Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Manufacturing
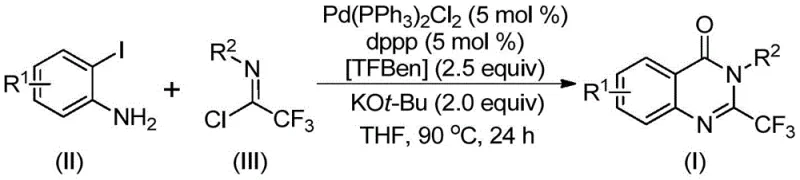
The pharmaceutical industry continuously seeks robust synthetic methodologies that balance high purity with operational safety, particularly when constructing complex heterocyclic scaffolds essential for modern drug discovery. Patent CN112125856A introduces a transformative approach to synthesizing 2-trifluoromethyl substituted quinazolinone derivatives, a privileged structural motif found in numerous bioactive molecules exhibiting anti-inflammatory, antiviral, and anticancer properties. This technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, distinguishing itself by replacing hazardous gaseous carbon monoxide with a stable solid surrogate, 1,3,5-tricarboxylate phenol ester (TFBen). For R&D directors and procurement specialists, this innovation represents a significant leap forward in process safety and scalability, enabling the efficient production of high-value pharmaceutical intermediates without the infrastructure burdens associated with high-pressure gas handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core, especially with specific trifluoromethyl substitution at the 2-position, has relied on methodologies fraught with significant operational challenges and safety risks. Traditional routes often involve the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, which frequently require harsh reaction conditions and expensive, pre-activated substrates that drive up manufacturing costs. Furthermore, alternative strategies utilizing isatoic anhydride or T3P-promoted tandem reactions often suffer from narrow substrate scopes and inconsistent yields, limiting their utility in diverse medicinal chemistry campaigns. Perhaps most critically, many conventional carbonylation processes necessitate the use of toxic carbon monoxide gas, imposing stringent safety protocols, specialized high-pressure equipment, and complex waste management procedures that severely hinder commercial scale-up and increase the overall cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these legacy methods, the methodology disclosed in CN112125856A offers a streamlined, operationally simple pathway that utilizes readily available starting materials such as o-iodoaniline and trifluoroethylimidoyl chloride. By employing TFBen as a solid carbon monoxide substitute, this novel approach effectively circumvents the dangers associated with handling toxic colorless gas, thereby simplifying the reactor setup and enhancing workplace safety profiles significantly. The reaction proceeds under relatively mild thermal conditions at 90°C in common organic solvents like tetrahydrofuran, demonstrating exceptional functional group tolerance that allows for the incorporation of diverse substituents including halogens, alkyl groups, and nitro moieties. This versatility ensures that the process is not merely a laboratory curiosity but a viable industrial solution capable of generating a wide array of derivatives for structure-activity relationship studies and bulk production alike.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic elegance of this transformation lies in its multi-step tandem sequence orchestrated by the palladium catalyst system, which facilitates the formation of multiple bonds in a single operational pot. The reaction is postulated to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride, generating a trifluoroacetamidine intermediate in situ. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring to form a reactive divalent palladium species, setting the stage for the crucial carbonylation step. Upon heating, the TFBen additive decomposes to release carbon monoxide in a controlled manner, which then inserts into the carbon-palladium bond to generate an acyl-palladium intermediate, effectively building the carbonyl functionality required for the quinazolinone ring closure.
