Scalable Synthesis of Prasugrel Intermediate: A Technical Breakthrough for Global API Manufacturing
Scalable Synthesis of Prasugrel Intermediate: A Technical Breakthrough for Global API Manufacturing
The pharmaceutical landscape for antiplatelet agents continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes for critical cardiovascular drugs. Patent CN101948479A introduces a transformative methodology for the synthesis of a key Prasugrel intermediate, designated as Structure (I). This innovation addresses long-standing bottlenecks in the production of this vital therapeutic agent by establishing a robust, four-step synthetic pathway that includes alkylation, borylation, deprotection, and coupling reactions. Unlike conventional methods that suffer from sluggish kinetics and complex purification requirements, this novel approach leverages accessible raw materials and streamlined operational protocols to achieve superior yields. For R&D directors and supply chain strategists, this represents a significant opportunity to optimize the manufacturing footprint of antiplatelet medications while adhering to stringent quality standards.
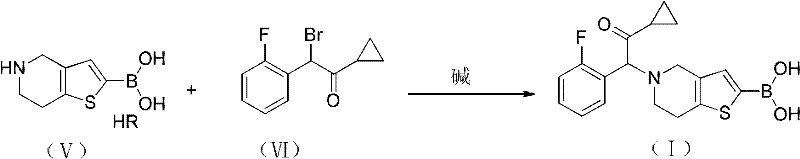
The strategic implementation of this synthetic route allows for the seamless transition from laboratory scale to commercial production, ensuring a reliable supply of high-purity intermediates. By focusing on the structural integrity of the tetrahydrothieno[3,2-c]pyridine core and optimizing the introduction of the cyclopropyl ketone side chain, the process minimizes the formation of deleterious by-products. This technical advancement not only enhances the economic viability of Prasugrel production but also aligns with modern green chemistry principles by reducing the generation of hazardous waste. As a result, manufacturers can secure a more resilient supply chain capable of meeting the growing global demand for effective cardiovascular therapies without compromising on environmental compliance or product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prasugrel and its precursors has been plagued by significant technical inefficiencies that hinder large-scale adoption. Prior art, such as the methods disclosed in EP542411, relies on reaction pathways characterized by slow reaction rates and a propensity for generating numerous side products. These inefficiencies result in lower overall yields and create substantial challenges during the purification phase, often requiring complex chromatographic separations that are impractical for industrial settings. Furthermore, alternative routes described in documents like WO2009062044 involve intermediates that are difficult to prepare and inherently expensive, driving up the cost of goods sold and limiting the economic feasibility of the final API. The cumulative effect of these drawbacks is a manufacturing process that is fragile, costly, and environmentally burdensome, failing to meet the rigorous demands of modern pharmaceutical supply chains.
The Novel Approach
In stark contrast, the methodology outlined in CN101948479A offers a paradigm shift by introducing a highly efficient and operationally simple synthetic strategy. This novel approach utilizes a trityl-protected tetrahydrothieno[3,2-c]pyridine derivative as a stable platform for subsequent functionalization, thereby mitigating the side reactions that plague unprotected analogues. The process employs a sequential alkylation and borylation strategy that proceeds under mild conditions, utilizing commercially available reagents such as trityl chloride and borate esters. This ensures that the reaction kinetics are favorable and that the intermediate species remain stable throughout the synthesis. By streamlining the workflow into distinct, high-yielding steps, this method drastically simplifies the isolation and purification of the target compound, making it ideally suited for continuous or batch processing in a commercial facility.
Mechanistic Insights into Trityl-Protection and Suzuki-Miyaura Type Coupling
The core of this synthetic innovation lies in the strategic manipulation of the nitrogen atom within the thienopyridine ring system. The initial alkylation step involves the reaction of 4,5,6,7-tetrahydrothieno[3,2-c]pyridine with trityl chloride in the presence of a base, such as triethylamine or potassium carbonate. This installation of the bulky trityl group serves a dual purpose: it protects the secondary amine from unwanted nucleophilic attacks during subsequent harsh conditions and enhances the solubility of the intermediate in organic solvents. Following protection, the molecule undergoes a directed lithiation at the C2 position using a strong organic base like n-butyllithium at cryogenic temperatures ranging from -20°C to 10°C. This precise metalation allows for the subsequent introduction of a boron moiety via reaction with a borate ester, creating a versatile organoboron species ready for cross-coupling.
