Advanced One-Pot Synthesis of Naphthoquinone Polycyclic Derivatives for Pharma and Materials
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access complex polycyclic scaffolds that serve as critical cores for bioactive molecules and functional materials. Patent CN113717182B introduces a groundbreaking methodology for the synthesis of naphthoquinone polycyclic derivatives, addressing the longstanding challenges associated with constructing these intricate molecular architectures. These derivatives, characterized by the general structure shown in Formula I, are not merely academic curiosities but represent potent candidates for anticancer, antitumor, and anti-inflammatory therapies, drawing inspiration from natural products like Calothrixin B and Kinamycin. Furthermore, their exceptional photophysical properties open doors for applications in fluorescent probing and optoelectronic materials. This technical insight report analyzes the transformative potential of this one-pot tandem cyclization strategy, offering a robust solution for reliable pharmaceutical intermediates supplier networks aiming to diversify their portfolios with high-value, complex heterocycles.
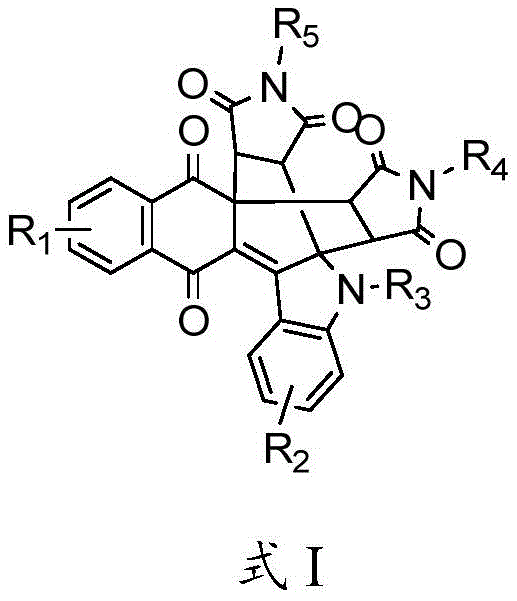
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of naphthoquinone-based polycyclic systems has been plagued by synthetic inefficiencies that hinder large-scale production and rapid structure-activity relationship (SAR) studies. Traditional approaches often necessitate the pre-functionalization of starting materials, requiring multiple discrete steps to install reactive handles before the final cyclization can occur. This multi-step lineage not only accumulates waste and increases the consumption of expensive reagents but also drastically lowers the overall yield due to losses at each isolation stage. Moreover, many existing protocols rely on precious metal catalysts or harsh reaction conditions that are incompatible with sensitive functional groups, limiting the substrate scope and complicating the purification process. For procurement teams, these factors translate into volatile supply chains and inflated costs for cost reduction in fine chemical manufacturing, making the adoption of such molecules in commercial drug development economically challenging.
The Novel Approach
In stark contrast, the methodology disclosed in CN113717182B utilizes a direct, one-pot tandem cyclization reaction that merges three distinct components—naphthoquinone, indole, and maleimide—into a single, highly complex polycyclic framework. As illustrated in the reaction scheme below, this approach eliminates the need for pre-functionalization, allowing for the direct assembly of the core structure under relatively mild thermal conditions. The reaction tolerates a broad spectrum of substituents on the indole and maleimide rings, enabling the rapid generation of diverse libraries for biological screening. By collapsing what was once a multi-step sequence into a single operational unit, this novel chemistry significantly enhances atom economy and process efficiency, providing a streamlined pathway for the commercial scale-up of complex pharmaceutical intermediates.
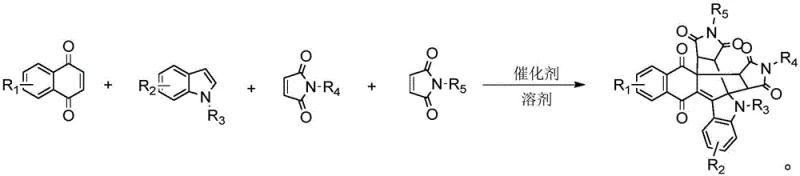
Mechanistic Insights into Lewis Acid-Catalyzed Tandem Cyclization
The success of this transformation hinges on the precise activation of the electrophilic and nucleophilic centers within the reaction mixture by a Lewis acid catalyst. The patent details the efficacy of various catalysts, including tris(pentafluorophenyl)borane (B(C5F6)3), metal triflates, and transition metal salts like FeCl3. Mechanistically, the catalyst likely coordinates with the carbonyl oxygens of the naphthoquinone or maleimide, increasing their electrophilicity and facilitating the initial Michael-type addition of the indole nucleophile. This is followed by a cascade of intramolecular cyclizations that forge the new C-C and C-N bonds required to close the polycyclic rings. The choice of solvent, such as acetonitrile or dichloroethane, plays a crucial role in stabilizing the charged intermediates and ensuring high conversion rates. Understanding this mechanistic nuance is vital for R&D directors aiming to optimize reaction parameters for specific substrate combinations to maximize yield and purity.
