Revolutionizing Folic Acid Production: A Green Chemistry Approach for Commercial Scale-Up
Revolutionizing Folic Acid Production: A Green Chemistry Approach for Commercial Scale-Up
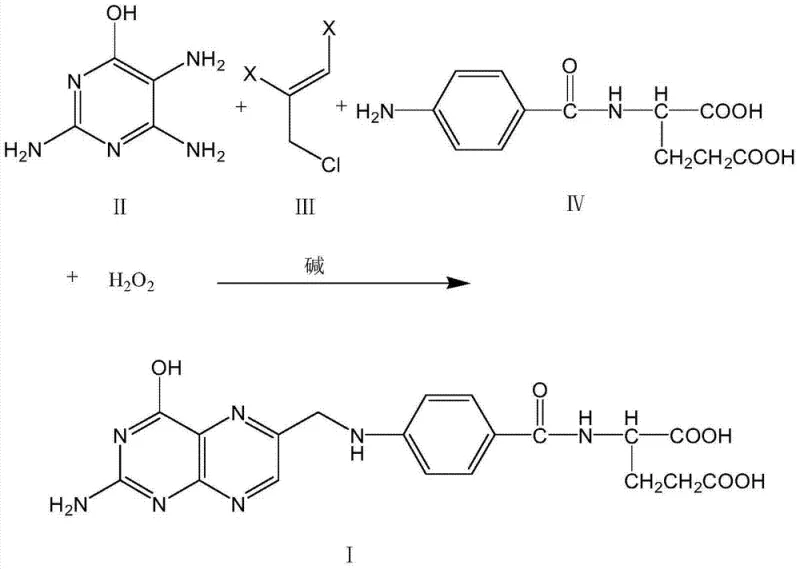
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with environmental sustainability. Patent CN103896945A presents a groundbreaking methodology for the synthesis of Folic Acid (Vitamin B9), addressing long-standing inefficiencies in traditional manufacturing. This innovation replaces the historically problematic 1,1,3-trichloroacetone with stable and accessible 1,2,3-trihalogen propylene derivatives. By integrating a mild oxidation step using agents like hydrogen peroxide alongside a phase-transfer catalyzed condensation, the process achieves exceptional selectivity. For R&D directors and procurement specialists, this represents a pivotal shift towards a more reliable folic acid supplier capability, ensuring that critical vitamin intermediates can be produced with minimal ecological footprint and maximum operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of folic acid has been plagued by the reliance on 1,1,3-trichloroacetone as a key building block for the pteridine ring system. The fundamental issue lies in the availability and quality of this reagent; commercial grades typically possess a purity of merely 40% to 50%, containing significant amounts of 1,3-dichloroacetone and other chlorinated impurities. These impurities participate in side reactions, generating difficult-to-remove byproducts such as pteroic acid, which must be strictly controlled to meet pharmacopeial standards of less than 0.6%. Furthermore, the crude product obtained from these traditional routes necessitates a cumbersome purification regimen involving acid dissolution and alkali melting. This not only drives up operational costs but also generates substantial volumes of hazardous wastewater, creating a severe burden on environmental compliance teams and complicating the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The patented methodology introduces a paradigm shift by utilizing 1,2,3-trihalogen propylene, specifically 1,2,3-trichloropropene or 1,2-dibromo-3-chloropropene, as the superior electrophilic partner. This reagent is chemically stable, commercially abundant, and free from the isomeric impurities that plague trichloroacetone. The reaction proceeds through a streamlined one-pot condensation of 2,4,5-triamino-6-hydroxy pyrimidine, the halogenated propylene, and N-p-benzoyl-L-glutamic acid. By employing a phase-transfer catalyst such as tetrabutylammonium chloride in solvents like tetrahydrofuran or isopropanol, the reaction kinetics are significantly enhanced under mild alkaline conditions. This approach eliminates the need for harsh refining steps, directly yielding a filter cake that requires only a simple ethanol beating wash to achieve sterling quality, thereby offering a clear path for cost reduction in vitamin manufacturing.
Mechanistic Insights into Phase-Transfer Catalyzed Cyclization and Oxidation
The core of this synthetic breakthrough lies in the precise orchestration of nucleophilic substitution and oxidative cyclization. In the initial stage, the amino groups of the 2,4,5-triamino-6-hydroxy pyrimidine act as nucleophiles, attacking the halogenated carbons of the 1,2,3-trihalogen propylene. The presence of a quaternary ammonium salt phase-transfer catalyst is critical here, as it facilitates the transport of hydroxide ions into the organic phase, maintaining the local pH between 5 and 6. This specific pH window is essential to promote the displacement of halide ions without degrading the sensitive glutamic acid moiety. The reaction temperature is carefully controlled between 40°C and 45°C to maximize the formation of the dihydropteridine intermediate while minimizing thermal decomposition. This controlled environment ensures that the structural integrity of the N-p-benzoyl protection group is maintained until the final cyclization event occurs.
