Advanced Cuprous Complex Catalyst Enables Efficient One-Pot Benzimidazole Synthesis
Advanced Cuprous Complex Catalyst Enables Efficient One-Pot Benzimidazole Synthesis
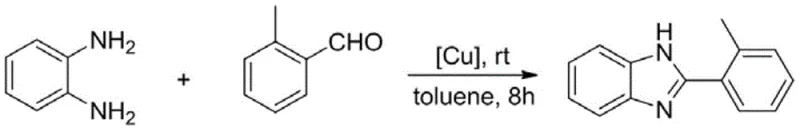
The pharmaceutical and agrochemical industries are constantly seeking robust catalytic systems that can streamline the production of high-value heterocyclic scaffolds. Patent CN111732600A introduces a groundbreaking advancement in this domain by disclosing a novel cuprous complex containing a meta-carborane ligand. This specific catalyst architecture addresses long-standing challenges in the synthesis of benzimidazole derivatives, which are pivotal structures in numerous bioactive molecules. Unlike traditional methods that rely on harsh acidic conditions or expensive noble metals, this innovation leverages the unique electronic and steric properties of the carborane cage to stabilize the copper center. The result is a highly efficient, air-stable catalyst capable of driving one-pot condensation reactions between o-phenylenediamines and aldehydes under remarkably mild conditions. For R&D directors and procurement specialists, this technology represents a significant opportunity to optimize synthetic routes, reduce raw material costs, and enhance the overall sustainability of manufacturing processes for key pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole derivatives has been plagued by significant operational inefficiencies and environmental concerns. Traditional protocols often necessitate the use of strong mineral acids to facilitate cyclization and dehydration, leading to corrosive waste streams and requiring specialized equipment resistant to acid attack. Furthermore, alternative oxidative methods utilizing oxidants frequently generate substantial amounts of toxic by-products, complicating downstream purification and increasing the burden on waste treatment facilities. More modern approaches have attempted to utilize gold catalysis or microwave-assisted cuprous salt catalysis; however, these methods introduce their own set of limitations. Gold catalysts are economically prohibitive for large-scale production due to the high cost of the precious metal, while microwave-assisted reactions often suffer from poor scalability and require specialized energy-intensive infrastructure. Additionally, many existing copper catalysts lack stability, being highly sensitive to air and moisture, which necessitates rigorous inert atmosphere handling and increases operational complexity.
The Novel Approach
The methodology outlined in patent CN111732600A offers a transformative solution by employing a specifically designed meta-carborane ligated cuprous complex. This novel catalyst enables the direct condensation of readily available o-phenylenediamines and various aldehyde compounds in a simple one-pot procedure. The reaction proceeds efficiently at room temperature in common organic solvents like toluene, eliminating the need for external heating or microwave irradiation. This mildness not only preserves energy but also expands the substrate scope to include sensitive functional groups that might degrade under harsher conditions. The process demonstrates exceptional universality, accommodating aromatic aldehydes with electron-donating or electron-withdrawing substituents, as well as aliphatic aldehydes, with reported yields ranging from 86% to 97%. By replacing expensive noble metals with a stable, reusable copper system and avoiding strong acids, this approach drastically simplifies the workflow and reduces the environmental footprint of benzimidazole production.
Mechanistic Insights into Meta-Carborane Ligand Stabilized Catalysis
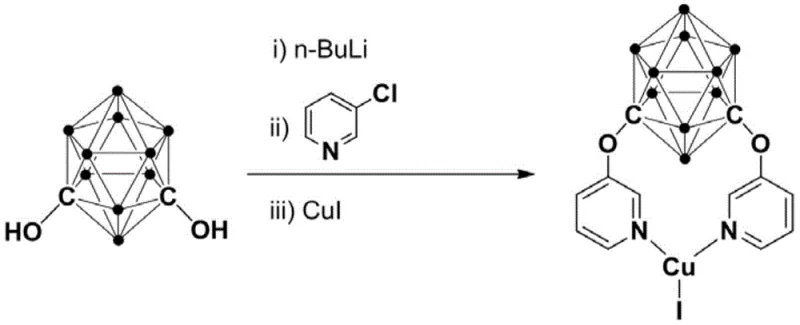
The superior performance of this catalytic system is fundamentally rooted in the unique structural attributes of the meta-carborane ligand. The carborane cage acts as a robust three-dimensional scaffold that imparts significant steric bulk around the copper center, effectively shielding it from deactivation pathways such as aggregation or oxidation by atmospheric oxygen. This steric protection is complemented by the electron-withdrawing nature of the boron cluster, which modulates the Lewis acidity of the copper ion, optimizing its ability to coordinate with the nitrogen atoms of the o-phenylenediamine substrate. The synthesis of the catalyst itself involves a precise sequence of organometallic transformations, starting with the deprotonation of meta-carborane diphenol using n-BuLi at cryogenic temperatures to ensure regioselectivity. Subsequent nucleophilic substitution with 3-chloropyridine installs the necessary nitrogen donor arms, which chelate the copper iodide to form the final active species. This rational design ensures that the catalyst remains homogeneous and active throughout the reaction cycle, facilitating the activation of the aldehyde carbonyl group and the subsequent nucleophilic attack by the amine.
