Advanced TBTU-Mediated Cyclization for High-Purity Lorlatinib Manufacturing
Advanced TBTU-Mediated Cyclization for High-Purity Lorlatinib Manufacturing
The pharmaceutical landscape for non-small cell lung cancer (NSCLC) treatment has been significantly advanced by third-generation ALK inhibitors, with Lorlatinib standing out as a critical therapeutic agent. As the industry seeks more efficient manufacturing routes for this complex macrocyclic compound, Patent CN112457293B emerges as a pivotal technological breakthrough. This patent details a novel application of TBTU (O-benzotriazole-N,N,N',N'-tetramethyluronium tetrafluoroborate) in conjunction with DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) to achieve superior cyclization yields. For R&D directors and procurement strategists, this methodology represents a shift from traditional, low-efficiency coupling agents to a more robust, economically viable system. By optimizing the condensation step, this process addresses the chronic challenges of low conversion rates and difficult purification that have plagued earlier synthetic routes, positioning it as a cornerstone for reliable API intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Lorlatinib relied heavily on coupling agents such as HATU or COMU, often paired with bases like DIPEA or TEA. The background art highlights severe inefficiencies in these legacy processes. For instance, methods utilizing HATU and DIPEA at low temperatures resulted in abysmal yields of merely 29%, rendering them commercially unviable for large-scale production. Even when heating was applied to 60-70°C for extended periods of 4 to 5 hours, yields only reached approximately 78%, which still leaves a substantial amount of valuable starting material wasted. Furthermore, alternative routes employing COMU with TEA struggled to exceed 60% yield. Beyond the poor atom economy, these conventional methods suffered from the necessity of using large excesses of expensive condensing agents and bases. This not only inflated raw material costs but also complicated the downstream purification process, as removing vast quantities of urea byproducts and salt residues required extensive washing and chromatography, thereby increasing the environmental footprint and production lead time.
The Novel Approach
The disclosed invention fundamentally reengineers the macrocyclization step by introducing a specific combination of TBTU and DBU. This synergistic pairing allows the reaction to proceed efficiently at moderate temperatures of 40-60°C within a shortened timeframe of just 2 to 3 hours. The data presented in the patent examples demonstrates a dramatic improvement, with yields consistently surpassing 87% and reaching as high as 89.4% in optimized embodiments. Crucially, this high efficiency is achieved with a significantly reduced stoichiometric requirement for the coupling agent, with molar ratios ranging from 1:0.5 to 1:1.5 relative to the substrate. This reduction in reagent load directly translates to a cleaner reaction profile, minimizing the formation of difficult-to-remove impurities. For a reliable API intermediate supplier, this translates to a process that is not only higher yielding but also inherently easier to scale, as the workup procedure is simplified to a straightforward quench with ammonium chloride followed by extraction and crystallization.
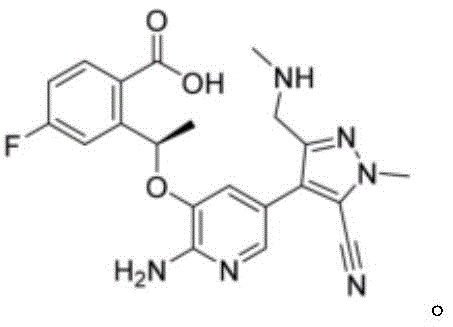
Mechanistic Insights into TBTU/DBU Mediated Macrocyclization
The success of this synthetic route lies in the specific mechanistic interplay between the uronium salt TBTU and the non-nucleophilic base DBU. In the context of forming the strained macrocyclic ring of Lorlatinib, the activation of the carboxylic acid moiety on Compound A is the rate-determining step. TBTU acts as a highly effective activating agent, converting the carboxylic acid into a reactive O-acylisourea intermediate or an active ester species in situ. Unlike HATU, which can sometimes lead to racemization or sluggish reactivity in sterically hindered environments, TBTU provides a balanced reactivity profile that is particularly well-suited for this specific substrate architecture. The presence of the fluorine atom and the bulky pyrazole-pyridine scaffold creates a challenging steric environment for the nucleophilic attack by the amine. The choice of DBU as the base is equally critical; its strong basicity and unique bicyclic structure facilitate the rapid deprotonation of the amine nucleophile without participating in unwanted side reactions. This ensures that the concentration of the free amine is maximized at the reaction site, driving the intramolecular cyclization forward with high kinetic favorability.
From an impurity control perspective, the TBTU/DBU system offers distinct advantages over the prior art. Conventional methods using DIPEA often resulted in purity levels as low as 79.6% to 83.9%, necessitating rigorous and yield-loss-inducing purification steps. In contrast, the new method consistently delivers crude product purities exceeding 97%, with final isolated purities reaching 98.2%. This suggests that the TBTU/DBU combination effectively suppresses the formation of oligomeric byproducts and epimerization at the chiral center. The mechanism likely involves a tighter ion pair or a more organized transition state that favors the intramolecular pathway over intermolecular polymerization. For quality assurance teams, this inherent selectivity reduces the burden on analytical testing and ensures that the final API meets stringent regulatory specifications for chiral purity and related substances with minimal additional processing.