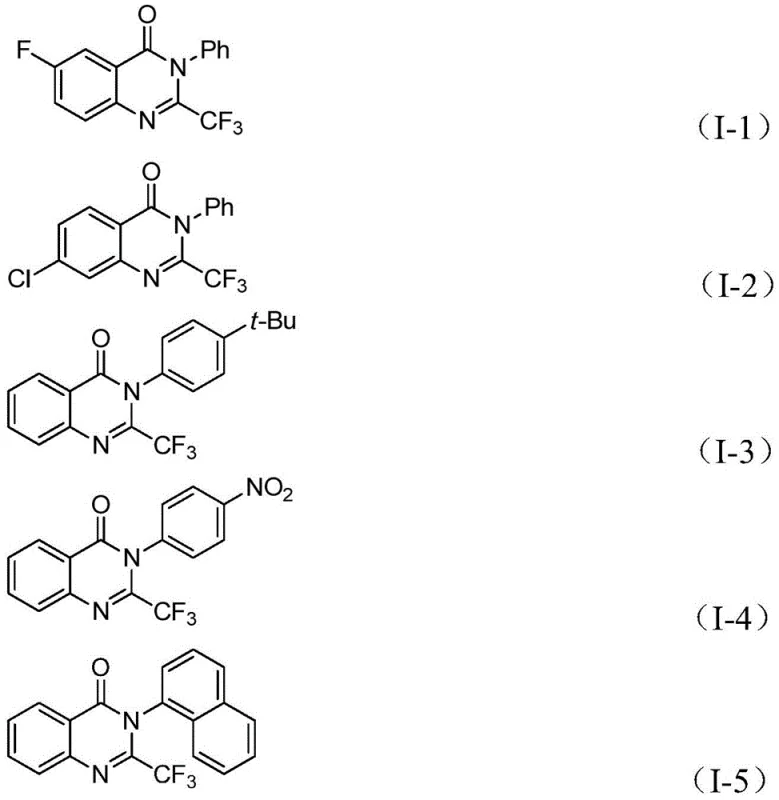
Following the CO insertion, the presence of a strong base such as potassium tert-butoxide promotes the formation of a palladium-nitrogen bond, leading to the generation of a seven-membered ring palladium intermediate that serves as the precursor to the final heterocycle. The catalytic cycle concludes with a reductive elimination step that releases the desired 2-trifluoromethyl substituted quinazolinone derivative while regenerating the active palladium species for further turnover. This intricate dance of organometallic steps ensures high atom economy and minimizes the formation of unwanted byproducts, resulting in a cleaner crude reaction profile that simplifies downstream purification efforts. The ability to tolerate various electronic environments on the aryl rings, as evidenced by the successful synthesis of derivatives with electron-withdrawing nitro groups and electron-donating tert-butyl groups, underscores the robustness of this catalytic system.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction parameters to maximize yield and purity, leveraging the specific conditions optimized in the patent examples. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, solid CO source, and substrates in an aprotic solvent, followed by heating to facilitate the tandem cyclization. While the general procedure is straightforward, attention to detail regarding the molar ratios of the catalyst system and the duration of heating is critical to ensuring complete conversion of the starting materials. For a comprehensive, step-by-step guide tailored to your specific production needs, please refer to the standardized synthesis protocol detailed below.
- Combine palladium catalyst, dppp ligand, potassium tert-butoxide, TFBen, trifluoroethylimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this patented methodology offers compelling advantages that directly address the pain points of cost volatility and logistical complexity in fine chemical manufacturing. The shift away from gaseous reagents and high-pressure equipment fundamentally alters the capital expenditure requirements for production facilities, allowing for more flexible and decentralized manufacturing capabilities. Moreover, the reliance on commercially available and inexpensive starting materials like o-iodoanilines and trifluoroethylimidoyl chlorides ensures a stable supply chain that is less susceptible to the fluctuations often seen with specialized or proprietary reagents. This stability is crucial for maintaining consistent production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the need for expensive gas containment systems, specialized monitoring equipment, and complex safety protocols, leading to substantial operational cost savings. Additionally, the use of a solid CO surrogate simplifies the reaction workup and reduces the burden on waste treatment facilities, further driving down the environmental compliance costs associated with the process. The high efficiency of the catalyst system also means that lower loading levels can be utilized without compromising yield, reducing the consumption of precious metal resources and minimizing the cost of metal scavenging in the final purification stages.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commodity chemicals that are widely produced and available from multiple global suppliers, mitigating the risk of single-source dependency. This broad availability ensures that production can continue uninterrupted even if one supplier faces logistical issues, providing a robust buffer against supply chain disruptions. Furthermore, the simplicity of the reaction conditions allows for rapid technology transfer between different manufacturing sites, enabling a distributed production network that can respond quickly to surges in demand for key pharmaceutical intermediates.
- Scalability and Environmental Compliance: The use of a solid carbon monoxide source inherently improves the safety profile of the reaction, making it easier to scale from kilogram to tonne quantities without encountering the exponential increase in risk associated with pressurized gases. This safety margin facilitates smoother regulatory approvals and reduces the time required for process validation at commercial scales. From an environmental standpoint, the reduced hazard profile and improved atom economy contribute to a greener manufacturing process, aligning with the increasing corporate sustainability goals and regulatory pressures facing the modern chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and specifications provided in the patent documentation. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers reflect the specific capabilities and limitations observed during the development of the process, ensuring transparency and accuracy for potential partners.
Q: What is the primary safety advantage of this synthesis method?
A: The method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, eliminating the need for handling toxic and hazardous gaseous carbon monoxide directly.
Q: What is the substrate scope for this reaction?
A: The process demonstrates excellent compatibility with various substituents on both the o-iodoaniline and the imidoyl chloride, including halogens, alkyl groups, and nitro groups.
Q: What catalyst system is employed?
A: The reaction utilizes a palladium catalyst, specifically bis(triphenylphosphine)palladium(II) dichloride, paired with 1,3-bis(diphenylphosphino)propane (dppp) as the ligand.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl quinazolinone derivative performs consistently in your downstream applications.
We invite you to engage with our technical procurement team to discuss how this innovative palladium-catalyzed route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a partnership built on technical excellence and mutual success.
