Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
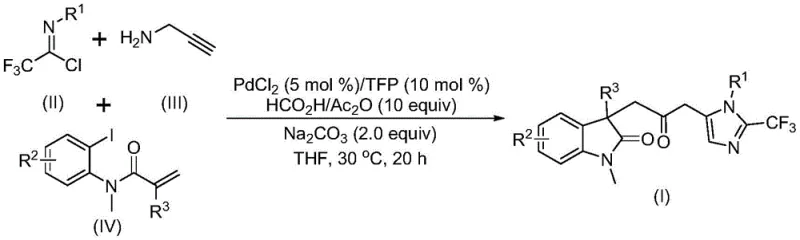
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multi-component synthesis strategy for producing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technological advancement addresses critical bottlenecks in traditional heterocycle synthesis by employing a transition metal palladium-catalyzed carbonylation cascade that operates under remarkably mild conditions. For R&D directors and process chemists, the ability to construct these intricate double heterocyclic systems in a single pot represents a significant leap forward in synthetic efficiency. The method eliminates the reliance on pre-functionalized carbonyl sources that often require harsh handling, instead utilizing a safe, in-situ generation of carbon monoxide. This innovation not only streamlines the synthetic route but also aligns with modern green chemistry principles, making it an attractive candidate for the development of next-generation active pharmaceutical ingredients (APIs) and high-value agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic compounds containing a carbonyl bridge has been fraught with significant synthetic challenges and safety hazards. Traditional approaches often rely on the direct coupling of two separate heterocyclic substrates, which frequently suffers from low atom economy and poor regioselectivity, leading to difficult purification processes and reduced overall yields. Another common pathway involves the oxidative cyclization of substrates bearing dual nucleophiles, a method that typically necessitates the use of stoichiometric oxidants and elevated temperatures, thereby increasing the risk of side reactions and thermal degradation of sensitive functional groups. Furthermore, conventional carbonylation reactions to install the bridging carbonyl group almost universally require the use of toxic carbon monoxide gas, demanding specialized high-pressure equipment and rigorous safety protocols that inflate capital expenditure and operational complexity. These limitations collectively hinder the rapid scale-up and cost-effective manufacturing of carbonyl-bridged biheterocycles, creating a supply chain bottleneck for drug developers who require reliable access to these complex scaffolds for clinical trials and commercial production.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated palladium-catalyzed multicomponent reaction that seamlessly integrates three distinct building blocks: trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamides. This transformative strategy leverages a mixture of formic acid and acetic anhydride as a safe and convenient surrogate for carbon monoxide gas, effectively bypassing the need for hazardous high-pressure CO infrastructure. The reaction proceeds efficiently at a mild temperature of 30°C in common organic solvents like tetrahydrofuran, demonstrating exceptional functional group tolerance across a wide range of substrates including those with halogen, nitro, and trifluoromethyl substituents. By enabling the simultaneous formation of multiple chemical bonds in a one-pot operation, this method drastically reduces the number of isolation steps and solvent consumption, directly translating to lower manufacturing costs and a reduced environmental footprint. The versatility of this system allows for the facile diversification of the final biheterocyclic product through simple modification of the starting materials, providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, initiating with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This key step generates an aryl-palladium intermediate which subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, forming a stable five-membered alkyl-palladium ring system characteristic of the indolinone core. Crucially, the carbon monoxide required for the bridging carbonyl is generated in situ from the decomposition of the formic acid and acetic anhydride additive mixture, which then inserts into the palladium-carbon bond to yield a reactive acyl-palladium species. Concurrently, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted intermolecular process to form a trifluoroacetamidine intermediate, which undergoes isomerization to a more reactive tautomer. The final cyclization event is driven by the nucleophilic attack of this amidine species onto the activated acyl-palladium complex, followed by reductive elimination to release the final carbonyl-bridged biheterocyclic product and regenerate the active palladium catalyst. This intricate dance of catalytic cycles ensures high turnover numbers and minimizes the accumulation of palladium black or other inactive species that could plague the reaction.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining high product purity. The low operating temperature of 30°C significantly suppresses thermal decomposition pathways that often lead to polymeric byproducts or tar formation in high-temperature cyclizations. Furthermore, the use of specific ligands such as trifurylphosphine (TFP) stabilizes the palladium center, preventing premature catalyst deactivation and ensuring consistent reaction kinetics throughout the 12 to 20-hour duration. The choice of sodium carbonate as a base provides a buffered environment that facilitates the necessary deprotonation steps without promoting hydrolysis of the sensitive imidoyl chloride or the final amide linkages. For quality control teams, this means the crude reaction mixture typically contains fewer structurally related impurities, simplifying the downstream purification via standard silica gel column chromatography. The robustness of the mechanism against varying electronic properties of the substrates—whether electron-rich methyl groups or electron-withdrawing nitro groups—ensures a consistent impurity profile across different batches, a critical factor for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize yield and reproducibility. The process begins with the preparation of the catalytic system, where palladium chloride and the trifurylphosphine ligand are combined with the carbon monoxide source additives before the introduction of the organic substrates. It is essential to maintain an inert atmosphere during the initial setup to protect the sensitive palladium catalyst from oxidation, although the reaction itself is tolerant to minor variations once initiated. The detailed standardized synthesis steps, including specific molar equivalents for the trifluoroethylimidoyl chloride, propargylamine, and acrylamide components, are outlined below to guide your technical team in replicating the high yields reported in the patent literature.
