Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale Manufacturing
Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale Manufacturing
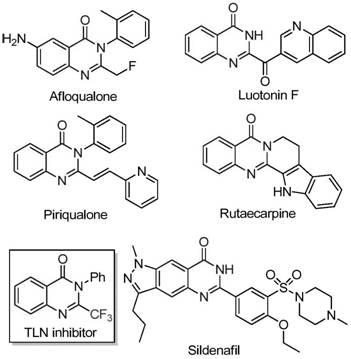
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring systems are pivotal structural motifs found in numerous bioactive molecules, exhibiting a wide spectrum of pharmacological activities including anti-cancer, anticonvulsant, anti-inflammatory, and antifungal properties. The strategic introduction of a trifluoromethyl group into these heterocycles further enhances their metabolic stability, lipophilicity, and bioavailability, making them prime candidates for next-generation drug development. This report analyzes the technical merits of this novel iron-catalyzed route, highlighting its potential to revolutionize the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons like trifluoroacetic anhydride or ethyl trifluoroacetate with substrates such as anthranilamide or isatoic anhydride. While effective in laboratory settings, these traditional pathways are plagued by significant industrial drawbacks. They often necessitate severe reaction conditions that pose safety risks and energy inefficiencies during scale-up. Furthermore, the reliance on expensive fluorinated reagents drives up the raw material costs substantially, creating a bottleneck for cost-sensitive manufacturing processes. The narrow substrate scope of these older methods also limits the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR), thereby slowing down the drug discovery pipeline. Additionally, the purification of products from these harsh reactions can be cumbersome, leading to lower overall yields and increased waste generation.
The Novel Approach
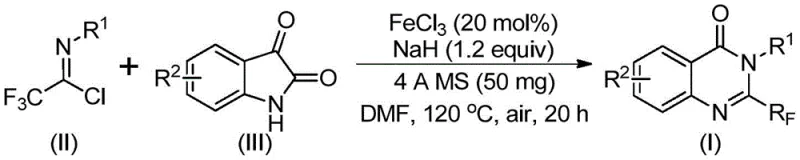
In stark contrast, the methodology outlined in patent CN111675662B offers a paradigm shift by utilizing readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials. This innovative route employs a cheap and abundant metal iron catalyst, specifically ferric chloride (FeCl3), to drive the cyclization. The process is characterized by its operational simplicity and mild conditions, proceeding effectively in common organic solvents like DMF. By shifting away from expensive anhydrides to more accessible imidoyl chlorides, the method drastically reduces the cost of goods sold (COGS). The reaction demonstrates excellent functional group tolerance, allowing for the synthesis of a diverse array of derivatives with substituents at various positions on the aromatic rings. This versatility is crucial for generating libraries of compounds for biological screening without the need for protecting group strategies or specialized reagents.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of base-promoted nucleophilic attack and transition metal catalysis. Initially, the strong base, sodium hydride (NaH), facilitates the formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This step generates a transient trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring closure. The presence of ferric chloride is critical at this stage, acting as a Lewis acid catalyst that promotes the decarbonylation and cyclization steps. The iron center likely coordinates with the carbonyl oxygen and the nitrogen atoms, lowering the activation energy barrier for the intramolecular rearrangement. This catalytic cycle allows the reaction to proceed smoothly even with sterically hindered or electronically diverse substrates, ensuring high conversion rates. The use of 4A molecular sieves in the reaction mixture further drives the equilibrium forward by sequestering water or other small molecule byproducts, thereby enhancing the overall efficiency of the transformation.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives. The specific coordination environment provided by the iron catalyst minimizes side reactions such as polymerization or hydrolysis of the sensitive imidoyl chloride moiety. The reaction conditions are tuned to favor the formation of the thermodynamically stable quinazolinone ring system, resulting in a clean crude reaction profile. This inherent selectivity simplifies the downstream purification process, as fewer byproducts need to be removed via column chromatography or recrystallization. For R&D teams focused on purity specifications, understanding this mechanism allows for precise optimization of catalyst loading and base equivalents to suppress trace impurities, ensuring the final API intermediate meets stringent regulatory standards.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these valuable intermediates with high reproducibility. The procedure involves a straightforward one-pot reaction where all reagents are combined in a Schlenk tube or reactor under controlled atmospheric conditions. The specific stoichiometry of ferric chloride to sodium hydride is optimized to balance catalytic activity with safety, typically employing a molar ratio that ensures complete consumption of the limiting reagent. The reaction temperature profile is critical, starting at a moderate 40°C to initiate the coupling and then ramping up to 120°C to drive the cyclization to completion. Detailed standardized synthetic steps for replicating this high-yielding process are provided in the technical guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C and continue reacting for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-catalyzed technology translates into tangible strategic benefits. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing exotic and costly fluorinating agents with commodity chemicals like isatin and simple aromatic amines, companies can mitigate supply risk and stabilize pricing. The elimination of precious metal catalysts removes the need for expensive metal scavenging steps and complex waste treatment protocols associated with heavy metal residues. This not only lowers the direct manufacturing costs but also reduces the environmental footprint of the production facility, aligning with modern green chemistry initiatives. Furthermore, the scalability of the process from gram to multi-kilogram levels ensures a reliable supply continuity for clinical and commercial demands.
- Cost Reduction in Manufacturing: The substitution of expensive reagents with low-cost alternatives like ferric chloride and readily available isatins results in significant raw material savings. The process avoids the use of precious metals such as palladium or rhodium, which are subject to volatile market pricing and supply constraints. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, leading to a leaner and more cost-efficient production model. These cumulative factors contribute to a substantially lower cost of goods, providing a competitive edge in the marketplace.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commercially available in bulk quantities from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch output. This reliability is crucial for maintaining uninterrupted supply chains for downstream pharmaceutical customers who require just-in-time delivery of critical intermediates. The ability to source raw materials locally in many regions further shortens lead times and reduces logistics costs.
- Scalability and Environmental Compliance: The reaction operates under relatively mild conditions compared to traditional high-pressure or cryogenic methods, making it easier to scale up in standard glass-lined or stainless steel reactors. The use of iron, a non-toxic and abundant metal, simplifies waste disposal and regulatory compliance regarding heavy metal limits in final products. The high atom economy of the cyclization step minimizes the generation of hazardous byproducts, supporting sustainable manufacturing practices. This alignment with environmental, social, and governance (ESG) goals is increasingly important for securing contracts with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this FeCl3-catalyzed method over traditional synthesis?
A: This method utilizes inexpensive iron catalysts and readily available starting materials like isatin and trifluoroethylimidoyl chloride, avoiding the severe reaction conditions and expensive substrates often required in conventional trifluoroacetic anhydride routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to its simple operation, cheap raw materials, and good functional group tolerance.
Q: What represents the optimal reaction temperature profile for maximum yield?
A: The optimal protocol involves a two-stage heating process: initial reaction at 40°C for 8-10 hours followed by heating to 120°C for 18-20 hours, which ensures high conversion rates across various substrate derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel derivatives or bulk supply of established intermediates, our infrastructure is designed to support your growth from preclinical research through to commercial launch.
We invite you to contact our technical procurement team to discuss how this advanced FeCl3-catalyzed technology can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your unique requirements, ensuring a seamless partnership that drives value and innovation in your pharmaceutical pipeline.
