Advanced Synthetic Route for Methotrexate Intermediate Compound A Enhancing Commercial Scalability
Advanced Synthetic Route for Methotrexate Intermediate Compound A Enhancing Commercial Scalability
The pharmaceutical industry constantly seeks robust manufacturing pathways for critical oncology agents, and the recent disclosure in patent CN115504897A presents a transformative approach to synthesizing Compound A, a pivotal intermediate for Methotrexate. Methotrexate remains a cornerstone folate reductase inhibitor used globally as an antineoplastic agent and for managing autoimmune conditions, making the supply security of its precursors paramount for global health infrastructure. This patent details a sophisticated five-step sequence starting from readily available p-chlorobenzoyl chloride, strategically bypassing the severe limitations of historical methods that relied on hazardous high-pressure equipment. By shifting the paradigm from autoclave-dependent reactions to ambient pressure catalytic processes, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to de-risk their supply chains. The technical depth of this invention lies not just in the chemical transformations but in the holistic optimization of safety, yield, and operational simplicity, addressing the triad of concerns faced by R&D, procurement, and supply chain leadership simultaneously.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical precedents for synthesizing diethyl p-methylaminobenzoyl-L-glutamate, such as those documented in US4136101a and US3892801a, are fraught with significant industrial bottlenecks that hinder modern cost reduction in API manufacturing. These legacy processes mandate the use of autoclaves to facilitate reactions under high pressure, creating substantial capital expenditure barriers and introducing elevated safety risks associated with pressurized vessel operations. Furthermore, alternative routes described in literature, including specific disclosures in the Journal of Chinese Medicine Industry, rely heavily on benzene as a primary solvent, a practice that is increasingly untenable due to stringent environmental regulations and the known carcinogenicity of benzene. These conventional pathways often suffer from suboptimal yields that render them economically unviable for large-scale production, forcing manufacturers to contend with excessive waste generation and inefficient raw material utilization. The cumulative effect of these technical deficiencies is a fragile supply chain vulnerable to regulatory shutdowns and equipment failures, necessitating a urgent transition toward more sustainable and scalable chemical engineering solutions.
The Novel Approach
The innovative methodology outlined in the patent data introduces a streamlined synthetic trajectory that fundamentally resolves the safety and efficiency deficits of the prior art through mild reaction conditions and superior atom economy. Instead of high-pressure hydrogenation, the new route employs a transfer hydrogenation strategy using ammonium formate and a palladium catalyst, allowing the critical deprotection step to proceed safely at atmospheric pressure and moderate temperatures around 50°C. The elimination of benzene in favor of methanol and dichloromethane drastically simplifies solvent recovery and waste management, aligning the process with modern green chemistry principles while maintaining high product quality. As illustrated in the comprehensive reaction scheme below, the sequence flows logically from esterification to amination, followed by hydrogenolysis, hydrolysis, and final amide coupling, with each step demonstrating exceptional conversion efficiency.
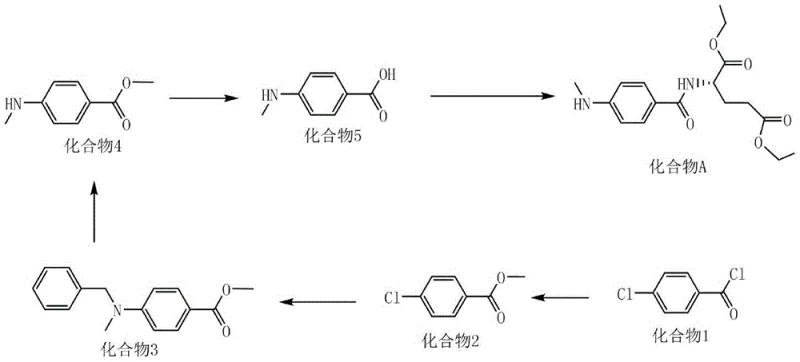
This cohesive design ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved without the need for specialized high-pressure infrastructure, thereby lowering the barrier to entry for qualified manufacturers and enhancing overall market availability.
Mechanistic Insights into Catalytic Hydrogenolysis and Amide Coupling
A deep mechanistic understanding of the critical transformation steps reveals why this pathway offers superior impurity control and reproducibility compared to traditional methods. The hydrogenolysis step, which converts Compound 3 to Compound 4, utilizes a transfer hydrogenation mechanism where ammonium formate serves as the hydrogen donor in the presence of a Pd/C catalyst. This approach avoids the handling of compressed hydrogen gas, mitigating explosion risks while ensuring selective cleavage of the benzyl group without affecting the ester functionalities, a selectivity challenge that often plagues harsher reduction conditions. The subsequent hydrolysis step employs sodium hydroxide under controlled conditions to saponify the methyl ester, generating the free carboxylic acid necessary for the final coupling, with pH adjustment protocols ensuring precise isolation of the intermediate to prevent racemization or degradation.
The final convergence of the synthesis involves the coupling of the generated acid (Compound 5) with L-glutamic acid diethyl ester hydrochloride, a reaction mediated by the uronium-based coupling reagent HBTU. This activation strategy is crucial for forming the amide bond with high fidelity, minimizing the formation of deletion sequences or side products that could complicate downstream purification. The use of HBTU, potentially in conjunction with bases like DIEA, facilitates rapid activation of the carboxylate, driving the reaction to completion at room temperature over a 16-hour period. As shown in the detailed coupling scheme, this step effectively links the aromatic moiety with the glutamate backbone, establishing the core scaffold of the Methotrexate intermediate with high stereochemical integrity.
