Scalable Photocatalytic Synthesis of High-Purity 1,2,4-Triazole Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust pathways for constructing nitrogen-containing heterocycles, particularly 1,2,4-triazoles, due to their pervasive presence in bioactive molecules ranging from antifungals to anticancer agents. Patent CN111518042B introduces a groundbreaking approach to synthesizing these critical scaffolds through a visible-light-driven photocatalytic cyclization. Unlike traditional thermal methods that often demand harsh conditions, this technology leverages photon energy to drive the reaction at mild temperatures between 50-80°C. This shift not only enhances safety profiles by eliminating the need for explosive azide reagents but also opens new avenues for functional group tolerance. For R&D teams and procurement specialists, this represents a significant evolution in accessing high-purity pharmaceutical intermediates with improved process safety and operational simplicity.
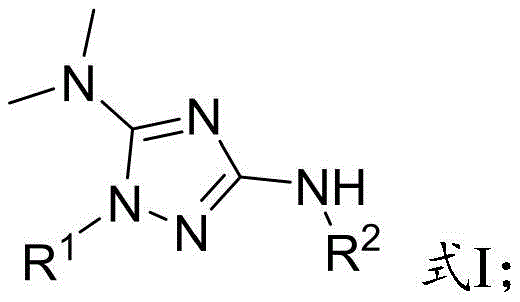
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazole frameworks has heavily relied on the use of azide compounds as key building blocks. While effective in forming the triazole ring, azides are notoriously unstable and possess high explosive potential, posing severe safety risks during storage, transport, and large-scale manufacturing. Furthermore, conventional thermal cyclizations often require elevated temperatures and strong acidic or basic conditions, which can lead to the degradation of sensitive functional groups on the substrate. These harsh parameters frequently result in complex impurity profiles, necessitating extensive and costly downstream purification steps. For supply chain managers, the reliance on hazardous raw materials translates into higher insurance costs, stricter regulatory compliance burdens, and potential disruptions in logistics due to safety classifications.
The Novel Approach
The methodology disclosed in CN111518042B fundamentally reimagines the synthetic strategy by replacing hazardous azides with stable hydrazine and isothiocyanate precursors. By utilizing visible light irradiation in the presence of a photocatalyst, the reaction proceeds through a radical-mediated mechanism that is both energetically efficient and chemically selective. This photochemical approach operates under remarkably mild conditions, typically requiring temperatures no higher than 80°C, which preserves the integrity of delicate substituents. The elimination of explosive reagents drastically reduces the safety footprint of the manufacturing process. Additionally, the use of commercially available organic dyes or metal complexes as catalysts ensures that the process remains cost-effective and scalable, addressing the critical needs of industrial production without compromising on yield or purity.
Mechanistic Insights into Photocatalytic Radical Cyclization
The core of this innovation lies in the intricate photocatalytic cycle that facilitates the formation of the triazole ring. Upon irradiation, the photocatalyst, such as Rose Bengal or a Ruthenium complex, absorbs photon energy to reach an excited state. This excited species interacts with the thiourea intermediate, formed in situ from the reaction of hydrazine and isothiocyanate, inducing the dissociation of the C-N bond to generate a thiourea radical. Subsequently, this radical species undergoes oxidation, potentially mediated by oxygen or the catalyst itself, to form a radical cation. This highly reactive intermediate then engages in an intramolecular cyclization with the adjacent nitrogen center, closing the triazole ring. This mechanism avoids the high-energy transition states associated with thermal cycloadditions, allowing the reaction to proceed smoothly at lower temperatures while maintaining high regioselectivity.
From an impurity control perspective, this radical pathway offers distinct advantages over ionic mechanisms. The mild reaction environment minimizes side reactions such as hydrolysis or polymerization that are common under harsh thermal conditions. The specific interaction between the photocatalyst and the substrate ensures that the cyclization occurs preferentially at the desired position, leading to a cleaner crude reaction profile. This inherent selectivity reduces the burden on purification teams, as fewer byproducts are generated that mimic the polarity of the target molecule. For quality control departments, this translates to a more consistent impurity spectrum, facilitating easier validation and regulatory filing for the final active pharmaceutical ingredient derived from these intermediates.
