Revolutionizing Quinazolinone Production: A Scalable Iron-Catalyzed Route for High-Purity Intermediates
Revolutionizing Quinazolinone Production: A Scalable Iron-Catalyzed Route for High-Purity Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways for nitrogen-containing heterocycles, particularly quinazolinones, due to their profound biological significance. As detailed in the recent patent CN111675662B, a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds has been disclosed, offering a transformative approach to accessing these valuable scaffolds. Quinazolinone derivatives are ubiquitous in medicinal chemistry, serving as core structures for agents exhibiting anti-cancer, anticonvulsant, anti-inflammatory, antifungal, and antimalarial activities. The strategic introduction of a trifluoromethyl group into these heterocyclic systems further enhances their pharmacokinetic profiles by improving electronegativity, bioavailability, metabolic stability, and lipophilicity. This patent represents a significant leap forward for any organization acting as a reliable pharmaceutical intermediate supplier, as it replaces complex, low-yielding traditional routes with a streamlined, iron-catalyzed protocol that utilizes commercially accessible starting materials.
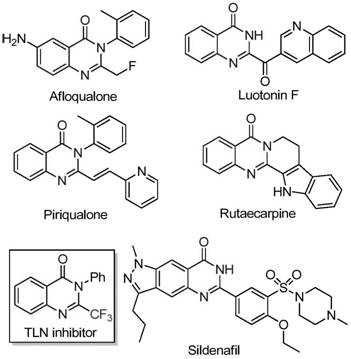
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has been fraught with challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Conventional literature methods typically rely on the cyclization of synthons containing trifluoromethyl groups, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide, anthranilic acid, or isatoic anhydride. While chemically feasible, these established protocols are severely limited by harsh reaction conditions, the requirement for expensive and specialized reaction substrates, generally low yields, and a narrow scope of substrate applicability. These limitations create bottlenecks in cost reduction in API manufacturing, as the reliance on costly reagents and difficult purification processes inflates the overall production budget. Furthermore, the sensitivity of these traditional reactions often necessitates stringent anhydrous conditions and inert atmospheres, complicating the operational workflow for large-scale production facilities.
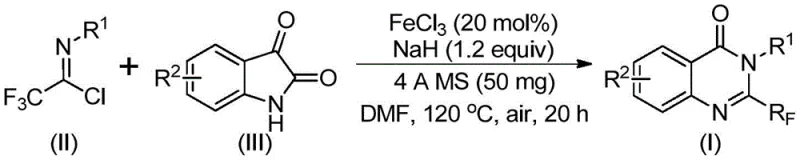
In stark contrast, the novel approach described in patent CN111675662B utilizes readily available trifluoroethylimidoyl chloride and isatin as the primary building blocks, driven by a cheap metal iron catalytic system. This methodology fundamentally shifts the economic and operational paradigm of quinazolinone synthesis. By employing ferric chloride (FeCl3) as the catalyst and sodium hydride (NaH) as the base in a polar aprotic solvent like DMF, the reaction achieves excellent conversion rates under relatively mild thermal conditions. The process involves an initial reaction phase at 40°C followed by heating to 120°C, allowing for a controlled progression of the cyclization. This two-stage thermal profile ensures high selectivity and minimizes the formation of degradation by-products, thereby simplifying the downstream purification process. The use of 4A molecular sieves further aids in maintaining anhydrous conditions essential for the success of the imidoyl chloride coupling, demonstrating a thoughtful integration of practical laboratory techniques into a scalable industrial protocol.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation offers fascinating insights into how base-metal catalysis can achieve results previously reserved for more expensive transition metals. During the reaction, an alkali-promoted carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and the isatin substrate. This initial coupling generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring closure. The presence of the iron catalyst is critical for facilitating the decarbonylation and cyclization steps that follow. Specifically, the iron species likely coordinates with the carbonyl oxygen of the isatin moiety, activating it towards nucleophilic attack and subsequent rearrangement. This iron-catalyzed decarbonylation is the key differentiator that allows for the direct construction of the quinazolinone core with the retention of the trifluoromethyl group at the 2-position. Understanding this mechanism is vital for R&D directors focusing on purity and impurity profiles, as it highlights the specific chemical transformations where side reactions might occur and how the chosen conditions mitigate them.
