Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for bioactive heterocyclic scaffolds, particularly those exhibiting broad therapeutic potential. Patent CN112538054B discloses a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a privileged structural motif found in numerous bioactive molecules ranging from antifungal agents to antitumor drugs. This innovation addresses critical bottlenecks in existing synthetic methodologies by introducing a transition metal palladium-catalyzed carbonylation tandem cyclization reaction. The process utilizes readily available chlorohydrazones and sodium azide as starting materials, achieving high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process chemistry, enabling the cost-effective production of high-value drug candidates.
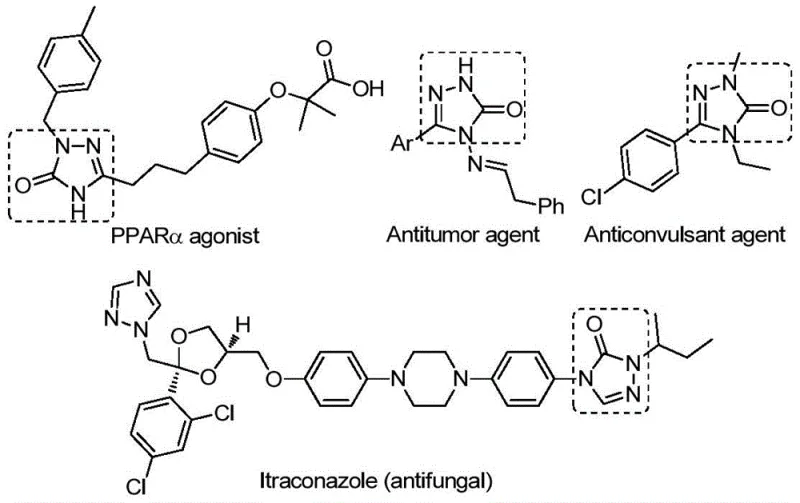
The versatility of the 1,2,4-triazole-3-one core is evident in its diverse biological applications, as illustrated by structures acting as PPARα agonists, antitumor agents, and anticonvulsants. The ability to efficiently construct this core with various substituents is paramount for medicinal chemistry campaigns aiming to optimize potency and pharmacokinetic profiles. The disclosed method not only simplifies the synthetic pathway but also enhances the overall atom economy, making it an attractive candidate for cost reduction in API manufacturing. By leveraging this patented technology, manufacturers can access a wider chemical space with reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one compounds has been plagued by significant technical challenges that hinder large-scale adoption. Traditional protocols often rely on the cyclization of benzoyl hydrazide with urea under strong basic conditions or the tandem reaction of hydrazides with isocyanates. These legacy methods frequently suffer from harsh reaction conditions, such as extreme temperatures or the use of corrosive reagents, which pose safety risks and complicate equipment maintenance. Furthermore, many conventional routes require pre-activation of substrates, adding unnecessary steps to the synthetic sequence and driving up material costs. The narrow substrate scope of these older methods limits the diversity of derivatives that can be accessed, often resulting in low yields when functionalized aryl or alkyl groups are introduced. Such inefficiencies create substantial barriers for supply chain heads looking to secure consistent volumes of high-purity 1,2,4-triazole-3-one derivatives.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated palladium-catalyzed system that dramatically streamlines the synthesis. By employing chlorohydrazones and sodium azide in the presence of a Pd2(dba)3 catalyst and Xantphos ligand, the reaction proceeds through a smooth carbonylation tandem cyclization. This method operates under relatively mild conditions, typically around 100°C in 1,4-dioxane, eliminating the need for extreme thermal stress. The use of TFBen as a solid carbon monoxide substitute is particularly ingenious, as it avoids the handling hazards associated with gaseous CO while ensuring efficient carbonyl insertion. This strategy not only improves safety but also enhances the reproducibility of the reaction across different batch sizes. The broad functional group tolerance allows for the introduction of diverse substituents at the R1 and R2 positions, facilitating the rapid generation of compound libraries for drug discovery without compromising yield or purity.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process further. The reaction likely initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, TFBen decomposes under thermal conditions to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, forming a key acyl palladium species. Subsequent interaction with sodium azide leads to the formation of an acyl azide intermediate, which undergoes a Curtius rearrangement to produce an isocyanate species. Finally, an intramolecular nucleophilic addition occurs, closing the ring to yield the final 1,2,4-triazole-3-one product. This elegant cascade minimizes the accumulation of unstable intermediates, thereby reducing the formation of side products and simplifying downstream purification.

