Optimizing Diclofenac Production: A Novel Lewis Acid Catalyzed Cyclization Route for Commercial Scale
Introduction to Advanced Diclofenac Manufacturing Technologies
The global demand for non-steroidal anti-inflammatory drugs (NSAIDs) continues to drive innovation in process chemistry, particularly for high-volume molecules like Diclofenac. Patent CN103145574A introduces a strategically significant advancement in the synthesis of this critical pharmaceutical intermediate, offering a pathway that balances operational simplicity with chemical efficiency. This proprietary methodology outlines a robust four-step sequence that transforms readily available aniline derivatives into the target indole-based structure through a novel N-chloroamide intermediate. By leveraging specific Lewis acid catalysis, the process circumvents many of the historical bottlenecks associated with traditional ring-closure strategies. For technical decision-makers evaluating potential technology transfers, this patent represents a viable alternative that aligns with modern green chemistry principles while maintaining rigorous quality standards. The following analysis details how this route can be integrated into existing manufacturing frameworks to enhance both product integrity and process economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Diclofenac has relied on several distinct synthetic pathways, each carrying inherent logistical and safety burdens that complicate large-scale operations. Traditional routes often involve the Willgerodt-Kindler reaction, which necessitates the use of elemental sulfur and morpholine, creating significant challenges in waste management and odor control within a production facility. Other established methods utilize Ullmann-type coupling reactions requiring copper powder or expensive palladium catalysts alongside iodo-substituted starting materials, which drastically inflate raw material costs and introduce risks of heavy metal contamination in the final API. Furthermore, some legacy processes involve multi-step sequences with low overall yields due to harsh reaction conditions that promote side reactions and degradation of sensitive intermediates. These factors collectively contribute to higher production costs, extended cycle times, and increased regulatory scrutiny regarding residual impurities and environmental impact.
The Novel Approach
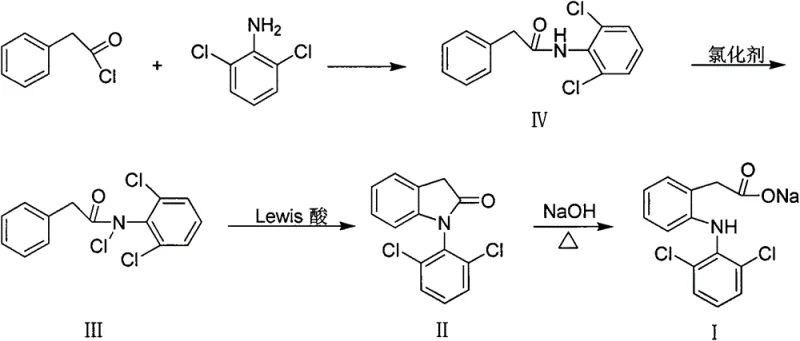
In contrast, the methodology described in the patent data proposes a streamlined approach centered on the formation and cyclization of an N-chloroamide species. This strategy begins with the straightforward acylation of 2,6-dichloroaniline to form the precursor amide, followed by a highly efficient N-chlorination step using accessible reagents like tert-butyl hypochlorite or sodium hypochlorite systems. The core innovation lies in the subsequent Lewis acid-mediated intramolecular cyclization, which effectively closes the ring to form the indolinone scaffold without the need for precious metal catalysts or toxic sulfur reagents. This shift not only simplifies the reaction workflow but also enhances the safety profile of the manufacturing process by utilizing more benign chemical inputs. The final hydrolysis step is equally robust, proceeding under standard alkaline conditions to yield the target carboxylate salt with high conversion rates. This cohesive strategy offers a compelling value proposition for manufacturers seeking to optimize their supply chains and reduce dependency on volatile commodity markets for specialized reagents.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The heart of this synthetic innovation is the Lewis acid-catalyzed cyclization of the N-chloro-N-(2,6-dichlorophenyl)phenylacetamide intermediate. Mechanistically, this transformation involves the activation of the N-Cl bond by a strong Lewis acid, such as anhydrous aluminum trichloride, which generates a highly reactive nitrenium ion or a similar electrophilic nitrogen species in situ. This activated species then undergoes an intramolecular electrophilic aromatic substitution, attacking the ortho-position of the pendant phenyl ring to close the five-membered indolinone ring system. The choice of solvent, typically dichloromethane or similar halogenated hydrocarbons, plays a critical role in stabilizing the charged intermediates and ensuring the reaction proceeds with high regioselectivity. By carefully controlling the temperature, often initiating at 0°C and allowing a gradual warm-up, the process minimizes the risk of intermolecular side reactions or over-chlorination, which are common pitfalls in electrophilic aromatic substitutions. This precise control over the reaction trajectory is essential for maintaining a clean impurity profile, a key requirement for regulatory compliance in pharmaceutical manufacturing.