Furthermore, the impurity control mechanism inherent in this design is robust. The use of specific stoichiometric ratios, such as a molar ratio of ferric chloride to sodium hydride of approximately 0.2:1.2, ensures that the basic environment is sufficient to drive the deprotonation steps without causing excessive decomposition of the sensitive imidoyl chloride. The tolerance for various substituents on the aryl rings (R1 and R2), including alkyl, halogen, methoxy, and nitro groups, indicates that the electronic properties of the substrates do not drastically inhibit the catalytic cycle. This broad functional group tolerance is a hallmark of a mature synthetic method, suggesting that the catalyst system is resilient against poisoning by common heteroatoms or electron-withdrawing groups. For quality control teams, this implies a consistent impurity spectrum that can be easily managed through standard chromatographic techniques, ensuring that the final high-purity pharmaceutical intermediates meet rigorous regulatory standards.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-yielding transformations in a laboratory or pilot plant setting. The procedure is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions that often complicate process chemistry. The reaction utilizes a Schlenk tube or similar vessel where the catalyst, base, molecular sieves, and substrates are combined in an organic solvent. The specific choice of solvent, preferably DMF, acetonitrile, or dioxane, plays a crucial role in solubilizing the ionic intermediates and facilitating the heat transfer required for the 120°C step. The reaction time is optimized to balance completeness with efficiency, typically ranging from 24 to 48 hours depending on the specific substrate electronics. Detailed standardized synthesis steps see the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then increase temperature to 120°C and react for an additional 18-20 hours.
- Filter the reaction mixture, mix with silica gel, and perform column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply reliability. The shift from precious metal catalysts or expensive activated reagents to commodity chemicals like ferric chloride and sodium hydride represents a fundamental reduction in the bill of materials. This change does not merely lower the per-kilogram cost of the catalyst; it also simplifies the logistics of raw material sourcing, as these iron salts are globally available in bulk quantities with stable pricing. Consequently, this leads to substantial cost savings in the overall manufacturing budget, allowing companies to maintain competitive pricing for their final API products even in volatile market conditions. The elimination of expensive transition metal catalysts also removes the need for costly and time-consuming heavy metal scavenging steps during purification, further streamlining the production timeline.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of high-cost reagents with inexpensive, earth-abundant iron catalysts. Traditional methods often require palladium or other noble metals, which not only carry a high price tag but also introduce supply chain risks associated with geopolitical instability in mining regions. By utilizing ferric chloride, manufacturers can decouple their production costs from the fluctuating prices of precious metals. Additionally, the high atom economy of the reaction, where the trifluoroethylimidoyl chloride and isatin are efficiently incorporated into the final product with minimal waste, contributes to a greener and more cost-effective process. The simplified workup procedure, involving basic filtration and chromatography, reduces the consumption of solvents and silica gel, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as isatin and aromatic amines ensures a robust and continuous supply chain. Isatin is a commodity chemical produced on a large scale for various industrial applications, meaning that supply disruptions are highly unlikely. Similarly, the aromatic amines required to synthesize the trifluoroethylimidoyl chloride are widely produced intermediates in the dye and agrochemical industries. This abundance of raw materials means that procurement teams can source inputs from multiple vendors, reducing the risk of single-source dependency. The stability of these raw materials also allows for long-term storage without significant degradation, enabling manufacturers to build strategic inventory buffers against market fluctuations or logistical delays.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration at the gram level with a clear path to industrial application. The reaction conditions, utilizing standard heating and stirring in common solvents like DMF, are easily transferable to large-scale reactors found in modern chemical plants. From an environmental perspective, the use of iron, a non-toxic metal, aligns with the increasing regulatory pressure to reduce heavy metal contamination in pharmaceutical waste streams. The process generates fewer hazardous by-products compared to traditional methods involving harsh acylating agents, simplifying waste treatment and disposal. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The data reflects a balance between high yield and operational simplicity, making it a strong candidate for process development.
Q: What are the key advantages of the FeCl3 catalytic system over traditional methods?
A: The FeCl3 catalytic system utilizes inexpensive and abundant iron salts compared to precious metal catalysts, significantly reducing raw material costs while maintaining high reaction efficiency and broad substrate tolerance.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states that the method is simple to operate, uses readily available starting materials, and has been demonstrated at the gram level with potential for expansion to industrial scale applications.
Q: What is the typical purity profile of the resulting quinazolinone compounds?
A: The process involves a straightforward post-treatment including filtration and column chromatography, which effectively removes impurities and by-products, ensuring high-purity specifications suitable for pharmaceutical intermediate standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the technology disclosed in patent CN111675662B and is fully prepared to leverage this iron-catalyzed methodology for your project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate we deliver meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project specifics,索取 specific COA data, and obtain comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates while maximizing your operational efficiency.