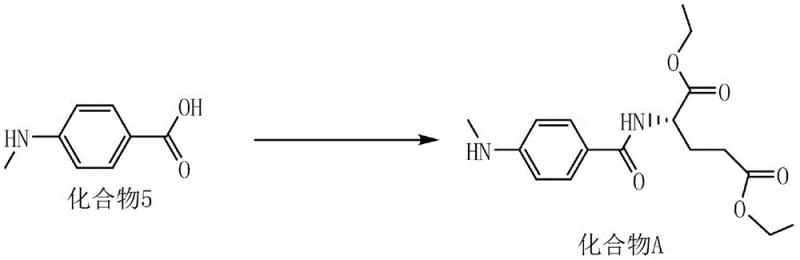
Such precise control over the coupling mechanics is essential for meeting the rigorous purity specifications required for oncology drug substances, ensuring that the final API meets all regulatory standards for clinical use.
How to Synthesize Diethyl p-methylaminobenzoyl-L-glutamate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent embodiments to maximize yield and minimize impurity profiles. The process begins with the esterification of p-chlorobenzoyl chloride in methanol under reflux, followed by nucleophilic substitution with benzylmethylamine using potassium carbonate as a base to scavenge generated acid. The subsequent catalytic deprotection and hydrolysis steps must be monitored closely to ensure complete conversion before proceeding to the final coupling, where stoichiometry and reagent quality are critical for success. Detailed standard operating procedures regarding temperature ramps, workup phases, and crystallization conditions are essential for technology transfer and GMP compliance.
- Esterify p-chlorobenzoyl chloride with methanol at 65°C to form methyl p-chlorobenzoate.
- React the ester with benzylmethylamine in the presence of potassium carbonate to introduce the amine functionality.
- Perform catalytic hydrogenolysis using Pd/C and ammonium formate to remove the benzyl protecting group.
- Hydrolyze the methyl ester using sodium hydroxide to generate the free carboxylic acid intermediate.
- Condense the acid with L-glutamic acid diethyl ester hydrochloride using HBTU coupling reagent to yield Compound A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible operational benefits that extend beyond simple chemical yield improvements. By eliminating the requirement for high-pressure autoclaves, manufacturers can utilize standard glass-lined or stainless steel reactors that are more widely available and easier to maintain, significantly reducing capital investment costs and maintenance downtime. The shift away from benzene to less toxic solvents reduces the regulatory burden associated with solvent residue testing and hazardous waste disposal, leading to substantial cost savings in environmental compliance and waste treatment operations. Furthermore, the high yields reported in the examples, such as the 94.38% yield in the initial esterification and 92.74% in the hydrogenolysis step, indicate a highly efficient mass balance that minimizes raw material waste and maximizes output per batch.
- Cost Reduction in Manufacturing: The removal of high-pressure equipment requirements allows for production in standard facilities, drastically lowering the barrier to entry and operational overhead. Additionally, the use of transfer hydrogenation eliminates the need for expensive high-purity hydrogen gas infrastructure, replacing it with inexpensive ammonium formate, which further drives down variable production costs. The high efficiency of the coupling step using HBTU ensures that expensive chiral starting materials are not wasted, optimizing the cost of goods sold for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like p-chlorobenzoyl chloride and methanol ensures that raw material sourcing is robust and less susceptible to geopolitical disruptions compared to specialized reagents. The simplified process flow reduces the number of unit operations and potential failure points, resulting in more predictable lead times and consistent batch-to-batch quality. This stability is crucial for long-term supply agreements with major pharmaceutical companies who require guaranteed continuity of supply for life-saving medications.
- Scalability and Environmental Compliance: Operating at atmospheric pressure and moderate temperatures makes the process inherently safer and easier to scale from pilot plant to multi-ton commercial production without encountering heat transfer or pressure limitations. The avoidance of carcinogenic solvents like benzene aligns the manufacturing process with increasingly strict global environmental regulations, future-proofing the supply chain against regulatory changes. This environmental stewardship enhances the corporate social responsibility profile of the supply chain, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway for Methotrexate intermediates. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these nuances is critical for evaluating the feasibility of adopting this technology within existing manufacturing frameworks.
Q: Why is the new synthesis method safer than prior art US4136101a?
A: The prior art requires high-pressure autoclave equipment which poses significant industrial safety risks. The novel method operates at atmospheric pressure using transfer hydrogenation, eliminating the need for specialized high-pressure vessels and reducing operational hazards.
Q: How does this route improve environmental compliance compared to older methods?
A: Previous methods disclosed in journals utilized benzene as a solvent, which is a known carcinogen and strictly regulated. This new protocol utilizes methanol and dichloromethane, significantly reducing toxicological risks and simplifying waste treatment protocols for large-scale manufacturing.
Q: What represents the key yield improvement in this synthetic strategy?
A: While older routes suffered from low yields unsuitable for scale production, this optimized pathway achieves individual step yields exceeding 85%, with the initial esterification reaching over 94%, ensuring a robust overall mass balance for commercial operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl p-methylaminobenzoyl-L-glutamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and high-quality supply of critical oncology intermediates like Compound A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our materials meet the exacting standards required for API synthesis. Our commitment to technical excellence allows us to navigate complex chemical landscapes, delivering intermediates that facilitate the efficient production of life-saving medicines.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient manufacturing process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability.
