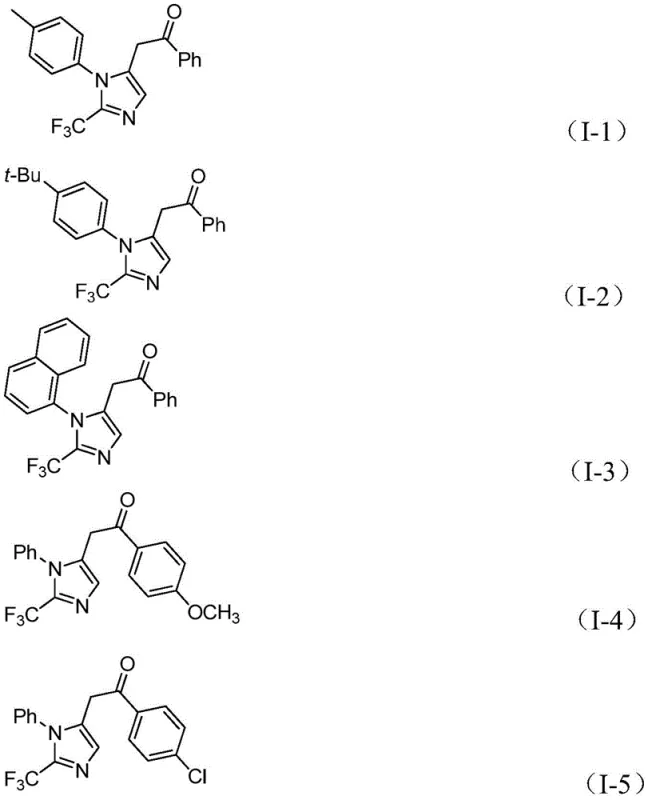
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway proposed in the patent involves a sophisticated sequence of organometallic transformations initiated by base-promoted intermolecular carbon-nitrogen bond formation. Initially, the reaction between trifluoroethylimidoyl chloride and propargylamine yields a trifluoroacetamidine intermediate, which subsequently undergoes isomerization. The palladium catalyst then facilitates alkynyl amine palladation to generate a vinyl-palladium species, followed by further isomerization to an alkyl-palladium intermediate. This critical step sets the stage for the subsequent carbonylation event, where carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond to form an acyl-palladium complex. The cycle concludes with the oxidative addition of the diaryliodonium salt to form a tetravalent palladium intermediate, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
From a quality control perspective, understanding this mechanism is vital for impurity management. The use of mild conditions (30°C) minimizes thermal degradation pathways that often plague high-temperature syntheses. Furthermore, the specific stoichiometry of reagents, such as the excess of trifluoroethylimidoyl chloride relative to propargylamine, drives the equilibrium towards the desired product while suppressing side reactions. The choice of tetrahydrofuran (THF) as the preferred solvent ensures optimal solubility of all polar and non-polar components, facilitating homogeneous catalysis and consistent reaction kinetics. This deep mechanistic understanding allows process chemists to fine-tune parameters like catalyst loading (PdCl2/PPh3) and additive ratios to maximize yield and purity, ensuring the final API intermediate meets stringent regulatory specifications for commercial distribution.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory optimization and industrial scale-up. The procedure involves charging a reactor with the palladium catalyst system, additives, and substrates in an organic solvent, followed by heating to a modest 30°C. Detailed standardized synthesis steps, including precise molar ratios and workup procedures involving silica gel filtration and column chromatography, are outlined in the technical guide below to ensure reproducibility and high yield.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this patented technology are substantial. The shift towards this palladium-catalyzed method eliminates the dependency on hazardous high-pressure gas infrastructure and expensive specialized fluorinating agents, leading to significant operational cost reductions. The reliance on commercially available starting materials like aromatic amines and propargylamine ensures a stable and resilient supply chain, mitigating risks associated with raw material shortages. Additionally, the mild reaction conditions reduce energy consumption and equipment wear, contributing to a lower overall carbon footprint and enhanced environmental compliance.
- Cost Reduction in Manufacturing: The utilization of inexpensive palladium chloride and triphenylphosphine as the catalytic system, combined with the avoidance of high-pressure CO gas, drastically lowers capital expenditure and operating costs. The ability to run reactions at 30°C instead of elevated temperatures reduces energy bills and extends the lifespan of reactor vessels. Furthermore, the high atom economy and yield minimize waste disposal costs, providing a clear financial advantage over legacy synthetic routes that generate substantial byproduct streams.
- Enhanced Supply Chain Reliability: The starting materials, including trifluoroethylimidoyl chloride and diaryliodonium salts, are derived from widely available commodity chemicals such as aromatic amines and aryl iodides. This broadens the supplier base and reduces the risk of single-source dependency. The robustness of the reaction across diverse substrates means that supply chain disruptions for specific precursors can often be managed by switching to alternative analogs without re-validating the entire process, ensuring continuous production flow for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable to gram levels with potential for multi-kilogram production, supported by simple post-processing steps like filtration and chromatography. The use of in situ CO generation removes the regulatory burden and safety risks associated with storing and transporting toxic carbon monoxide cylinders. This aligns with modern green chemistry principles, facilitating easier permitting and reducing the environmental impact of manufacturing operations, which is increasingly important for maintaining social license to operate in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method, focusing on practical aspects of catalyst performance, substrate scope, and safety protocols. These insights are derived directly from the experimental data and embodiments provided in the patent documentation to assist technical teams in evaluating feasibility.
Q: What is the optimal temperature for this palladium-catalyzed carbonylation?
A: The patent specifies a mild reaction temperature of 30°C, which balances reaction efficiency with energy consumption and safety.
Q: Can this method tolerate diverse functional groups on the aryl rings?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating substituents such as methyl, tert-butyl, halogens, trifluoromethyl, and nitro groups.
Q: What serves as the carbon monoxide source in this protocol?
A: Carbon monoxide is generated in situ from a mixture of formic acid and acetic anhydride, eliminating the need for hazardous high-pressure CO gas cylinders.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111423381A for developing next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl imidazole intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and accelerate your time to market.