Impurity control in this catalytic system is inherently managed by the high selectivity of the copper complex. Because the reaction conditions are neutral and mild, side reactions such as polymerization of the aldehyde or over-oxidation of the diamine are minimized. The catalyst's stability against air and water means that strict anhydrous conditions are not required, reducing the risk of hydrolysis-related impurities that often plague sensitive organometallic reactions. Furthermore, the clean reaction profile allows for straightforward isolation of the product, often requiring only simple concentration and column chromatography or recrystallization. This high level of chemoselectivity is crucial for pharmaceutical applications where regulatory limits on impurities are stringent. The robust nature of the meta-carborane ligand ensures that the catalyst does not decompose into free copper ions that could contaminate the final API, thereby simplifying the metal removal process and ensuring compliance with heavy metal specifications.
How to Synthesize Cuprous Complex Efficiently
The preparation of this high-performance catalyst follows a streamlined protocol that is amenable to standard laboratory and pilot plant setups. The process begins with the careful handling of n-BuLi to generate the lithiated carborane intermediate, followed by the introduction of the pyridine linker and finally the copper source. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Deprotonate meta-carborane diphenol with n-BuLi at low temperature (-80 to -75°C) in THF/n-hexane.
- Warm to room temperature and react with 3-chloropyridine for nucleophilic substitution.
- Add CuI to the reaction mixture to form the final cuprous complex, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers compelling economic and logistical benefits. The shift from precious metal catalysts to a copper-based system immediately alleviates the cost volatility associated with gold or palladium markets. Moreover, the ability to run reactions at room temperature significantly reduces energy consumption compared to thermal or microwave-assisted processes, contributing to lower utility costs and a smaller carbon footprint. The use of commodity chemicals like o-phenylenediamine and substituted benzaldehydes as starting materials ensures a reliable and diverse supply base, mitigating the risk of raw material shortages. The simplified workup procedure, which avoids extensive aqueous washes required for acid neutralization, reduces solvent usage and waste disposal costs, further enhancing the overall cost-efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts results in substantial direct material cost savings. Additionally, the mild reaction conditions negate the need for energy-intensive heating or specialized microwave equipment, leading to reduced operational expenditures. The high catalytic efficiency means that lower catalyst loadings can be utilized while maintaining high yields, optimizing the cost-per-kilogram of the final product. By minimizing the formation of by-products, the downstream purification burden is lessened, reducing the consumption of silica gel and solvents required for chromatography.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as copper iodide, n-BuLi, and common aldehydes ensures a resilient supply chain that is not dependent on single-source specialty reagents. The catalyst's stability towards air and moisture simplifies storage and transportation logistics, removing the need for complex inert gas packaging or cold chain shipping. This robustness allows for greater flexibility in inventory management and reduces the risk of production delays caused by reagent degradation. Consequently, manufacturers can maintain consistent production schedules and meet tight delivery deadlines with greater confidence.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis minimizes unit operations, making the process inherently easier to scale from kilogram to multi-ton production levels. The absence of strong acids and toxic oxidants aligns with green chemistry principles, facilitating easier regulatory approval and reducing the environmental compliance burden. Waste generation is significantly curtailed due to the high atom economy and selectivity of the reaction, lowering the costs associated with hazardous waste treatment. This environmentally friendly profile enhances the corporate sustainability metrics of the manufacturing entity, appealing to eco-conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: Why is the meta-carborane ligand significant for catalyst stability?
A: The meta-carborane cage provides exceptional steric bulk and electron-withdrawing properties, rendering the cuprous center insensitive to air and moisture, which is critical for industrial handling.
Q: What are the primary advantages over traditional gold-catalyzed methods?
A: Unlike gold catalysts which are prohibitively expensive, this cuprous complex uses abundant copper, operates at room temperature without microwave assistance, and avoids strong acidic conditions.
Q: Is this catalyst suitable for large-scale pharmaceutical manufacturing?
A: Yes, the protocol uses cheap substrates like o-phenylenediamine and aldehydes, generates minimal waste, and achieves high yields (86-97%) under mild conditions, facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cuprous Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the meta-carborane cuprous complex in modern pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate delivered meets the highest industry standards. We are committed to supporting our clients in navigating the complexities of process development, offering tailored solutions that maximize efficiency and minimize risk.
We invite you to collaborate with our technical procurement team to explore how this novel catalyst can optimize your specific synthetic routes. Contact us today to request a Customized Cost-Saving Analysis tailored to your production volume and requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us be your partner in achieving cost-effective, sustainable, and high-quality chemical manufacturing.
