Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
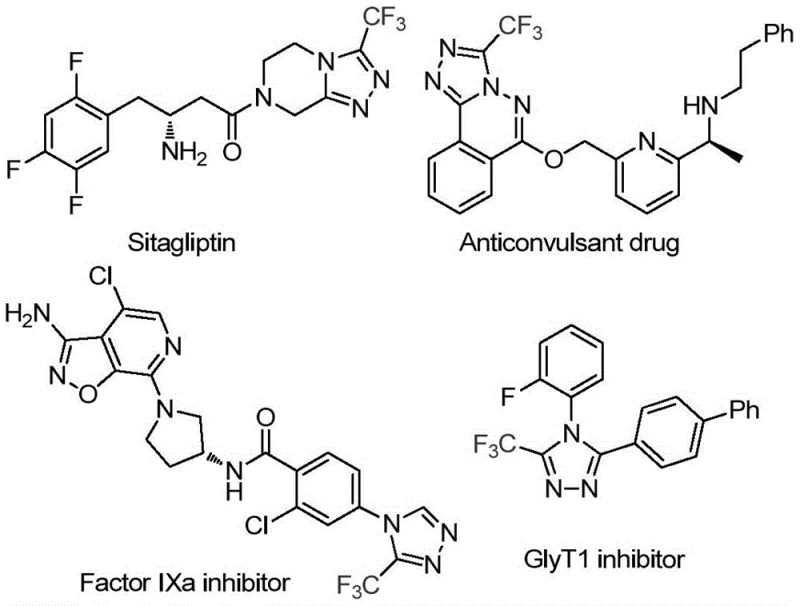
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A recent technological breakthrough documented in patent CN114920707B introduces a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, a structural motif prevalent in numerous high-value therapeutic agents. As illustrated in the provided chemical structures, these triazole cores are integral to the design of potent drugs such as Sitagliptin, Factor IXa inhibitors, and GlyT1 inhibitors, where the trifluoromethyl group plays a pivotal role in enhancing metabolic stability and lipophilicity. This novel methodology distinguishes itself by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a reaction medium but as a direct carbon source, thereby streamlining the synthetic pathway and reducing the reliance on specialized reagents. For R&D directors and procurement specialists alike, this represents a significant shift towards more atom-economical and operationally simple processes that can be readily adapted for commercial scale-up without compromising on purity or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted 1,2,4-triazoles has often relied on multi-step sequences involving harsh reaction conditions, expensive transition metal catalysts, or sensitive reagents that necessitate rigorous exclusion of moisture and oxygen. Conventional protocols frequently require the use of specialized C1 synthons which can be costly and difficult to handle on a large scale, leading to increased production costs and complex waste streams. Furthermore, many existing methods suffer from limited substrate scope, failing to accommodate diverse functional groups without significant loss in efficiency or requiring extensive protection-deprotection strategies. The necessity for anhydrous and anaerobic environments in many legacy processes imposes a substantial burden on manufacturing infrastructure, demanding specialized equipment such as Schlenk lines or gloveboxes, which drastically increases capital expenditure and operational complexity for fine chemical manufacturers aiming for high-throughput production.
The Novel Approach
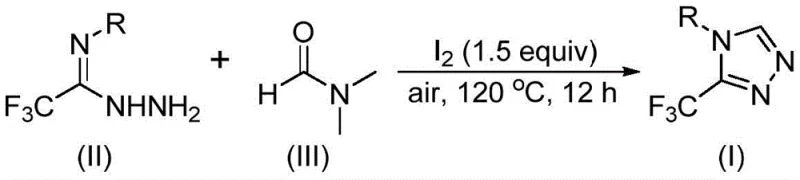
In stark contrast to these cumbersome traditional routes, the method disclosed in CN114920707B offers a remarkably streamlined alternative that leverages the dual functionality of DMF. By employing molecular iodine as a mild promoter under an air atmosphere, this process facilitates a tandem cyclization reaction that efficiently constructs the triazole ring while incorporating the trifluoromethyl group in a single pot. The reaction proceeds at moderate temperatures ranging from 110 to 130°C over a period of 10 to 15 hours, conditions that are easily achievable in standard industrial reactors without the need for cryogenic cooling or high-pressure vessels. This approach not only simplifies the operational workflow by eliminating the need for inert gas protection but also utilizes DMF, a solvent that is inexpensive, readily available globally, and familiar to process chemists, thereby ensuring a smooth transition from laboratory discovery to pilot plant operations.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the versatile reactivity of DMF, which can act as a carbon source through either its formyl or N-methyl group depending on the specific reaction pathway activated by the iodine promoter. When the formyl group serves as the carbon source, it undergoes a condensation reaction with the trifluoroethyliminohydrazide substrate to generate a hydrazone intermediate, which subsequently experiences intramolecular cyclization accompanied by the elimination of dimethylamine to yield the desired 3-trifluoromethyl-1,2,4-triazole product. Alternatively, when the N-methyl group participates, DMF initially reacts with iodine to form an amine salt species, which then engages in nucleophilic addition with the hydrazide followed by elimination steps to generate an azadiene intermediate that undergoes oxidative aromatization. This dual-pathway capability underscores the robustness of the method, allowing for flexibility in substrate design while maintaining high reaction efficiency across a broad range of electronic environments on the aromatic ring.