Following the condensation, the reaction mixture is cooled to ambient temperatures, typically around 20°C, before the introduction of a mild oxidizing agent. The patent specifies the use of hydrogen peroxide, peracetic acid, or benzoyl hydroperoxide to effect the final aromatization of the pteridine ring. This oxidation step converts the dihydro-intermediate into the fully conjugated folic acid structure. The choice of a mild oxidant is strategic; it prevents the over-oxidation of the glutamic acid side chain or the cleavage of the amide bonds, which are common failure modes in harsher oxidative environments. The result is a highly selective transformation that yields a product with HPLC purity consistently exceeding 96.5%, often reaching upwards of 98%, demonstrating superior impurity control mechanisms compared to legacy processes.
How to Synthesize High-Purity Folic Acid Efficiently
The execution of this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent data. The process begins with the suspension of the pyrimidine starting material in a suitable organic solvent, followed by the sequential addition of the halogenated propylene and the glutamic acid derivative. The maintenance of the pH gradient via the slow addition of aqueous base is a critical process parameter that dictates the rate of halide displacement. Once the condensation is complete, the oxidative workup must be performed under controlled temperatures to prevent exothermic runaway. For detailed operational parameters, stoichiometric ratios, and specific workup procedures required for GMP manufacturing, please refer to the standardized protocol below.
- Condense 2,4,5-triamino-6-hydroxy pyrimidine with 1,2,3-trihalogen propylene and N-p-benzoyl-L-glutamic acid in a solvent like THF or Isopropanol using a phase-transfer catalyst at 40-45°C.
- Maintain pH 5-6 by dripping aqueous sodium hydroxide solution during the reaction to ensure optimal nucleophilic substitution and cyclization.
- Cool the mixture to 20°C, add a mild oxidant such as hydrogen peroxide or benzoyl hydroperoxide, stir for 3-4 hours, and purify the filter cake via ethanol beating.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this trihalogen propylene-based route offers profound strategic advantages beyond mere technical elegance. The primary benefit is the stabilization of the raw material supply chain; unlike 1,1,3-trichloroacetone, which suffers from inconsistent quality and limited availability, 1,2,3-trihalogen propylene is a commodity chemical with a robust global supply network. This shift mitigates the risk of production stoppages due to raw material shortages. Furthermore, the simplification of the downstream processing—specifically the elimination of acid-soluble and alkali-melting refining stages—translates directly into reduced utility consumption and lower waste disposal costs. The process inherently generates less hazardous wastewater, aligning with increasingly stringent environmental regulations and reducing the liability associated with effluent treatment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of complex purification infrastructure. Traditional methods require significant capital expenditure for corrosion-resistant equipment capable of handling strong acids and bases during the refining stage. By achieving high purity directly from the reaction precipitate, this new method removes the need for such specialized hardware. Additionally, the higher reaction selectivity means that less starting material is wasted on byproduct formation, improving the overall atom economy. The use of recyclable solvents like tetrahydrofuran or isopropanol further enhances the cost-efficiency profile, allowing for substantial cost savings in large-scale production runs without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of niche intermediate markets. By switching to widely available halo-propylene derivatives, manufacturers can decouple their production schedules from the bottlenecks associated with specialty chlorinated ketones. The robustness of the reaction conditions, which tolerate a range of solvents and mild oxidants, provides operational flexibility. This flexibility allows production facilities to adapt quickly to regional availability of reagents, ensuring reducing lead time for high-purity folic acid batches. The consistent quality of the input materials also reduces the variance in batch-to-batch analysis, streamlining the quality assurance release process and accelerating time-to-market for finished vitamin products.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, but this methodology is designed with green chemistry principles at its core. The absence of heavy metal catalysts and the use of benign oxidants like hydrogen peroxide, which decomposes into water and oxygen, significantly lowers the environmental toxicity profile of the effluent. This makes the process highly scalable, as the permitting requirements for wastewater discharge are less burdensome compared to traditional routes. The simplified workflow, involving fewer unit operations, reduces the physical footprint required for manufacturing, facilitating easier technology transfer between sites and ensuring that commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel folic acid synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this greener manufacturing standard.
Q: Why is 1,2,3-trihalogen propylene preferred over 1,1,3-trichloroacetone in folic acid synthesis?
A: Traditional methods rely on 1,1,3-trichloroacetone, which typically has a low commercial purity of only 40-50%, leading to significant impurities like pteroic acid. The novel route uses 1,2,3-trihalogen propylene, a stable and commercially available reagent that ensures higher reaction selectivity and final product purity exceeding 98%.
Q: How does this new method improve environmental compliance compared to conventional processes?
A: Conventional folic acid production requires harsh acid-soluble and alkali-melting refining steps that generate substantial wastewater. This patented method achieves high purity through a direct condensation-oxidation sequence followed by simple ethanol beating, drastically reducing waste generation and eliminating the need for complex purification infrastructure.
Q: What represents the critical quality attribute for folic acid in this synthesis?
A: The critical quality attribute is the control of pteroic acid impurities, which must remain below 0.6% according to European Pharmacopoeia standards. By avoiding low-purity trichloroacetone derivatives that introduce chlorinated byproducts, this method consistently achieves HPLC purity greater than 96.5%, often reaching 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Folic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN103896945A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of folic acid or related intermediate meets the highest international pharmacopeial standards. We are committed to leveraging our process chemistry expertise to optimize these green routes for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to explore the full potential of this environmentally friendly production method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative approach can enhance your supply chain resilience and product quality.
