Advanced Non-Metal Photocatalytic Strategy for High-Purity Nitrogen Heterocycle Manufacturing and Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally benign methods for constructing complex nitrogen-containing heterocyclic scaffolds, which serve as critical structural motifs in countless bioactive molecules. Patent CN109134362B introduces a groundbreaking methodology for introducing nitrogen heterocycles through the decarboxylation of carboxylates utilizing light-induced non-metal catalytic activity. This innovation represents a significant paradigm shift from traditional transition-metal-catalyzed cross-coupling reactions, offering a pathway that is not only chemically efficient but also economically and environmentally superior. By leveraging visible light photocatalysis with simple iodide salts and phosphine ligands, this technology enables the direct functionalization of quinolines, isoquinolines, and related heterocycles under exceptionally mild conditions. The ability to achieve high-efficiency catalytic conversion at room temperature without the need for precious metals addresses two of the most persistent pain points in fine chemical manufacturing: cost control and product purity compliance.
For R&D directors and process chemists, the implications of this technology are profound. The method utilizes active carboxylic esters, specifically N-(acyloxy)phthalimides, which undergo single-electron transfer upon photoexcitation to generate alkyl radicals. These radicals subsequently engage in a Minisci-type addition with protonated nitrogen heterocycles. This mechanism bypasses the need for pre-functionalized halides or organometallic reagents, streamlining the synthetic route and reducing the number of unit operations required to reach the target molecule. Furthermore, the broad substrate scope documented in the patent suggests that this protocol can be applied to a diverse array of complex intermediates, facilitating rapid library synthesis for drug discovery programs while maintaining a green chemistry profile that aligns with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of nitrogen-containing heterocycles has relied heavily on transition-metal catalysis, particularly using expensive noble metals such as iridium, ruthenium, or palladium. While these methods are effective, they introduce significant logistical and financial burdens to the manufacturing supply chain. The high cost of noble metal catalysts directly impacts the bill of materials, and more critically, the removal of trace metal residues from the final active pharmaceutical ingredient (API) requires rigorous and often costly purification steps. Regulatory agencies impose strict limits on heavy metal content in drug substances, necessitating additional processing such as scavenging resins or repeated recrystallizations, which inevitably lower overall yield and extend production timelines. Additionally, many traditional photoredox methods require specialized equipment or harsh reaction conditions that complicate scale-up efforts and increase energy consumption, making them less attractive for large-volume commercial production.
The Novel Approach
The methodology described in Patent CN109134362B offers a compelling alternative by replacing noble metal photocatalysts with a simple, non-metallic system comprising an iodide salt (such as sodium iodide) and a phosphine ligand (such as triphenylphosphine). This substitution fundamentally alters the economic equation of heterocycle synthesis. The reagents are commodity chemicals that are readily available in bulk quantities at a fraction of the cost of organometallic complexes. Moreover, the absence of heavy metals simplifies the downstream processing significantly; there is no need for specialized metal scavenging, reducing both the time and solvent usage associated with purification. The reaction proceeds efficiently under blue LED irradiation at room temperature, utilizing common organic solvents like acetone or acetonitrile. This mildness ensures excellent functional group compatibility, allowing sensitive moieties to survive the reaction conditions, which is crucial for late-stage functionalization of complex drug candidates.
Mechanistic Insights into Light-Induced Non-Metal Catalytic Decarboxylation
The core of this innovation lies in the unique interaction between the iodide salt, the phosphine ligand, and the light source to generate a reactive catalytic species capable of driving the decarboxylative coupling. Upon irradiation with blue light (wavelength range 365 nm to 500 nm, optimally 440-456 nm), the system facilitates a single-electron transfer process that activates the N-(acyloxy)phthalimide ester. This activation leads to the fragmentation of the ester bond, releasing carbon dioxide and generating a nucleophilic alkyl radical. This radical species is highly reactive and selectively attacks the electron-deficient position of the protonated nitrogen heterocycle, which is activated by the presence of an acid additive such as trifluoroacetic acid. The resulting radical cation intermediate is then oxidized to restore aromaticity, yielding the alkylated heterocyclic product. This mechanistic pathway is distinct from traditional Minisci reactions that often require stoichiometric oxidants or silver salts, offering a cleaner and more atom-economical route.
From a quality control perspective, the mechanism ensures high selectivity and minimizes the formation of unwanted byproducts. The use of simple iodide salts avoids the introduction of complex ligand frameworks that could degrade into difficult-to-remove impurities. The patent data demonstrates that this system tolerates a wide variety of functional groups, including esters, ethers, halogens, and protected amines, without compromising the efficiency of the radical generation or the coupling step. For instance, substrates containing Boc-protected amines or thioethers remain intact throughout the reaction, highlighting the chemoselectivity of the process. This robustness is essential for R&D teams aiming to synthesize diverse analogues for structure-activity relationship (SAR) studies, as it reduces the need for extensive protecting group strategies and allows for more convergent synthetic designs.
