Advanced Palladium-Catalyzed Carbonylation for Scalable 1,2,4-Triazole-3-one Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 1,2,4-Triazole-3-one Production
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance efficiency with economic viability, particularly for nitrogen-containing heterocycles which serve as privileged scaffolds in drug discovery. A significant breakthrough in this domain is detailed in patent CN112538054B, which discloses a highly efficient preparation method for 1,2,4-triazole-3-one compounds. These heterocyclic structures are not merely academic curiosities; they are foundational cores found in a vast array of bioactive molecules exhibiting potent antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant activities. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies allows us to offer superior process solutions that align with the rigorous demands of modern API manufacturing. The innovation lies in transitioning from cumbersome multi-step protocols to a streamlined, transition metal-catalyzed carbonylation tandem cyclization reaction that utilizes readily accessible starting materials.
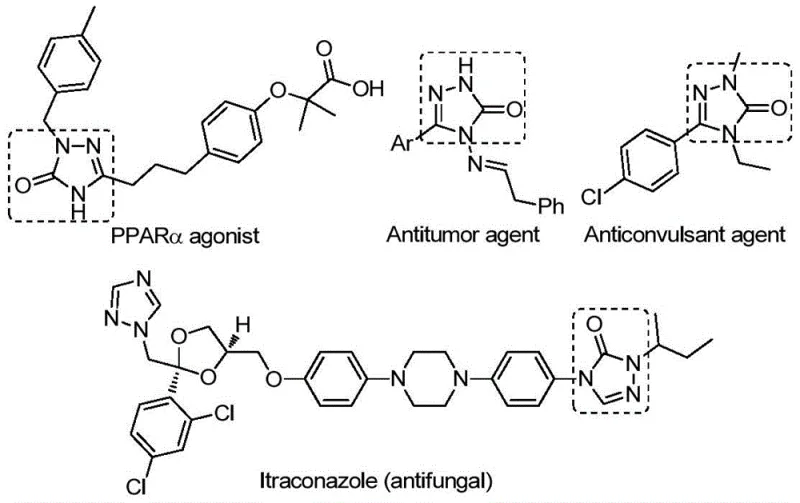
The strategic value of this technology cannot be overstated for R&D directors focusing on library expansion. The patent highlights that 1,2,4-triazole-3-one compounds have been successfully employed as tyrosinase inhibitors, CB receptor modulators, angiotensin AT1 receptor antagonists, and NK1 antagonists. By mastering the synthesis of these cores, chemical manufacturers can significantly accelerate the development pipeline for new therapeutic agents. The method described in CN112538054B specifically addresses the historical bottlenecks associated with constructing this five-membered nitrogen-containing ring system, offering a pathway that is both operationally simple and chemically elegant. This sets the stage for a deeper technical evaluation of how this novel approach outperforms legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazol-3-one compounds has been plagued by significant operational and chemical inefficiencies that hinder large-scale adoption. Traditional literature reports typically rely on methods such as the cyclization of benzoyl hydrazide with urea under the influence of potassium hydroxide, or the tandem cyclization of hydrazides with isocyanates. Other established routes involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines. These conventional pathways suffer from a multitude of drawbacks, including excessively harsh reaction conditions that pose safety risks, complicated multi-step sequences that erode overall yield, and the frequent necessity for pre-activation of reaction substrates. Furthermore, these older methods often exhibit narrow substrate scopes, failing to tolerate diverse functional groups, which severely limits their utility in the synthesis of complex drug candidates requiring specific substitution patterns.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112538054B represents a paradigm shift towards green and efficient chemistry. This novel approach employs a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, utilizing cheap and easily obtainable chlorohydrazones and sodium azide as the primary starting materials. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at moderate temperatures, eliminating the need for extreme conditions. Crucially, this method demonstrates exceptional substrate compatibility, allowing for the synthesis of 1,2,4-triazole-3-one compounds substituted with various groups through rational substrate design. The operational simplicity is further enhanced by the use of TFBen (phenyl 1,3,5-tricarboxylate) as a solid carbon monoxide substitute, bypassing the hazards associated with handling gaseous CO. This streamlined process not only improves reaction efficiency but also widens the practical applicability of the synthesis for industrial purposes.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
For the discerning R&D director, understanding the mechanistic underpinnings of this transformation is vital for troubleshooting and optimization. The reaction likely initiates with the oxidative addition of the palladium(0) catalyst, specifically tris(dibenzylideneacetone)dipalladium (Pd2(dba)3), into the carbon-chlorine bond of the chlorohydrazone substrate (Formula II). This step generates a critical divalent palladium intermediate. Simultaneously, the additive TFBen undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, forming an acyl-palladium intermediate. Subsequently, this intermediate reacts with sodium azide to generate an acyl azide compound. This acyl azide then undergoes a classic Curtius rearrangement to form an isocyanate intermediate, which finally participates in an intramolecular nucleophilic addition reaction to close the ring, yielding the final 1,2,4-triazole-3-one compound (Formula I). This intricate cascade effectively constructs the heterocyclic core in a single pot.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The use of specific ligands like Xantphos (4,5-bis(diphenylphosphino)-9,9-dimethylxanthene) in a precise 1:2 molar ratio with the palladium source ensures high catalytic turnover and selectivity. The choice of aprotic solvents, particularly 1,4-dioxane, is paramount as it effectively promotes the reaction progress compared to polar aprotic solvents like DMSO or DMF which were found to be less conducive. The broad tolerance for functional groups on both the R1 and R2 positions—ranging from alkyl and aryl groups to halogens and alkoxy substituents—suggests that side reactions such as dehalogenation or unwanted polymerization are minimized under these optimized conditions. This robustness ensures that the resulting high-purity pharmaceutical intermediates meet stringent quality specifications required for downstream drug synthesis.