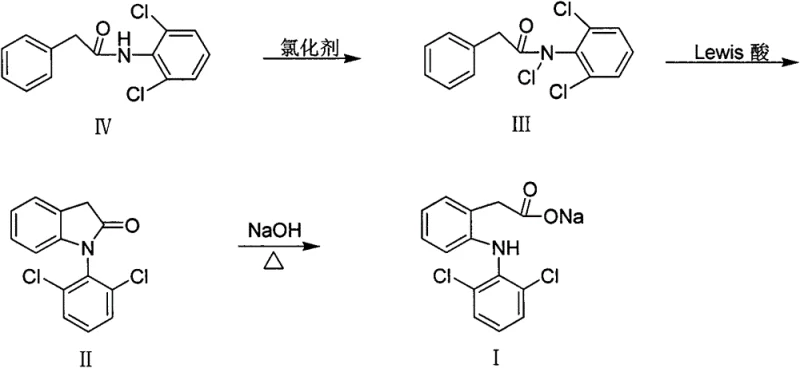
Furthermore, the mechanism inherently supports effective impurity control by limiting the formation of structural isomers. The steric environment created by the 2,6-dichloro substitution on the aniline ring directs the cyclization specifically to the desired position, preventing the formation of unwanted regioisomers that would be difficult to separate later. The use of stoichiometric or near-stoichiometric amounts of the Lewis acid ensures complete conversion of the N-chloro precursor while avoiding excess reagent that could lead to decomposition. Post-reaction workup procedures, involving aqueous quenching and extraction, are designed to efficiently remove aluminum salts and other inorganic byproducts, leaving the organic indolinone product in high purity. This mechanistic clarity provides R&D teams with the confidence to scale the process, as the critical parameters are well-defined and the chemical behavior is predictable under the specified conditions.
How to Synthesize Diclofenac Efficiently
Implementing this synthesis route requires strict adherence to the sequential addition of reagents and temperature controls outlined in the patent examples to ensure optimal yield and safety. The process begins with the preparation of the N-(2,6-dichlorophenyl)phenylacetamide, followed by its conversion to the N-chloro derivative using controlled chlorination conditions. The subsequent cyclization step is the most critical, demanding the careful addition of the Lewis acid catalyst to the organic solution of the N-chloroamide. Finally, the resulting indolinone is subjected to alkaline hydrolysis to open the ring and form the final diclofenac salt. Detailed standardized operating procedures for each of these stages, including specific molar ratios, solvent volumes, and agitation speeds, are essential for successful technology transfer. For a comprehensive breakdown of the exact experimental conditions and workup protocols, please refer to the technical guide below.
- Prepare N-(2,6-dichlorophenyl)phenylacetamide (IV) by reacting 2,6-dichloroaniline with phenylacetyl chloride in the presence of a base like pyridine or triethylamine.
- Convert the amide (IV) to N-chloro-N-(2,6-dichlorophenyl)phenylacetamide (III) using chlorinating agents such as tert-butyl hypochlorite or sodium hypochlorite/acetic acid.
- Perform intramolecular cyclization of compound (III) using a Lewis acid catalyst like anhydrous aluminum trichloride in an organic solvent to form 1-(2,6-dichlorophenyl)-2-indolinone (II).