How to Synthesize 1,2,4-Triazole Derivatives Efficiently
Implementing this photocatalytic protocol requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of a homogeneous mixture containing the hydrazine compound, isothiocyanate derivative, tetramethylguanidine, and the chosen photocatalyst in a polar organic solvent such as acetonitrile or DMSO. The molar ratios are critical, with a preferred stoichiometry of 1:1:1.5 for the hydrazine, isothiocyanate, and base respectively. Once the solution is prepared, it is subjected to light irradiation with a wavelength range of 200-1000 nm, with blue light (around 465 nm) often proving optimal. The reaction is maintained at a controlled temperature, typically around 60°C, for a duration ranging from 6 to 24 hours depending on the specific substituents. Detailed standardized synthesis steps are provided in the guide below.
- Mix hydrazine compound, isothiocyanate compound, tetramethylguanidine, and a photocatalyst (e.g., Rose Bengal) in a polar organic solvent.
- Subject the mixture to illumination (200-1000 nm) and maintain temperature between 50-80°C for cyclization.
- Purify the resulting reaction liquid via column chromatography using petroleum ether and ethyl acetate to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers substantial strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain. By eliminating the need for specialized, hazard-classified azide reagents, companies can source standard hydrazines and isothiocyanates from a broader vendor base, enhancing supply security and reducing raw material costs. Furthermore, the mild reaction conditions significantly lower energy consumption compared to high-temperature thermal processes, contributing to overall manufacturing cost reduction. The enhanced safety profile also reduces facility maintenance costs related to explosion-proof infrastructure and lowers insurance premiums, providing a clear financial upside for long-term production planning.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous azide reagents directly lowers the bill of materials. Additionally, the use of low-loading photocatalysts (0.5-2 mol%) and the ability to operate at near-ambient temperatures reduces utility costs associated with heating and cooling. The simplified workup procedure, often requiring only standard column chromatography, minimizes solvent usage and waste disposal fees. These factors combine to create a leaner, more cost-efficient manufacturing process that improves margin potential for high-volume commercial production.
- Enhanced Supply Chain Reliability: Sourcing stable hydrazine and isothiocyanate precursors is significantly more reliable than managing the logistics of explosive azides. This stability allows for larger batch sizes and longer storage times without degradation, enabling manufacturers to build strategic inventory buffers. The robustness of the photocatalytic method against minor fluctuations in reaction conditions further ensures consistent output, reducing the risk of batch failures that can disrupt downstream drug formulation schedules. This reliability is crucial for maintaining continuous supply to global pharmaceutical partners.
- Scalability and Environmental Compliance: The photochemical nature of this reaction is inherently scalable using modern flow chemistry reactors or large-scale batch photoreactors. The process generates minimal hazardous waste, aligning with green chemistry principles and increasingly strict environmental regulations. The absence of heavy metal catalysts in some embodiments (using organic dyes like Rose Bengal) further simplifies the removal of metal residues, ensuring the final product meets stringent limits for elemental impurities. This environmental compatibility facilitates smoother regulatory approvals and enhances the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic triazole synthesis. These insights are derived directly from the experimental data and claims within patent CN111518042B, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D directors assessing process feasibility and procurement teams evaluating vendor capabilities.
Q: Why is this photocatalytic method safer than conventional triazole synthesis?
A: Conventional methods often rely on explosive azide reagents at room temperature. This patent utilizes stable hydrazine and isothiocyanate precursors under mild light irradiation, significantly reducing explosion risks and handling hazards.
Q: What photocatalysts are compatible with this synthesis route?
A: The process supports both organic dyes such as Rose Bengal, Eosin B, and Eosin Y, as well as metal complexes like tris(2,2'-bipyridyl)ruthenium dichloride, offering flexibility in catalyst sourcing.
Q: What purity levels can be achieved with this purification method?
A: Through column chromatography purification using petroleum ether and ethyl acetate, the method consistently achieves purity levels between 98.5% and 99.9%, suitable for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111518042B can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole intermediate delivered meets the highest global standards for safety and efficacy.
We invite you to collaborate with our technical team to explore how this novel synthesis route can optimize your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate the purity of our intermediates, our experts are ready to assist. Contact our technical procurement team today to request route feasibility assessments and discover how our commitment to innovation and quality can drive value for your organization.