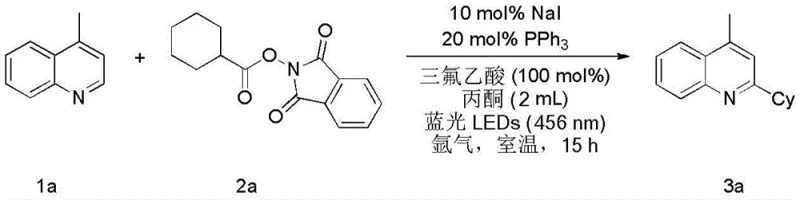
How to Synthesize 2-Cyclohexyl-4-methylquinoline Efficiently
To implement this technology in a laboratory setting, the procedure involves mixing the nitrogen heterocycle, the active carboxylate ester, sodium iodide, and triphenylphosphine in an organic solvent under an inert atmosphere. The reaction is initiated by exposure to blue LED light and stirred at room temperature for a defined period, typically ranging from 10 to 20 hours depending on the specific substrate reactivity. Following the reaction, standard aqueous workup and chromatographic purification yield the desired product with high purity. The detailed standardized synthesis steps for this specific transformation are outlined below, providing a clear roadmap for replication and optimization.
- In a Schlenk reaction tube, combine sodium iodide (10 mol%), triphenylphosphine (20 mol%), and the active carboxylate ester (1.5 equivalents) under an inert argon atmosphere.
- Add the nitrogen-containing heterocyclic compound (1 equivalent), trifluoroacetic acid (1 equivalent), and anhydrous acetone as the solvent to the reaction mixture.
- Irradiate the reaction system with blue LEDs (456 nm) at room temperature with continuous stirring for 15 hours, then quench with water and extract with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this non-metal photocatalytic technology presents a strategic opportunity to optimize manufacturing costs and enhance supply reliability. The most immediate impact is seen in the raw material expenditure; by eliminating the dependency on volatile and expensive noble metal markets, companies can stabilize their cost of goods sold (COGS). The reagents used in this process, such as sodium iodide and triphenylphosphine, are produced on a massive industrial scale for other applications, ensuring a stable and competitive supply market. This reduces the risk of supply chain disruptions that are often associated with specialized catalytic reagents sourced from limited suppliers. Furthermore, the simplified purification process translates directly into reduced solvent consumption and waste generation, lowering the environmental compliance costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts removes a significant cost driver from the production budget. Without the need for expensive iridium or ruthenium complexes, the direct material cost per kilogram of product is drastically lowered. Additionally, the removal of metal scavenging steps reduces the consumption of auxiliary materials and labor hours required for purification. The mild reaction conditions also mean lower energy costs, as the process does not require heating or cooling beyond ambient temperature control, contributing to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like iodide salts and common phosphine ligands mitigates the risk of supply bottlenecks. Unlike specialized organometallic catalysts that may have long lead times or limited availability, the key components of this reaction are widely stocked by global chemical distributors. This ensures consistent production schedules and reduces the likelihood of delays caused by raw material shortages. The robustness of the reaction also means that variations in reagent quality are less likely to impact the final outcome, providing greater stability in the supply chain.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of gram-scale amplification, indicating that the chemistry is robust enough for larger production runs. The use of common solvents like acetone and acetonitrile simplifies solvent recovery and recycling processes. Moreover, the absence of heavy metals simplifies the regulatory filing process for new drug applications, as there is no need to validate complex metal clearance protocols. This aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams evaluating this technology for integration into their existing workflows.
Q: What is the primary advantage of this photocatalytic method over traditional Minisci reactions?
A: The primary advantage is the elimination of expensive noble metal catalysts like iridium or ruthenium. This method utilizes simple iodide salts and phosphine ligands, which significantly reduces raw material costs and eliminates the risk of toxic heavy metal residues in the final pharmaceutical product.
Q: Does this process require specialized high-energy equipment?
A: No, the process operates efficiently at room temperature using standard blue LED light sources (456 nm). It does not require high-pressure reactors or extreme thermal conditions, making it compatible with standard laboratory and pilot plant glassware.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful gram-scale amplification experiments with high conversion rates. The mild reaction conditions and simple workup procedures involving standard extraction and chromatography indicate strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in modern pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN109134362B can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with specialized photochemical reactors capable of handling large-scale irradiation processes, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that moving from bench-scale to commercial production requires not just chemical expertise but also deep engineering knowledge, which our team delivers consistently.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this metal-free approach can optimize your budget. Please contact us to request specific COA data for similar heterocyclic intermediates and comprehensive route feasibility assessments. Let us help you accelerate your development timeline while reducing your environmental impact and production costs.
