Optimizing Diclofenac Sodium Production: A Novel Acylation and Rearrangement Strategy for Commercial Scale
Optimizing Diclofenac Sodium Production: A Novel Acylation and Rearrangement Strategy for Commercial Scale
The global demand for non-steroidal anti-inflammatory drugs (NSAIDs) continues to drive the need for more efficient and sustainable manufacturing processes for key active pharmaceutical ingredients like Diclofenac Sodium. Patent CN113429308A introduces a groundbreaking synthetic methodology that fundamentally restructures the production pathway, moving away from traditional, energy-intensive routes toward a streamlined acylation and rearrangement strategy. This innovation leverages o-aminophenyl acetate as a pivotal starting material, enabling a sequence of acylation, Chapman rearrangement, and aminolysis that significantly reduces operational complexity. For pharmaceutical manufacturers and procurement leaders, this represents a critical opportunity to enhance supply chain resilience while adhering to increasingly stringent environmental and quality standards. The technical breakthrough lies in the ability to bypass the notorious Ullmann condensation steps, thereby mitigating the risks associated with heavy metal contamination and harsh reaction conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Diclofenac Sodium has been plagued by significant technical bottlenecks that impact both cost and quality consistency. Traditional routes often rely on the Ullmann condensation of o-chlorobenzoic acid derivatives with 2,6-dichloroaniline, a process that necessitates high temperatures, stoichiometric amounts of copper catalysts, and extended reaction times. These conditions not only consume substantial energy but also generate complex impurity profiles that are difficult to purge during downstream processing. Furthermore, the subsequent decarboxylation and cyclization steps required in older methodologies introduce additional unit operations, each representing a potential point of yield loss and safety hazard. The reliance on toxic solvents and the generation of heavy metal waste streams create substantial disposal costs and regulatory burdens, making these legacy processes increasingly untenable in a modern, green chemistry-focused manufacturing environment.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated cascade reaction sequence initiated by the acylation of o-aminophenyl acetate. This method effectively decouples the formation of the diphenylamine backbone from the harsh conditions previously required. By employing 2,6-dichlorophenoxyacid esters or chloro-acyl chlorides as acylating agents, the process establishes the critical carbon-nitrogen bond under much milder thermal conditions. The subsequent intramolecular Chapman rearrangement allows for the precise construction of the molecular architecture without the need for external high-energy inputs. This strategic shift not only shortens the overall synthetic timeline but also inherently improves the safety profile of the operation. The ability to proceed directly to hydrolysis under alkaline conditions to yield the final sodium salt eliminates several isolation and purification steps, thereby maximizing overall throughput and minimizing material handling losses.
Mechanistic Insights into Acylation and Chapman Rearrangement
The core of this technological advancement rests on the precise control of the acylation and subsequent rearrangement mechanisms. The reaction begins with the nucleophilic attack of the amino group in o-aminophenyl acetate on the carbonyl carbon of the acylating agent, facilitated by catalysts such as sodium methoxide or zinc salts. This step forms a stable amide intermediate which serves as the precursor for the critical structural reorganization. The use of alkoxide catalysts is particularly advantageous as they promote the reaction kinetics without introducing transition metals that could persist as trace impurities in the final API. The reaction environment is carefully tuned to ensure that the acylation proceeds to completion before the temperature is adjusted to trigger the rearrangement, ensuring high conversion rates and minimizing the formation of unreacted starting materials.
Following acylation, the system undergoes a Chapman rearrangement, a [1,3]-sigmatropic shift that migrates the acyl group from the oxygen to the nitrogen atom, or in this specific context, facilitates the formation of the diphenylamine structure through the intermediate species. 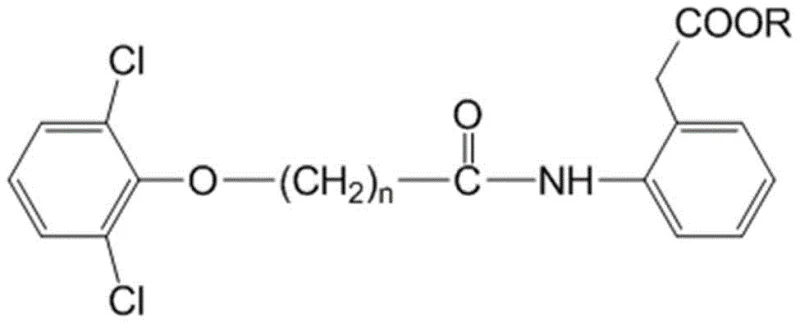 This rearrangement is thermodynamically driven and occurs smoothly under the established alkaline conditions. The resulting intermediate, characterized by the specific connectivity shown in the structural diagrams, is then subjected to aminolysis and hydrolysis. The presence of the ester moiety in the starting material is crucial, as it allows for a one-pot saponification at the end of the sequence. This mechanistic elegance ensures that the carboxylic acid functionality required for the final Diclofenac structure is revealed only at the final stage, preventing premature side reactions and ensuring the integrity of the molecular scaffold throughout the synthesis.
This rearrangement is thermodynamically driven and occurs smoothly under the established alkaline conditions. The resulting intermediate, characterized by the specific connectivity shown in the structural diagrams, is then subjected to aminolysis and hydrolysis. The presence of the ester moiety in the starting material is crucial, as it allows for a one-pot saponification at the end of the sequence. This mechanistic elegance ensures that the carboxylic acid functionality required for the final Diclofenac structure is revealed only at the final stage, preventing premature side reactions and ensuring the integrity of the molecular scaffold throughout the synthesis.