Impurity control in such complex tandem reactions is paramount, especially when targeting high-purity naphthoquinone derivatives for biological testing. The patented method demonstrates remarkable selectivity, minimizing the formation of side products that typically arise from uncontrolled polymerization or over-oxidation of the quinone moiety. The use of specific catalyst loading ratios (e.g., 5 mol% to 10 mol%) and controlled temperature profiles (ranging from room temperature to 120°C) allows for the fine-tuning of reaction kinetics. This kinetic control ensures that the desired cyclization pathway outcompetes potential degradation routes, resulting in cleaner crude reaction mixtures that simplify downstream purification. Such robustness in impurity profiling is essential for meeting the stringent quality standards required in the production of active pharmaceutical ingredients (APIs) and advanced functional materials.
How to Synthesize Naphthoquinone Polycyclic Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational protocols to ensure reproducibility and safety. The process begins with the precise weighing of the three key starting materials—naphthoquinone, substituted indole, and maleimide—according to the molar ratios specified in the patent embodiments. These components are introduced into a reaction vessel along with the chosen solvent and catalyst system. The detailed standardized synthesis steps for optimizing this reaction are provided in the guide below, which outlines the critical parameters for temperature control, reaction time, and workup procedures necessary to achieve the reported yields of up to 93%.
- Mix naphthoquinone, indole, and maleimide substrates in a reaction vessel with a suitable solvent such as acetonitrile.
- Add a Lewis acid catalyst like tris(pentafluorophenyl)borane or metal salts and heat the mixture to 100-120°C.
- After reaction completion, perform extraction and purification via column chromatography to isolate the polycyclic derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this one-pot synthesis methodology offers tangible strategic advantages beyond mere chemical elegance. By eliminating the need for pre-functionalized building blocks, the process drastically reduces the number of raw materials that need to be sourced, qualified, and stocked. This simplification of the bill of materials (BOM) directly contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering inventory holding costs and minimizing the risk of supply disruptions associated with specialized reagents. Furthermore, the use of commodity chemicals like naphthoquinone and common indoles ensures a stable and reliable supply base, mitigating the volatility often seen with exotic synthetic precursors.
- Cost Reduction in Manufacturing: The consolidation of multiple synthetic steps into a single pot operation inherently reduces energy consumption, labor hours, and solvent usage. Without the need for intermediate isolations and purifications, the overall processing time is significantly shortened, leading to substantial operational expenditure (OpEx) savings. The elimination of expensive transition metal catalysts in favor of more accessible Lewis acids further drives down the cost of goods sold (COGS), making these high-value derivatives more commercially viable for large-scale applications.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, enhances the resilience of the manufacturing process. This flexibility allows production facilities to adapt to varying resource availability without compromising product quality. Additionally, the high substrate tolerance means that alternative suppliers for specific indole or maleimide derivatives can be qualified more easily, reducing dependency on single-source vendors and strengthening the overall supply chain security for critical drug candidates.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, typically involving standard extraction and column chromatography, facilitates easier scale-up from gram to kilogram quantities. The reduced generation of chemical waste due to higher atom economy aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these naphthoquinone polycyclic derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and performance of this synthetic route.
Q: What are the primary applications of these naphthoquinone derivatives?
A: These compounds exhibit significant biological activity, including anticancer and anti-inflammatory properties, and possess excellent photophysical characteristics suitable for fluorescent materials and chemoreceptors.
Q: Does this synthesis method require pre-functionalized substrates?
A: No, a key advantage of this patented method is that it achieves tandem cyclization without the need for pre-functionalization of the substrates, simplifying the synthetic route.
Q: What catalysts are compatible with this reaction system?
A: The process supports a wide range of catalysts including Lewis acids like B(C5F6)3, metal triflates, and transition metal salts such as FeCl3 and Cu(OTf)2.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoquinone Derivatives Supplier
As the demand for complex heterocyclic scaffolds continues to grow in the fields of oncology and materials science, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from discovery to market. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of naphthoquinone derivatives meets the highest international standards, providing you with the confidence needed to advance your drug development pipelines.
We invite you to leverage our technical expertise to optimize your supply chain and accelerate your time-to-market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your strategic goals in the competitive landscape of pharmaceutical intermediates.