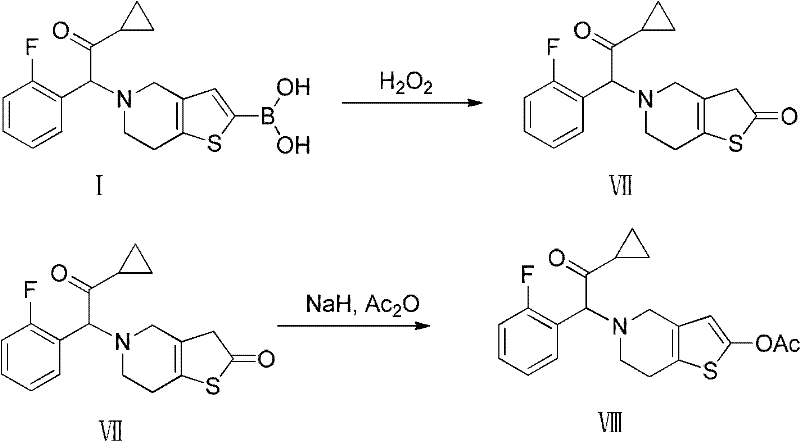
The final assembly of the pharmacophore is achieved through a coupling reaction between the deprotected amine salt and a specific alpha-bromo ketone derivative. This step effectively links the heterocyclic core with the 2-fluorophenyl cyclopropyl ketone side chain, which is essential for the biological activity of the final drug. The use of a base-mediated coupling in an aprotic solvent ensures high conversion rates while minimizing the degradation of sensitive functional groups. Furthermore, the subsequent conversion of this intermediate to the active Prasugrel molecule involves a controlled oxidation of the sulfur atom followed by acetylation. This mechanistic pathway ensures that the stereochemical and structural integrity of the molecule is preserved, resulting in a product with a clean impurity profile that meets the stringent specifications required for clinical application.
How to Synthesize Prasugrel Intermediate Efficiently
The synthesis of this critical pharmaceutical intermediate requires precise control over reaction parameters to maximize yield and purity. The process begins with the protection of the thienopyridine nitrogen, followed by a low-temperature borylation to install the necessary coupling handle. After removing the protecting group under acidic conditions, the final coupling with the bromo-ketone completes the construction of the core scaffold. Each step has been optimized to balance reaction speed with selectivity, ensuring that the process is robust enough for multi-kilogram production. For detailed operational parameters, including specific stoichiometry, temperature ramps, and workup procedures, please refer to the standardized synthesis guide below.
- Perform alkylation of 4,5,6,7-tetrahydrothieno[3,2-c]pyridine with trityl chloride under alkaline conditions to form the N-trityl protected derivative.
- Execute borylation using an organic strong base and borate ester at low temperatures (-20 to 10°C) to introduce the boronic acid functionality.
- Conduct deprotection under acidic conditions followed by a coupling reaction with 2-bromo-1-cyclopropyl-2-(2-fluorophenyl)ethanone to yield the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The reliance on cheap and easily obtainable raw materials significantly de-risks the supply chain, reducing dependency on exotic or single-source reagents that can cause production delays. The simplicity of the operational steps translates directly into reduced manufacturing cycle times and lower labor costs, as the process does not require specialized equipment or extreme conditions that drive up utility expenses. Moreover, the high yield and ease of purification mean that less raw material is wasted, leading to a more sustainable and cost-efficient production model that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the use of commodity chemicals for the alkylation and borylation steps drive down the direct material costs substantially. By avoiding complex purification techniques like preparative HPLC and relying instead on standard crystallization and extraction methods, the operational expenditure is significantly lowered. This economic efficiency allows for a more competitive pricing structure for the final API, providing a strategic advantage in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Since all key starting materials, including the thienopyridine derivative and the bromo-ketone, are commercially available from multiple vendors, the risk of supply disruption is minimized. The robustness of the chemical transformations ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that can disrupt inventory planning. This reliability is crucial for maintaining continuous production schedules and meeting the delivery commitments of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process generates minimal three wastes (waste water, waste gas, and solid waste) compared to traditional methods, easing the burden on environmental treatment facilities and reducing disposal costs. The mild reaction conditions and absence of heavy metal catalysts simplify the regulatory compliance landscape, facilitating faster approval for commercial scale-up. This environmental friendliness not only reduces the ecological footprint but also future-proofs the manufacturing site against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this Prasugrel intermediate. These insights are derived directly from the patent data and are intended to clarify the operational benefits and chemical characteristics of the described methodology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new synthetic route compared to prior art?
A: This route eliminates the slow reaction rates and difficult purification steps associated with previous methods (such as EP542411). It utilizes commercially available raw materials and achieves higher yields with significantly reduced environmental waste.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is suitable for large-scale production due to simple operational steps, mild reaction conditions, and the use of inexpensive, readily available starting materials.
Q: How is the purity of the final intermediate controlled?
A: Purity is managed through the strategic use of a trityl protecting group which minimizes side reactions during the borylation step, followed by efficient recrystallization processes that remove impurities effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prasugrel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical clients. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our dedication to technical excellence ensures that the complex chemistry involved in producing heterocyclic intermediates is managed with the utmost precision and care.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance the efficiency and reliability of your Prasugrel manufacturing operations.
