Advanced Chiral Phosphine Ligands for High-Purity Pharmaceutical Intermediate Manufacturing
The pharmaceutical industry continuously demands more efficient and selective methods for constructing chiral centers, which are critical for the biological activity of modern drugs. Patent CN103087105A introduces a groundbreaking class of chiral phosphine ligands, specifically designed to overcome the limitations of traditional asymmetric catalysis. These novel ligands, characterized by a unique dibenzo[d][1,3]oxaphosphole backbone substituted with bulky aryl groups, enable the highly enantioselective hydrogenation of beta-aryl enamides. This technological advancement is particularly significant for the synthesis of key pharmaceutical intermediates found in bioactive molecules such as antimalarials, anti-HIV agents, and alpha-adrenoceptor blockers. The core innovation lies in the structural rigidity and steric environment provided by the ligand, which translates directly into superior catalytic performance.
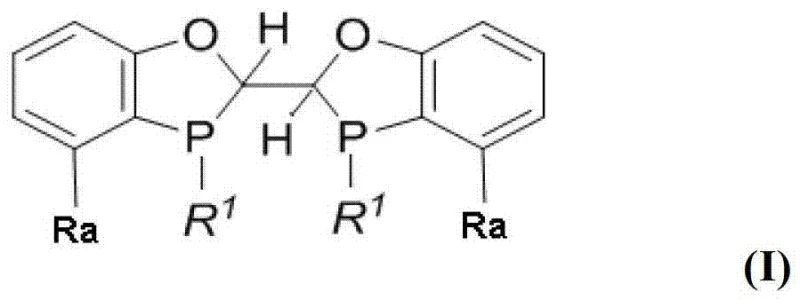
For research and development directors focusing on process optimization, the structural versatility of these ligands offers a powerful tool for fine-tuning reaction outcomes. The general formula allows for independent variation of the substituents on the phosphorus atom (R1) and the aromatic rings (Ra), enabling chemists to modulate the electronic and steric properties of the catalyst. This adaptability ensures that the catalyst can be optimized for specific substrate classes, ranging from acyclic E-enamides to complex cyclic and heterocyclic systems. By leveraging this tunable architecture, manufacturers can achieve consistent high-quality outputs, minimizing the need for downstream purification steps that often erode profit margins in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral beta-aryl amides has relied heavily on resolution of racemic mixtures or the use of stoichiometric chiral auxiliaries. Resolution methods are inherently inefficient, theoretically discarding 50% of the starting material, which creates significant waste disposal challenges and inflates raw material costs. Furthermore, processes utilizing chiral auxiliaries require additional synthetic steps for attachment and removal, increasing the overall process mass intensity and extending production timelines. Even existing asymmetric hydrogenation methods often suffer from limited substrate scope or require prohibitively expensive catalyst loadings to achieve acceptable enantioselectivity. These inefficiencies create bottlenecks in the supply chain, making it difficult to secure reliable supplies of high-purity intermediates at a competitive cost.
The Novel Approach
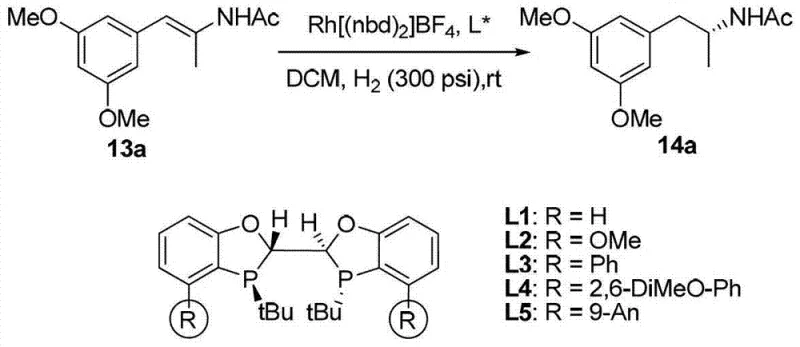
The technology disclosed in CN103087105A represents a paradigm shift by utilizing a catalytic amount of a highly active chiral rhodium complex. Unlike conventional methods, this approach does not consume chiral sources stoichiometrically; instead, a single catalyst molecule can turnover thousands of times. The novel ligands feature bulky anthracenyl or naphthyl groups that create a well-defined chiral pocket around the metal center. This precise spatial arrangement directs the hydrogenation to occur exclusively on one face of the double bond, resulting in products with exceptional optical purity. Comparative studies within the patent data indicate that ligands with larger aromatic substituents significantly outperform simpler analogues, demonstrating that steric bulk is a critical design element for maximizing enantioselectivity in this reaction class.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The efficacy of this catalytic system stems from the intricate interplay between the ligand architecture and the transition metal center. Upon coordination with a rhodium precursor, such as Rh(nbd)2BF4, the bidentate phosphine ligand forms a stable cationic complex. The rigid backbone prevents unfavorable conformational changes during the catalytic cycle, ensuring that the chiral information is effectively transferred to the substrate. The mechanism likely proceeds through a standard dihydride pathway where the beta-aryl enamide coordinates to the rhodium center. The bulky R1 and Ra groups shield specific quadrants of the metal, forcing the substrate to bind in a specific orientation that favors the formation of one enantiomer over the other. This high degree of stereocontrol is maintained even at elevated temperatures and pressures, highlighting the robustness of the catalyst-ligand interaction.
