Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are ubiquitous in modern medicinal chemistry, found in the molecular frameworks of prominent drugs such as Sitagliptin and Deferasirox, where the trifluoromethyl group plays a pivotal role in enhancing metabolic stability and lipophilicity. The innovation lies in a novel transition metal co-catalyzed system that overcomes the limitations of classical approaches, offering a streamlined pathway for generating these valuable intermediates.
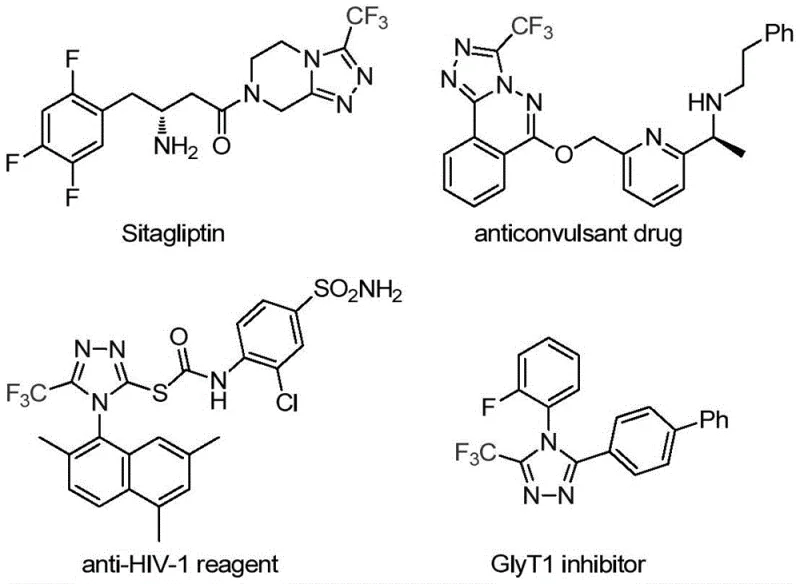
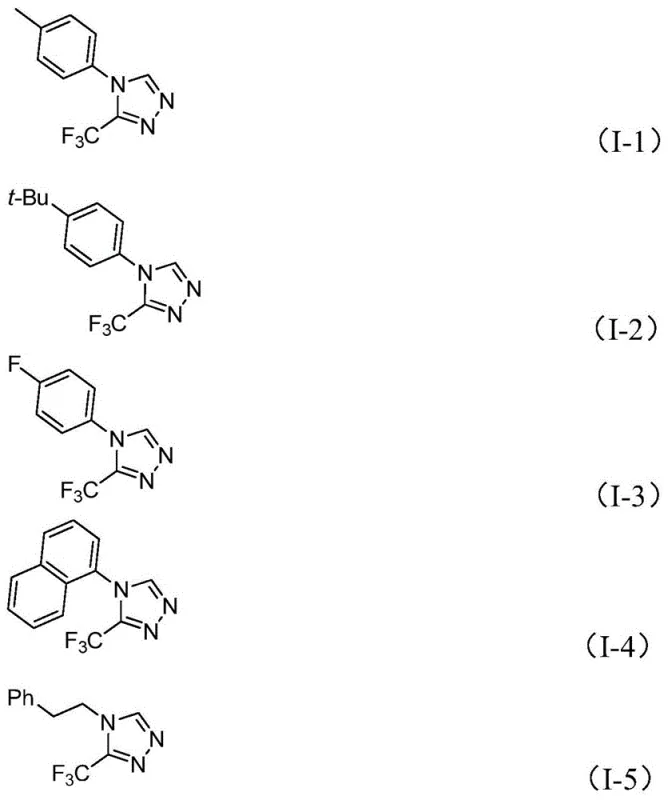
For research and development teams, the ability to access diverse triazole derivatives with high purity is essential for lead optimization. This patent provides a versatile platform where the R group can be extensively modified, ranging from simple phenyl rings to complex naphthyl or phenethyl groups. By leveraging this technology, manufacturers can secure a reliable supply of high-purity pharmaceutical intermediates that are crucial for the development of next-generation therapeutics targeting various diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that often suffer from significant operational drawbacks. Traditional literature reports primarily describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical routes frequently require harsh reaction conditions, involving strong acids or bases and elevated temperatures that can degrade sensitive functional groups. Furthermore, alternative multi-component reactions utilizing diazonium salts or trifluorodiazoethane often involve hazardous reagents and complex safety protocols, making them less desirable for large-scale manufacturing environments. The limited scope of substrates in these older methods also restricts the chemical diversity accessible to medicinal chemists, hindering the rapid exploration of structure-activity relationships.