- Mix palladium chloride, trifurylphosphine ligand, sodium carbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction vessel under stirring.
- Maintain the reaction temperature at 30°C for 12 to 20 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond mere chemical efficiency. The elimination of toxic carbon monoxide gas from the process inventory removes a significant regulatory and safety burden, allowing facilities to operate with reduced insurance premiums and simplified environmental compliance reporting. Moreover, the starting materials utilized in this protocol, such as propargylamine and various substituted acrylamides, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency and ensuring supply continuity even during market fluctuations. The ability to run the reaction at ambient pressure and low temperature further reduces energy consumption compared to traditional high-pressure carbonylation processes, contributing to substantial operational cost savings over the lifecycle of the product. These factors combine to create a resilient supply chain capable of supporting long-term commercial production without the volatility associated with hazardous reagent logistics.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction infrastructure and the high atom economy of the multicomponent design. By avoiding the need for specialized high-pressure autoclaves required for gaseous CO reactions, manufacturers can utilize standard glass-lined or stainless steel reactors, significantly lowering capital investment barriers. Additionally, the high conversion rates and selectivity observed in the patent examples mean that less raw material is wasted on byproduct formation, directly improving the cost of goods sold (COGS). The use of inexpensive palladium chloride as the catalyst precursor, rather than more exotic and costly palladium complexes, further optimizes the catalyst cost per kilogram of product. These cumulative efficiencies allow for a more competitive pricing structure for the final biheterocyclic intermediates, enhancing margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of robust, shelf-stable reagents that do not require cryogenic storage or special transport permits. Unlike gaseous reagents which are subject to strict transportation regulations and potential delivery disruptions, the liquid and solid components of this reaction can be stockpiled safely, providing a buffer against market shortages. The broad substrate scope implies that if a specific substituted acrylamide becomes unavailable, alternative analogs can often be substituted with minimal process re-optimization, offering flexibility in raw material sourcing. This adaptability is crucial for maintaining uninterrupted production schedules in the face of global logistical challenges, ensuring that critical pharmaceutical intermediates reach the market without delay.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the homogeneous nature of the reaction mixture and the absence of exothermic hazards associated with high-pressure gas additions. The process has already been validated on a gram scale with consistent results, indicating a clear path to kilogram and multi-ton manufacturing with predictable heat transfer and mixing dynamics. From an environmental standpoint, the replacement of toxic CO gas with formic acid derivatives significantly reduces the facility's hazard profile, simplifying waste treatment and emission control measures. The use of common organic solvents like THF, which can be readily recovered and recycled through distillation, further aligns the process with sustainability goals and reduces the volume of hazardous waste requiring disposal, making it an environmentally responsible choice for modern chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on reaction parameters, safety considerations, and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production workflows or R&D pipelines.
Q: Does this synthesis method require toxic carbon monoxide gas?
A: No, the process utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, eliminating the need for handling hazardous CO gas cylinders and enhancing operational safety.
Q: What is the substrate compatibility for this palladium-catalyzed reaction?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy, nitro, and trifluoromethyl groups on the aromatic rings without significant yield loss.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (30°C) with commercially available reagents and has been successfully demonstrated on a gram scale, indicating strong potential for kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop concept to industrial reality. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that employ advanced analytical techniques to verify identity and potency. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry, providing you with a secure and reliable source for your most challenging chemical building blocks.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements and target specifications. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions about your supply chain strategy. Contact us today to explore how our expertise in carbonyl-bridged biheterocyclic synthesis can drive value and efficiency in your pharmaceutical manufacturing operations.
