Advanced Iron-Catalyzed Cyclization for Scalable 2-Trifluoromethyl Quinazolinone Production
Advanced Iron-Catalyzed Cyclization for Scalable 2-Trifluoromethyl Quinazolinone Production
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic scaffolds that possess high biological activity and metabolic stability. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring systems are ubiquitous in medicinal chemistry, serving as core structures for molecules exhibiting anti-cancer, anticonvulsant, anti-inflammatory, and antifungal properties. The strategic introduction of a trifluoromethyl group into these heterocycles further enhances their electronegativity, lipophilicity, and bioavailability, making them prime candidates for next-generation drug development. This novel methodology leverages a cost-effective iron catalytic system to achieve what previously required harsh conditions or expensive reagents, marking a pivotal shift towards more sustainable and economically viable API intermediate manufacturing.
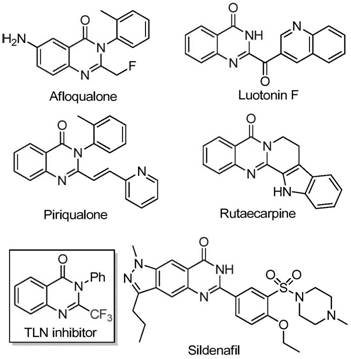
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has been fraught with significant technical and economic challenges. Traditional literature methods predominantly rely on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with trifluoroacetic anhydride or ethyl trifluoroacetate. These conventional pathways are often severely limited by the requirement for severe reaction conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purge. Furthermore, the reliance on expensive fluorinating agents and the narrow substrate scope restrict the versatility of these methods, making them less attractive for the rapid diversification required in modern drug discovery campaigns. The low yields frequently associated with these older protocols also contribute to higher waste generation and increased overall production costs, creating a bottleneck for supply chain reliability.
The Novel Approach
In stark contrast, the innovative route described in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin as starting raw materials, catalyzed by inexpensive ferric chloride. This approach fundamentally alters the economic landscape of producing these valuable intermediates by replacing precious metal catalysts or harsh reagents with a benign iron system. The reaction demonstrates exceptional functional group tolerance, allowing for the incorporation of various substituents such as alkyl, halogen, and methoxy groups at different positions on the aromatic rings without compromising yield. As illustrated in the reaction scheme below, the process involves a tandem sequence that efficiently constructs the quinazolinone core while installing the critical trifluoromethyl group in a single operational setup. This streamlined strategy not only simplifies the synthetic workflow but also significantly enhances the atom economy of the transformation.
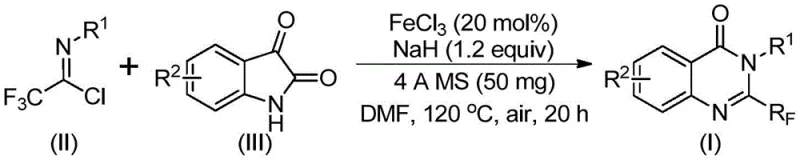
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The success of this transformation lies in the synergistic interaction between the iron catalyst and the base promoter within the reaction medium. Mechanistically, the process initiates with an alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate, generating a transient trifluoroacetamidine intermediate. This intermediate then undergoes an iron-catalyzed decarbonylation and subsequent cyclization reaction, effectively isomerizing to form the stable 2-trifluoromethyl-substituted quinazolinone scaffold. The use of ferric chloride is particularly advantageous as it facilitates the activation of the C-Cl bond and promotes the necessary rearrangement steps under relatively mild thermal conditions. This mechanistic pathway avoids the high-energy barriers typical of non-catalyzed thermal cyclizations, thereby preserving the integrity of sensitive substituents on the substrate.