How to Synthesize Lorlatinib Efficiently
The operational protocol for this synthesis is designed for robustness and reproducibility in a GMP environment. The process begins with the dissolution of the key intermediate, Compound A, in a polar aprotic solvent such as DMF, although toluene and acetonitrile are also viable options depending on solubility profiles. The addition of TBTU and DBU is performed under controlled stirring to ensure homogeneity before the system is gently heated. The reaction progress is monitored via TLC to confirm complete consumption of the starting material, typically within the 2 to 3-hour window. Upon completion, the reaction is quenched carefully with a saturated ammonium chloride solution, which neutralizes the base and decomposes any remaining activated species. The product is then extracted into an organic phase, concentrated, and washed with n-hexane to remove non-polar impurities, yielding the target molecule in high purity. For detailed standard operating procedures and safety guidelines, please refer to the technical documentation below.
- Dissolve Compound A in an organic solvent such as DMF or toluene, then add the condensing agent TBTU and the alkali promoter DBU.
- Heat the reaction mixture to 40-60°C and maintain this temperature for 2-3 hours while monitoring progress via TLC.
- Quench the reaction with ammonium chloride solution, extract with ethyl acetate, concentrate, wash with n-hexane, and dry to obtain pure Lorlatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this TBTU-mediated process offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the material bill of materials (BOM). By replacing expensive and less efficient coupling agents like HATU and COMU with the more cost-effective TBTU, and by reducing the required molar equivalents of both the coupling agent and the base, the direct material cost per kilogram of API is substantially lowered. Furthermore, the reduction in reagent load means that less waste is generated per unit of product, which significantly decreases the costs associated with waste disposal and environmental compliance. This aligns perfectly with the industry's push towards greener chemistry and sustainable manufacturing practices, potentially lowering the carbon footprint of the supply chain.
- Cost Reduction in Manufacturing: The elimination of excessive reagent usage directly impacts the bottom line. In traditional processes, the need for large excesses of coupling agents to drive the reaction to completion results in significant material waste. The new method achieves near-quantitative conversion with near-stoichiometric amounts of reagents. Additionally, the simplified workup procedure—requiring only a basic aqueous quench and solvent wash rather than complex chromatographic separations—reduces labor hours and solvent consumption. This streamlined workflow allows for faster batch turnover times, effectively increasing the throughput of existing manufacturing assets without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis and the availability of specialized reagents. TBTU and DBU are commodity chemicals with stable global supply chains, unlike some specialized peptide coupling reagents that may face shortages. The robustness of the reaction conditions (40-60°C) also means that the process is less sensitive to minor fluctuations in temperature control, reducing the risk of batch failures. This reliability ensures a steady flow of high-purity intermediates to downstream formulation partners, mitigating the risk of drug shortages in the market.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this method's mild thermal requirements make it highly scalable. The ability to run the reaction in solvents like toluene, which has a lower boiling point and is easier to recover than DMF, offers further flexibility for large-scale operations. Moreover, the high purity of the crude product minimizes the need for recrystallization solvents, reducing the overall solvent intensity of the process. This makes the technology attractive for contract manufacturing organizations (CMOs) looking to offer cost reduction in pharmaceutical manufacturing while adhering to strict environmental regulations regarding solvent emissions and waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring that stakeholders have a clear understanding of the process capabilities and limitations. Understanding these nuances is essential for making informed decisions about technology transfer and vendor selection.
Q: Why is the TBTU/DBU system superior to HATU/DIPEA for Lorlatinib synthesis?
A: The TBTU/DBU system provides significantly higher yields (up to 89.4% vs 29-78%) and purity (>98%) while operating under milder conditions and requiring shorter reaction times, reducing overall processing costs.
Q: What are the optimal reaction conditions for this macrocyclization?
A: The optimal conditions involve heating the system to 40-60°C for 2-3 hours in solvents like DMF or toluene, using a molar ratio of Compound A:TBTU:DBU of approximately 1:1:1.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial development due to reduced reagent consumption, simplified post-treatment operations, and high catalytic efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lorlatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs like Lorlatinib depends on a partner who can bridge the gap between laboratory innovation and industrial reality. Our technical team has extensively analyzed the TBTU/DBU cyclization pathway and is fully equipped to execute this process with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral integrity and trace impurity profiling, to guarantee that every batch meets the highest global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this high-yield route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Let us help you secure a competitive edge in the market with a supply of high-purity Lorlatinib that is both economically and technically superior.