- Hydrolyze the indolinone (II) in a sodium hydroxide solution, optionally with a phase transfer catalyst, to yield the final Diclofenac sodium salt (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial strategic benefits by decoupling production from volatile markets for precious metals and specialized coupling reagents. The reliance on commodity chemicals such as aluminum trichloride, sodium hydroxide, and common organic solvents ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or sudden price spikes. Additionally, the elimination of toxic sulfur compounds and heavy metal catalysts significantly reduces the complexity and cost of waste treatment and environmental compliance, leading to lower operational expenditures over the lifecycle of the product. The simplicity of the unit operations also translates to reduced equipment maintenance and shorter cleaning cycles between batches, further enhancing overall plant efficiency. These factors combine to create a manufacturing process that is not only economically attractive but also environmentally sustainable, aligning with the increasing corporate focus on green chemistry initiatives.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or copper catalysts with inexpensive Lewis acids like aluminum trichloride results in a direct and significant reduction in raw material costs per kilogram of finished product. Moreover, the high yields reported in the chlorination and cyclization steps minimize the loss of valuable intermediates, thereby improving the overall mass balance and reducing the cost of goods sold. The avoidance of complex purification steps required to remove trace heavy metals further lowers processing costs, as specialized scavenging resins or additional crystallization steps are rendered unnecessary. This economic efficiency allows for more competitive pricing strategies in the global market while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as 2,6-dichloroaniline and phenylacetyl chloride, the process mitigates the risk of supply interruptions that often plague routes dependent on single-source specialty reagents. The robustness of the reaction conditions means that the process can be executed in a wide range of standard pharmaceutical manufacturing facilities without the need for specialized high-pressure or cryogenic equipment. This flexibility enables diversified sourcing strategies and the ability to rapidly ramp up production capacity in response to market demand fluctuations. Consequently, supply chain managers can achieve greater predictability in lead times and inventory planning, ensuring consistent availability of the final API for downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard laboratory glassware and easily adaptable to large-scale reactors used in commercial production. The use of common solvents like dichloromethane and toluene facilitates efficient solvent recovery and recycling systems, minimizing waste generation and environmental footprint. Furthermore, the absence of persistent organic pollutants and heavy metals in the waste stream simplifies regulatory reporting and reduces the burden on wastewater treatment facilities. This alignment with environmental, social, and governance (ESG) goals makes the technology particularly attractive for companies aiming to enhance their sustainability credentials while expanding their manufacturing capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Diclofenac synthesis route, based on the detailed data provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific manufacturing needs. Understanding these nuances is critical for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this N-chloroamide cyclization route over traditional methods?
A: This method avoids the use of toxic sulfur and morpholine required in Willgerodt-Kindler reactions and eliminates the need for expensive copper catalysts or iodo-substituted starting materials found in Ullmann-type couplings. It utilizes readily available chlorinating agents and common Lewis acids, significantly simplifying the supply chain and reducing raw material costs.
Q: How does the process ensure high purity and minimize impurities during the cyclization step?
A: The use of specific Lewis acids like anhydrous aluminum trichloride under controlled temperatures (0°C to room temperature) promotes selective intramolecular electrophilic substitution. This specificity minimizes poly-chlorination side reactions and ensures the formation of the desired indolinone core with high regioselectivity, facilitating easier downstream purification.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable due to the use of common industrial solvents like dichloromethane and toluene, and the reaction conditions are mild (ranging from 0°C to reflux). The elimination of hazardous reagents and the robustness of the hydrolysis step make it well-suited for multi-ton production in standard pharmaceutical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diclofenac Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying robust and scalable synthetic routes to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that technologies like the one described in CN103145574A can be seamlessly transitioned from the lab to full-scale manufacturing. We are committed to delivering high-purity Diclofenac and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance ensures that every batch conforms to international pharmacopoeia standards, providing our partners with the confidence they need to proceed with their own drug development and commercialization efforts.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits of switching to this Lewis acid-catalyzed process. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your supply chain. Let us partner with you to optimize your Diclofenac production and drive value across your entire organization.