Furthermore, the inclusion of 4A molecular sieves plays a critical role in controlling the reaction environment and minimizing impurity formation. By sequestering water produced during the condensation steps, the molecular sieves drive the equilibrium towards product formation, preventing hydrolysis of the reactive imidoyl chloride species. This careful management of the reaction milieu ensures high conversion rates and reduces the burden on downstream purification processes. The robustness of this catalytic cycle allows for a wide range of substrates, including those with electron-withdrawing and electron-donating groups, to be processed with consistent efficiency. Such mechanistic clarity provides R&D teams with the confidence to adapt this chemistry for the synthesis of diverse analog libraries, accelerating the lead optimization phase of drug development projects.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it highly suitable for both laboratory scale-up and eventual commercial production. The protocol involves mixing the catalyst, base, molecular sieves, and substrates in a polar aprotic solvent such as DMF, followed by a staged heating profile that optimizes reaction kinetics. The initial lower temperature phase allows for the controlled formation of the intermediate, while the subsequent higher temperature phase drives the cyclization to completion. Detailed standardized operating procedures regarding stoichiometry, solvent volumes, and workup techniques are essential for maintaining batch-to-batch consistency. For a comprehensive breakdown of the specific experimental steps and conditions required to achieve optimal results, please refer to the technical guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly address the pain points of cost volatility and material availability in the fine chemical sector. By shifting away from proprietary or scarce reagents towards commodity chemicals like isatin and simple aromatic amines, manufacturers can secure a more resilient supply chain that is less susceptible to market fluctuations. The elimination of expensive transition metal catalysts, such as palladium or gold, which are often subject to geopolitical supply risks and price spikes, further stabilizes the cost structure of the final intermediate. Additionally, the use of common solvents like DMF simplifies waste management and solvent recovery processes, aligning with increasingly stringent environmental regulations without necessitating costly infrastructure upgrades.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with ferric chloride represents a drastic reduction in raw material costs, as iron salts are among the most affordable catalysts available in the global market. This change eliminates the need for expensive metal scavenging steps typically required to meet strict residual metal limits in pharmaceutical products, thereby simplifying the purification workflow and reducing processing time. The high yields reported across a broad substrate scope mean that less starting material is wasted, directly improving the overall mass balance and lowering the cost per kilogram of the active intermediate. Consequently, this efficiency translates into substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final drug product.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as isatin and substituted anilines ensures that production schedules are not disrupted by the scarcity of specialized reagents. These commodities are produced by numerous suppliers globally, providing procurement managers with multiple sourcing options to mitigate risk. The robustness of the reaction conditions, which tolerate air and moisture to a reasonable extent thanks to the molecular sieves, reduces the complexity of the manufacturing environment, allowing for production in standard facilities rather than requiring specialized inert atmosphere equipment. This accessibility significantly reduces lead times for high-purity pharmaceutical intermediates, enabling faster response to market demands.
- Scalability and Environmental Compliance: The simplicity of the post-treatment procedure, involving filtration and standard column chromatography or crystallization, facilitates easy scale-up from gram to multi-ton quantities without encountering unforeseen engineering hurdles. The process generates minimal hazardous waste compared to traditional methods that might utilize corrosive acids or toxic heavy metals, supporting a greener manufacturing footprint. This environmental compatibility is crucial for maintaining regulatory compliance in major markets and supports the sustainability goals of modern pharmaceutical companies. The ability to scale this process efficiently ensures a continuous supply of critical intermediates, safeguarding the production timelines of downstream API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is crucial for technical teams evaluating the adoption of this route for their specific pipeline projects. For further customization or specific data requests, our technical team is available to provide deeper insights.
Q: What are the key advantages of this FeCl3-catalyzed method over traditional synthesis?
A: This method utilizes cheap and readily available starting materials like isatin and avoids expensive trifluoroacetic anhydride. It operates under relatively mild conditions with high functional group tolerance and eliminates the need for precious metal catalysts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to simple operation, cheap catalysts, and convenient post-treatment procedures.
Q: What is the role of 4A molecular sieves in this reaction?
A: The 4A molecular sieves act as a desiccant to remove water generated during the reaction, driving the equilibrium forward and improving the overall yield and efficiency of the cyclization process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for complex heterocyclic intermediates in the pharmaceutical industry. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We are committed to delivering high-purity 2-trifluoromethyl quinazolinones that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch delivered adheres to the highest international standards, minimizing risk for your downstream processing.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can accelerate your timeline and optimize your budget. Let us be your partner in turning innovative chemistry into commercial reality.