From an impurity control perspective, the use of molecular iodine as a promoter is particularly advantageous because it avoids the introduction of heavy metal contaminants that are often associated with transition metal catalysis, thus simplifying the downstream purification process and ensuring the final product meets stringent pharmaceutical purity specifications. The reaction mechanism inherently favors the formation of the thermodynamically stable triazole ring, minimizing the generation of regioisomers or side products that could complicate the isolation of the target molecule. Moreover, the oxidative nature of the cyclization step ensures that any partially reduced intermediates are driven towards the fully aromatic product, resulting in a cleaner reaction profile that reduces the load on purification columns and enhances the overall mass balance of the process, a critical factor for maintaining cost-effectiveness in large-scale manufacturing.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol described in the patent provides a clear and reproducible framework for generating high-purity triazole derivatives suitable for drug development pipelines. The procedure involves simply combining the trifluoroethyliminohydrazide starting material with molecular iodine in DMF solvent, followed by heating under air to drive the cyclization to completion. This straightforward setup minimizes the risk of operator error and reduces the training time required for technical staff, making it an ideal candidate for technology transfer between sites. For detailed standardized synthesis steps and specific workup procedures, please refer to the guide below.
- Combine molecular iodine, trifluoroethyliminohydrazide, and DMF solvent in a reaction vessel under air.
- Heat the mixture to 110-130°C and maintain reaction for 10-15 hours to ensure complete conversion.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the drastic simplification of the raw material list; by utilizing DMF as both the solvent and the carbon source, the process eliminates the need to procure and inventory specialized, high-cost C1 building blocks that are often subject to volatile market pricing and supply constraints. This consolidation of reagents not only reduces the direct material cost per kilogram of product but also simplifies warehouse management and reduces the safety risks associated with handling a larger variety of hazardous chemicals, thereby lowering insurance and compliance overheads for the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the removal of strict inert atmosphere requirements lead to substantial operational savings. Without the need for nitrogen or argon purging systems and the associated energy consumption for maintaining low-oxygen environments, the utility costs for the reaction are significantly lowered. Furthermore, the use of common solvents and reagents like iodine ensures that the process remains resilient against supply chain disruptions, as these materials are produced in massive volumes globally and are unlikely to face shortages, providing a stable cost structure for long-term production contracts.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture, translates directly into improved batch consistency and reduced failure rates. In a commercial setting, batch failures due to accidental air leaks or moisture ingress are a major cause of supply delays; this method's inherent stability mitigates such risks, ensuring reliable delivery schedules for downstream API manufacturers. Additionally, the broad substrate scope demonstrated in the patent allows for the rapid synthesis of various analogues from readily available aromatic amines, enabling agile responses to changing market demands for different drug candidates without the need for extensive process re-optimization.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of standard equipment and the absence of pyrophoric or highly toxic reagents. The waste profile is also more manageable, as the primary byproducts are derived from the solvent and iodine, which can often be recovered or treated using established waste management protocols. This aligns well with modern green chemistry principles and increasingly stringent environmental regulations, reducing the liability and disposal costs associated with heavy metal waste streams, thus making the process more sustainable and attractive for environmentally conscious corporate partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed experimental data and beneficial effects outlined in the patent documentation. These insights are intended to clarify the practical implications of adopting this method for your specific production needs and to highlight the versatility of the chemistry involved.
Q: What are the key advantages of using DMF as a carbon source in this synthesis?
A: Using DMF eliminates the need for separate, expensive C1 building blocks, significantly reducing raw material costs and simplifying the reaction setup by serving as both solvent and reactant.
Q: Does this process require strict anhydrous or oxygen-free conditions?
A: No, the patented method operates efficiently under standard air atmosphere, removing the capital and operational expenses associated with inert gas lines and glovebox setups.
Q: What is the substrate scope for the R-group in the triazole product?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating and electron-withdrawing substituents like halogens, alkyls, and trifluoromethyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the fast-paced pharmaceutical landscape. Our team of expert process chemists has extensively evaluated the technology described in CN114920707B and confirmed its potential for delivering high-quality intermediates with exceptional purity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require material for early-stage clinical trials or full-scale commercial launch, our facilities are equipped to meet your volume requirements with consistent quality. Our rigorous QC labs employ state-of-the-art analytical techniques to verify stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole we supply meets the exacting standards required for GMP manufacturing.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs, helping you optimize your budget without sacrificing quality. We encourage you to contact our technical procurement team today to request specific COA data for our available batches and to discuss route feasibility assessments for your target molecules, ensuring a seamless and successful supply partnership.