Impurity control is inherently built into this catalytic cycle due to the high selectivity of the palladium system. The specific choice of ligands, such as Xantphos, stabilizes the active catalytic species and prevents non-productive decomposition pathways that often lead to tar formation in traditional syntheses. Moreover, the use of sodium azide in slight excess ensures complete consumption of the chlorohydrazone starting material, preventing the carryover of unreacted halides which can be difficult to remove later. The reaction conditions are optimized to balance reaction rate with selectivity, typically running for 16 to 30 hours to ensure full conversion without degrading the product. For quality assurance teams, this means a cleaner crude profile and higher recovery rates during isolation, directly translating to improved process economics and reduced waste generation.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol involves charging a reactor with the palladium catalyst, ligand, CO source, substrates, and solvent, followed by heating and stirring. The simplicity of the setup reduces the training burden on operators and minimizes the risk of procedural errors. Detailed standardized synthetic steps are provided below to guide technical teams in replicating these results with high fidelity.
- Combine Pd2(dba)3 catalyst, Xantphos ligand, TFBen carbon monoxide substitute, chlorohydrazone substrate, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers compelling advantages that address the core concerns of procurement managers and supply chain leaders. The shift towards more efficient synthetic routes is not merely a technical upgrade but a strategic move to enhance supply security and margin protection. By adopting this technology, companies can mitigate risks associated with volatile raw material markets and complex regulatory compliance regarding hazardous reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive and commercially available starting materials. Chlorohydrazones and sodium azide are commodity chemicals with stable supply chains, avoiding the premium pricing often associated with specialized organometallic reagents. Furthermore, the elimination of pre-activation steps reduces the overall number of unit operations, leading to significant savings in labor, energy, and solvent consumption. The high yields reported, reaching up to 96% in optimized examples, mean that less raw material is wasted per kilogram of product produced. This efficiency directly lowers the cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is critical for maintaining production schedules in the pharmaceutical sector. This method relies on robust reagents that are less susceptible to degradation or supply disruptions compared to sensitive organolithium or Grignard reagents used in alternative routes. The reaction tolerates a wide range of functional groups, meaning that variations in substrate quality from different suppliers are less likely to cause batch failures. Additionally, the use of a solid CO substitute like TFBen removes the logistical complexities and safety regulations associated with transporting and storing high-pressure carbon monoxide cylinders. This simplification of the supply chain infrastructure ensures a more reliable and resilient manufacturing process.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this protocol is designed with scalability in mind. The reaction conditions are moderate, avoiding the need for specialized high-pressure or cryogenic equipment, which facilitates easier technology transfer. From an environmental standpoint, the process generates fewer byproducts and utilizes solvents that can be recovered and recycled. The high atom economy of the tandem cyclization reduces the volume of chemical waste requiring treatment, aligning with modern green chemistry principles and stringent environmental regulations. This compliance reduces the burden on waste management systems and lowers the overall environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this route for their own production needs.
Q: What are the advantages of this Pd-catalyzed method over traditional synthesis?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this protocol uses cheap chlorohydrazones and operates under mild conditions with high substrate tolerance and excellent yields up to 96%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly mentions scalability from milligram to mole scales, utilizing commercially available reagents and standard post-processing techniques like filtration and chromatography.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen acts as a solid carbon monoxide substitute that releases CO under heating conditions, facilitating the insertion into the carbon-palladium bond to form the key acyl palladium intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the one described in CN112538054B for the production of complex heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity 1,2,4-triazole-3-one derivatives that meet the exacting standards of the pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative synthesis for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate your drug development pipeline while optimizing your manufacturing costs and securing your supply chain for the future.