How to Synthesize 1,2,4-Triazole-3-one Efficiently
The execution of this synthesis requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a generalized procedure where the palladium catalyst, ligand, CO source, chlorohydrazone, and sodium azide are combined in an organic solvent. The reaction is typically conducted at temperatures between 100°C and 120°C for a duration of 16 to 30 hours. Post-reaction processing involves standard filtration and purification techniques, such as silica gel column chromatography, to isolate the target molecule. While the laboratory scale (mmol level) has been proven effective, the principles remain consistent for larger batches. For a detailed breakdown of the specific operational steps, reagent quantities, and workup procedures, please refer to the standardized guide below.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), TFBen, chlorohydrazone substrate, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to allow the carbonylation and cyclization cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,2,4-triazole-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from complex, multi-step traditional syntheses to a one-pot catalytic process fundamentally alters the cost structure of manufacturing these valuable intermediates. By leveraging cheap and commercially available starting materials like chlorohydrazones and sodium azide, manufacturers can achieve substantial cost savings in raw material acquisition. Furthermore, the elimination of hazardous gaseous carbon monoxide in favor of a solid CO surrogate (TFBen) drastically simplifies the engineering controls required, reducing capital expenditure on specialized high-pressure reactors and safety infrastructure.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the high atom economy and the use of inexpensive reagents. Unlike traditional methods that may require expensive activated substrates or harsh reagents leading to waste, this palladium-catalyzed route utilizes catalytic amounts of metal and ligand. Although palladium is a precious metal, the high efficiency and turnover of the catalyst system, combined with the ability to recover and recycle the metal from the reaction mixture, ensure that the overall catalyst cost remains manageable. The simplified post-treatment process, involving basic filtration and chromatography, reduces labor hours and solvent consumption, contributing to a leaner manufacturing cost profile.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on commodity chemicals. Chlorohydrazones can be rapidly synthesized from corresponding acid chlorides, hydrazines, and triphenylphosphine, all of which are bulk chemicals with stable global supply chains. Sodium azide and the ligand Xantphos are also widely available from multiple vendors, mitigating the risk of single-source dependency. This diversification of the supply base ensures that production schedules are not disrupted by raw material shortages, providing a reliable flow of pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration at the mmol scale with clear pathways to kilogram and tonnage production. The use of 1,4-dioxane as a preferred solvent, while requiring careful handling, is a well-established industrial solvent with existing recovery and recycling protocols. Moreover, the reaction generates fewer byproducts compared to condensation methods that produce stoichiometric amounts of salt waste. This reduced waste profile aligns with increasingly stringent environmental regulations, lowering the costs associated with waste disposal and environmental compliance, thereby enhancing the long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production capabilities.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this novel approach utilizes cheap chlorohydrazones and avoids high-pressure CO gas, offering milder conditions and broader functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method uses commercially available reagents and standard organic solvents like dioxane, making it highly adaptable for scale-up from laboratory to commercial manufacturing without specialized high-pressure equipment.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen acts as a solid carbon monoxide substitute that releases CO under heating conditions, facilitating the formation of the acyl-palladium intermediate essential for the subsequent Curtius rearrangement and cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert chemists has thoroughly analyzed the potential of the Pd-catalyzed carbonylation route described in CN112538054B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one intermediate meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce time-to-market for your therapeutic candidates. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you navigate the complexities of fine chemical synthesis with confidence and precision.
