Advanced Asymmetric Reduction Technology for High-Purity Mitiglinide Intermediate Manufacturing
Advanced Asymmetric Reduction Technology for High-Purity Mitiglinide Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for active pharmaceutical ingredients (APIs) that balance high stereochemical control with economic viability. A pivotal advancement in this domain is documented in Chinese Patent CN101346352B, which discloses a novel asymmetric reduction method for producing (2S)-2-benzyl-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid, a critical intermediate for the antidiabetic drug Mitiglinide. This technology addresses longstanding challenges in the synthesis of benzylsuccinic acid derivatives by employing a specialized class of urea-type pyrrolidine diphosphine ligands coordinated with rhodium. For R&D directors and procurement specialists, this patent represents a significant leap forward, offering a pathway to achieve exceptional optical purity and reaction efficiency that surpasses conventional carbamate-based catalytic systems. The ability to produce this key pharmaceutical intermediate with high conversion rates and minimized catalyst loading translates directly into enhanced supply chain reliability and reduced manufacturing costs for global healthcare providers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Mitiglinide and its salts has relied on asymmetric hydrogenation techniques utilizing chiral diphosphine complexes, such as those based on ruthenium or rhodium with carbamate-type ligands like BPPM. However, these traditional methodologies suffer from inherent inefficiencies that hinder large-scale optimization. Specifically, the reaction kinetics associated with carbamate-type ligands are often sluggish, necessitating prolonged reaction times to reach acceptable conversion levels. Furthermore, to achieve satisfactory reaction rates, manufacturers are frequently forced to operate with low substrate-to-catalyst (S/C) molar ratios, which implies the consumption of substantial quantities of expensive noble metal catalysts. This not only inflates the raw material costs significantly but also complicates the downstream purification process, as removing trace amounts of heavy metals from the final API becomes a rigorous and costly regulatory requirement. Additionally, the low melting point of Mitiglinide makes recrystallization difficult, meaning that any lack of selectivity or incomplete conversion in the reduction step creates severe bottlenecks in obtaining pharmaceutical-grade purity.
The Novel Approach
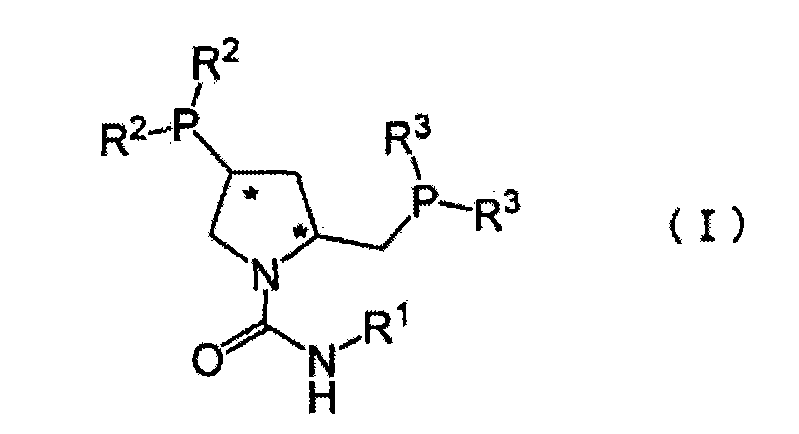
The innovative methodology outlined in the patent data fundamentally alters the economic and technical landscape of this synthesis by introducing urea-type pyrrolidine diphosphine compounds as the chiral ligands. Unlike their carbamate predecessors, these urea-based ligands, such as (2S,4S)-N-phenylaminocarbonyl-4-diphenylphosphino-2-diphenylphosphinomethylpyrrolidine (PCPPM), exhibit extraordinary catalytic activity and substrate specificity. This breakthrough allows the reaction to proceed rapidly even at high substrate concentrations, achieving near-quantitative conversion in a fraction of the time required by older methods. Crucially, the system supports extremely high S/C ratios, ranging from 5000 to over 20000, which drastically reduces the requisite amount of rhodium catalyst. For a reliable pharmaceutical intermediate supplier, this translates to a process that is not only faster and more selective but also inherently more cost-effective and environmentally sustainable due to the minimization of precious metal waste. The result is a streamlined manufacturing route capable of delivering high-purity intermediates with optical purity consistently exceeding 95% e.e., effectively bypassing the purification difficulties associated with previous generations of synthetic technology.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise molecular architecture of the catalyst system, which facilitates a highly stereoselective hydrogen transfer to the carbon-carbon double bond of the 2-benzylidene substrate. The urea-type pyrrolidine diphosphine ligand coordinates with the rhodium center to form a chiral environment that strongly favors the formation of the (2S)-enantiomer. The structural rigidity and electronic properties imparted by the urea linkage at the nitrogen atom of the pyrrolidine ring appear to enhance the stability of the active catalytic species and improve the discrimination between the prochiral faces of the olefinic substrate. This enhanced interaction ensures that the hydrogenation proceeds with exceptional enantioselectivity, minimizing the formation of the unwanted (2R)-isomer which would otherwise act as a difficult-to-remove impurity. The versatility of the ligand structure allows for further tuning through substituents on the phosphine aryl groups or the urea nitrogen, enabling chemists to optimize the catalyst for specific batch requirements or solvent systems without compromising the fundamental stereochemical outcome.