How to Synthesize Diclofenac Sodium Efficiently
The practical implementation of this synthesis route involves a series of well-defined operational steps that balance reaction efficiency with ease of execution. The process initiates with the preparation of the key starting material, o-aminophenyl acetate, typically derived from the nitration and subsequent hydrogenation of phenyl acetate. Once obtained, this amine is reacted with the appropriate acylating agent, such as ethyl 2,6-dichlorophenoxyacetate, in a solvent system like n-butanol. 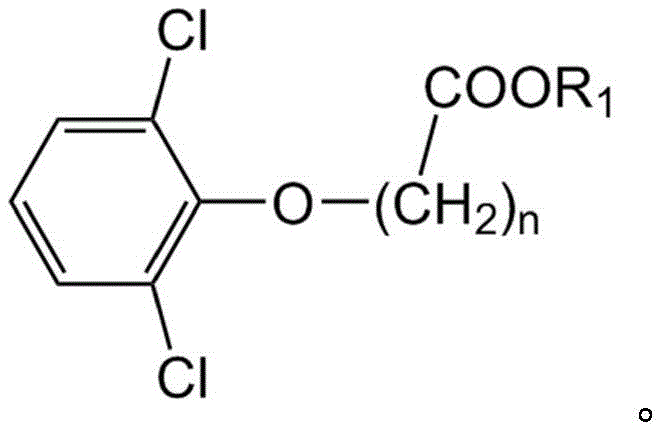 The reaction mixture is heated to moderate temperatures to drive the acylation, after which the conditions are adjusted to facilitate the rearrangement and subsequent aminolysis. The final step involves the addition of an inorganic base, such as sodium hydroxide, to hydrolyze the ester groups and form the sodium salt directly. Detailed standardized synthetic steps see the guide below.
The reaction mixture is heated to moderate temperatures to drive the acylation, after which the conditions are adjusted to facilitate the rearrangement and subsequent aminolysis. The final step involves the addition of an inorganic base, such as sodium hydroxide, to hydrolyze the ester groups and form the sodium salt directly. Detailed standardized synthetic steps see the guide below.
- Prepare o-aminophenyl acetate via nitration of phenyl acetate followed by hydrogenation reduction.
- Perform acylation with 2,6-dichlorophenoxyacid ester or chloro-acyl chloride to form the key amide intermediate.
- Execute Chapman rearrangement and aminolysis under alkaline conditions, followed by hydrolysis to obtain Diclofenac Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple yield improvements. The elimination of copper catalysts and high-pressure hydrogenation steps significantly reduces the dependency on specialized equipment and expensive metal recovery systems. This simplification of the infrastructure requirements translates directly into lower capital expenditure and reduced operational overheads. Furthermore, the use of recyclable solvents such as n-butanol and ethyl acetate aligns with sustainability goals, potentially lowering waste disposal fees and improving the environmental footprint of the manufacturing site. The robustness of the process also implies a more consistent supply of high-quality material, reducing the risk of batch failures that can disrupt downstream formulation schedules.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently drives down manufacturing costs by reducing the number of unit operations and eliminating the need for costly transition metal catalysts. By avoiding the Ullmann condensation, manufacturers save significantly on catalyst procurement and the extensive purification steps required to remove metal residues to ppm levels. Additionally, the milder reaction conditions result in lower energy consumption for heating and cooling, contributing to a leaner cost structure. The direct formation of the sodium salt in the final step further reduces processing time and reagent usage, creating a cumulative effect of substantial cost savings across the production lifecycle.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals like o-aminophenyl acetate and chloroacetic acid derivatives enhances the stability of the raw material supply chain. Unlike specialized intermediates that may have limited suppliers, the precursors for this route are produced at scale globally, mitigating the risk of shortages. The simplified process flow also reduces the lead time required for production cycles, allowing for more responsive inventory management. This agility is crucial for meeting fluctuating market demands for Diclofenac Sodium, ensuring that pharmaceutical customers can maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: From a scale-up perspective, the absence of exothermic hazards associated with traditional Friedel-Crafts or high-pressure reactions makes this process highly scalable. The温和 (mild) conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic materials of construction. Environmentally, the process generates significantly less hazardous waste, particularly regarding heavy metal sludge and halogenated solvent emissions. This compliance with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for environmentally conscious global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this new Diclofenac Sodium synthesis route?
A: The process eliminates the need for harsh Ullmann condensation conditions and decarboxylation steps found in traditional routes. It utilizes mild reaction temperatures and avoids toxic heavy metal catalysts, resulting in a simplified workflow and easier purification.
Q: How does this method improve impurity control compared to conventional methods?
A: By avoiding high-temperature cyclization and copper-catalyzed coupling, the formation of complex side products is minimized. The direct hydrolysis of the ester group under alkaline conditions allows for efficient removal of organic impurities, yielding high-purity Diclofenac Sodium suitable for pharmaceutical standards.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is designed for scalability. It uses commercially available raw materials like o-aminophenyl acetate and employs recyclable solvents such as n-butanol and ethyl acetate. The mild conditions reduce energy consumption and equipment stress, facilitating large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diclofenac Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Diclofenac Sodium meets the highest international pharmacopoeia standards. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, providing our clients with the confidence needed for regulatory filings.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized process can deliver superior value, reliability, and quality for your Diclofenac Sodium needs.