The Novel Approach
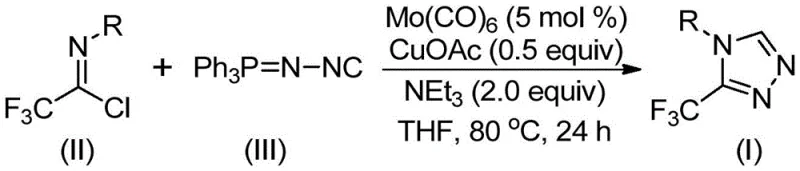
In stark contrast, the method disclosed in patent CN113307778A introduces a transformative strategy utilizing a molybdenum and copper co-catalytic system. This approach employs trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, reacting them in the presence of molybdenum hexacarbonyl and cuprous acetate. The reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C in common organic solvents like THF. This shift not only simplifies the operational complexity but also dramatically expands the substrate scope, allowing for the introduction of electron-donating and electron-withdrawing groups without compromising yield. The result is a robust, high-efficiency process that converts readily available starting materials into complex heterocyclic products with exceptional atom economy.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthesis hinges on the synergistic interaction between the molybdenum and copper catalysts, which facilitates a unique [3+2] cycloaddition pathway. Mechanistically, the molybdenum hexacarbonyl acts as a metal activator for the functionalized isonitrile, forming a reactive metal-isocyanide complex that primes the molecule for nucleophilic attack. Simultaneously, the cuprous acetate promotes the cycloaddition of this activated species to the trifluoroethylimidoyl chloride. This dual-catalyst system lowers the activation energy barrier significantly, enabling the formation of the five-membered triazole ring under thermal conditions that would be insufficient for non-catalyzed variants. Following the ring closure, a subsequent elimination of triphenylphosphine oxide, driven by trace water in the system, yields the final stable 3-trifluoromethyl-substituted 1,2,4-triazole product.
From an impurity control perspective, this mechanism offers distinct advantages. The mild thermal window (70-90°C) minimizes the formation of thermal degradation byproducts that often plague high-temperature cyclizations. Additionally, the specificity of the metal-ligand interactions ensures high regioselectivity, reducing the generation of isomeric impurities that are difficult to separate. The patent data demonstrates that this method tolerates a wide array of substituents, including halogens, alkyl groups, and even sensitive nitro groups, as evidenced by the successful synthesis of derivatives like I-3 (fluoro-substituted) and I-10 (nitro-substituted). This broad tolerance confirms the robustness of the catalytic cycle against electronic variations in the substrate.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized stoichiometry and reaction parameters outlined in the patent. The process is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes for every step, although standard Schlenk techniques are recommended for optimal catalyst longevity. The following guide summarizes the critical operational phases required to achieve the high yields reported in the experimental section, ensuring reproducibility and safety during the handling of reactive isonitriles and metal carbonyls.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents compelling economic and logistical benefits. The shift towards this methodology addresses several critical pain points associated with the sourcing of complex heterocyclic intermediates. By utilizing a catalyst system based on abundant transition metals and avoiding precious metals like palladium or rhodium, the overall cost of goods sold (COGS) can be significantly optimized. Furthermore, the reliance on commercially available starting materials reduces the risk of supply chain disruptions caused by specialized reagent shortages.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive catalysts such as cuprous acetate and molybdenum hexacarbonyl, which are far more cost-effective than noble metal alternatives. The reaction achieves high conversion rates, which minimizes the loss of valuable starting materials and reduces the burden on downstream purification processes. Additionally, the simplified workup procedure, involving basic filtration and standard column chromatography, eliminates the need for complex extraction or crystallization steps that often inflate manufacturing costs. This streamlined workflow translates directly into substantial cost savings in API manufacturing without sacrificing product quality.
- Enhanced Supply Chain Reliability: The starting materials, specifically trifluoroethylimidoyl chloride and functionalized isonitriles, are derived from commodity chemicals that are widely produced and stocked by global chemical suppliers. This accessibility ensures a stable and continuous supply of raw materials, mitigating the risks associated with single-source dependencies. The robustness of the reaction conditions also means that production schedules are less likely to be impacted by minor fluctuations in environmental controls or reagent quality, providing a predictable and reliable timeline for project milestones and delivery commitments.
- Scalability and Environmental Compliance: The patent explicitly notes the successful expansion of this method to gram-level scales, indicating a clear pathway for kilogram and ton-scale production. The use of THF as a preferred solvent aligns with standard industrial practices, facilitating easy solvent recovery and recycling systems that support green chemistry initiatives. Moreover, the high atom economy of the cycloaddition reaction results in reduced waste generation compared to traditional multi-step syntheses, simplifying waste treatment protocols and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects.
Q: What are the key advantages of this Mo/Cu co-catalyzed method over traditional synthesis?
A: This method utilizes cheap and readily available starting materials like functionalized isonitriles and trifluoroethylimidoyl chloride. It operates under mild conditions (70-90°C) compared to harsher traditional cyclization methods, offering higher reaction efficiency and broader substrate tolerance for diverse functional groups.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states that the method can be expanded to gram-level reactions with high efficiency. The use of standard organic solvents like THF and common catalysts suggests strong potential for commercial scale-up without requiring exotic equipment.
Q: What types of substituents are tolerated on the aromatic ring?
A: The process demonstrates excellent functional group tolerance. It successfully accommodates various substituents including alkyl groups (methyl, t-butyl), halogens (fluoro, chloro), alkoxy groups, and even electron-withdrawing groups like nitro, allowing for the synthesis of a wide library of derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your upcoming projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