Furthermore, the mechanism benefits from the specific choice of rhodium precursors, such as rhodium-1,5-cyclooctadiene-chloro complex, which readily forms the active hydride species under mild hydrogen pressures. The patent data highlights that the reaction maintains high efficiency across a broad range of conditions, including temperatures from 10°C to 50°C and hydrogen pressures as low as 0.5 MPa. This operational flexibility is critical for process safety and equipment longevity in a commercial plant setting. The robustness of the catalyst system means that variations in feedstock quality or minor fluctuations in process parameters do not lead to catastrophic drops in yield or selectivity. By understanding these mechanistic nuances, R&D teams can confidently implement this chemistry knowing that the catalyst system is engineered to withstand the rigors of industrial production while delivering the stringent purity profiles demanded by regulatory agencies for diabetes medications.
How to Synthesize (2S)-2-benzyl-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid Efficiently
Implementing this advanced asymmetric reduction route requires careful attention to catalyst preparation and reaction monitoring to fully leverage the kinetic advantages described in the patent. The process begins with the in situ generation of the active rhodium complex by mixing the urea-type ligand with the rhodium precursor in an alcoholic solvent, typically methanol or ethanol. This mixture is then introduced to the substrate, 2-benzylidene-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid, under an inert atmosphere before pressurizing with hydrogen. The detailed standardized synthesis steps see the guide below.
- Prepare the asymmetric catalyst by mixing a urea-type pyrrolidine diphosphine compound (Formula I) with a rhodium complex such as rhodium-1,5-cyclooctadiene-chloro complex in a solvent like methanol.
- Charge the reactor with the substrate 2-benzylidene-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid and the prepared catalyst solution, ensuring a substrate-to-catalyst molar ratio (S/C) between 5000 and 20000.
- Conduct the hydrogenation reaction under hydrogen pressure of 0.1 to 2 MPa at a temperature range of 10 to 50°C for approximately 4 to 10 hours until conversion exceeds 97%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this urea-ligand based asymmetric reduction technology offers transformative benefits that extend far beyond simple chemical yield improvements. The primary value driver is the drastic reduction in catalyst consumption enabled by the high S/C ratios, which directly lowers the bill of materials for every kilogram of intermediate produced. Since rhodium is a volatile and expensive commodity, minimizing its usage per batch provides a substantial buffer against market price fluctuations and significantly improves the overall gross margin of the manufacturing process. Moreover, the accelerated reaction kinetics mean that reactor turnover times are shortened, allowing existing production assets to generate more output over the same period without requiring capital expenditure on new equipment. This increase in throughput capability enhances supply security, ensuring that downstream formulation partners receive their API intermediates on schedule even during periods of high demand.
- Cost Reduction in Manufacturing: The ability to operate with substrate-to-catalyst ratios as high as 20000 fundamentally changes the cost structure of the synthesis. By eliminating the need for excessive amounts of noble metal catalysts, manufacturers can achieve significant cost savings on raw materials. Additionally, the high conversion rates reduce the burden on purification units, lowering solvent consumption and energy costs associated with distillation and crystallization steps. This leaner operational model ensures that the final cost of goods sold is optimized, making the end product more competitive in the global generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the urea-type catalyst system contributes to a more predictable and reliable supply chain. Because the reaction is less sensitive to minor deviations and completes in a short timeframe, the risk of batch failures or off-spec production is minimized. This consistency allows supply chain planners to maintain tighter inventory controls and reduce safety stock levels, freeing up working capital. Furthermore, the use of common solvents like methanol and mild reaction conditions simplifies logistics and storage requirements, reducing the complexity of managing hazardous materials across the supply network.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern green chemistry principles. The reduction in heavy metal usage decreases the toxic load of the waste stream, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The high atom economy and selectivity mean less waste is generated per unit of product, supporting sustainability goals. Scalability is further assured by the fact that the reaction performs well at high concentrations, meaning that scaling from pilot plant to commercial multi-ton production does not require disproportionate increases in solvent volume or reactor size, facilitating a smooth technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reduction technology for Mitiglinide production. These insights are derived directly from the experimental data and claims within the patent literature to provide clarity on process capabilities.
Q: What is the primary advantage of using urea-type ligands over carbamate-type ligands in this synthesis?
A: Urea-type pyrrolidine diphosphine ligands, such as PCPPM, demonstrate significantly superior catalytic activity compared to traditional carbamate-type ligands like BPPM. This results in much faster reaction rates, allowing for completion in roughly 4 hours versus extended periods, and enables the use of drastically lower catalyst loadings (higher S/C ratios) while maintaining high optical purity above 95% e.e.
Q: Can this asymmetric reduction process be scaled for industrial production?
A: Yes, the process is specifically designed for industrial applicability. The patent data indicates that the reaction proceeds efficiently even at high substrate concentrations and achieves nearly 100% conversion, which simplifies downstream purification and makes it highly suitable for large-scale commercial manufacturing of diabetes therapeutics.
Q: What represent the optimal reaction conditions for maximizing yield and purity?
A: Optimal conditions involve using methanol or ethanol as the solvent, maintaining a hydrogen pressure between 0.5 and 1 MPa, and keeping the reaction temperature between 10°C and 50°C. Under these conditions, using ligands like PCPPM or DCPCPPM with rhodium complexes yields the target acid with optical purity exceeding 95% e.e. and minimal residual starting material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mitiglinide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of diabetes therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the urea-type rhodium system described in CN101346352B, we can offer our partners a supply solution that balances cost, quality, and speed.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you secure a stable and economical supply of high-quality Mitiglinide intermediates for your global operations.