Furthermore, the stability of the catalyst contributes to its impressive turnover numbers. The patent data reports substrate-to-catalyst ratios (s/c) reaching up to 10,000 without significant loss of activity or selectivity. This suggests that the catalyst is resistant to deactivation pathways such as aggregation or oxidation, which commonly plague less robust systems. For process chemists, this means that residual metal levels in the final product can be kept low, simplifying the purification process and ensuring compliance with strict regulatory limits for heavy metals in pharmaceutical products. The ability to operate efficiently at low catalyst loadings is a direct result of the ligand's ability to stabilize the active rhodium species throughout the reaction duration.
How to Synthesize Chiral Bisphosphine Ligands Efficiently
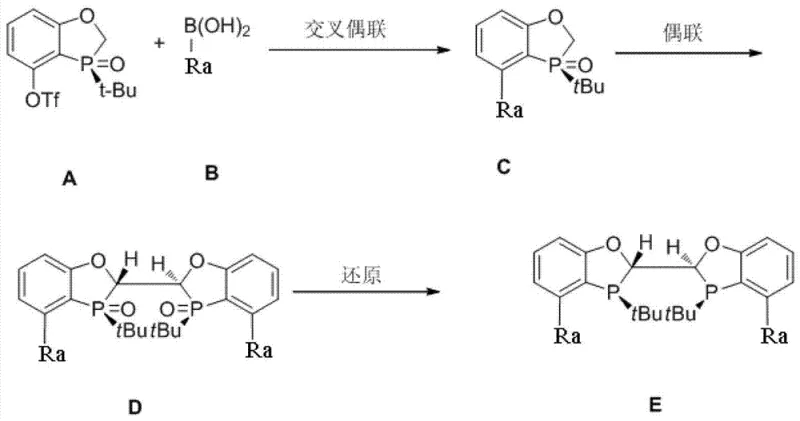
The preparation of these high-performance ligands follows a logical and scalable synthetic sequence that begins with readily available starting materials. The process initiates with a palladium-catalyzed cross-coupling reaction to install the requisite aryl groups onto the phospholene oxide core. This is followed by a critical oxidative dimerization step that links two monomeric units to form the bisphosphine oxide backbone. Finally, a reduction step converts the oxide into the active trivalent phosphine ligand. Each step has been optimized to ensure high yields and purity, making the overall route suitable for kilogram-scale production. The detailed standardized synthesis steps for implementing this route are provided in the guide below.
- Perform a palladium-catalyzed cross-coupling reaction between a trifluoromethanesulfonate precursor and a specific aryl boronic acid to install the bulky aromatic substituent.
- Execute an oxidative dimerization using a strong base like LDA and a metal oxidant such as copper(II) chloride to form the bisphosphine oxide backbone.
- Conduct a final reduction step using silane-based reducing agents to convert the phosphine oxide into the active trivalent phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers substantial strategic benefits beyond mere technical performance. The primary advantage lies in the drastic reduction of raw material consumption. By eliminating the 50% yield loss associated with racemic resolution, manufacturers can effectively double the output from the same amount of starting feedstock. This efficiency gain translates directly into lower unit costs and reduced dependency on volatile raw material markets. Additionally, the high turnover number of the catalyst means that the consumption of expensive precious metals like rhodium is minimized, further driving down the cost of goods sold. These factors combine to create a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the use of catalytic rather than stoichiometric chiral sources significantly lowers the overall production cost. The high efficiency of the catalyst reduces the requirement for expensive transition metals, while the simplified workflow decreases labor and utility expenses associated with extra synthetic steps. This streamlined process allows for a more competitive pricing structure without compromising on the quality or purity of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The synthetic route relies on commercially available building blocks such as boronic acids and triflates, which are sourced from a stable global supply network. This reduces the risk of supply disruptions caused by reliance on exotic or custom-synthesized reagents. Furthermore, the robustness of the catalyst ensures consistent batch-to-batch reproducibility, allowing supply chain planners to forecast production timelines with greater accuracy and confidence, thereby reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions are mild, typically operating at moderate temperatures and hydrogen pressures, which facilitates safe scale-up from pilot plant to commercial production. The high atom economy of the asymmetric hydrogenation process generates minimal waste compared to traditional resolution methods, aligning with green chemistry principles. This reduced environmental footprint simplifies waste management protocols and helps manufacturers meet increasingly stringent regulatory standards for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What level of enantioselectivity can be achieved with these ligands?
A: The patented technology demonstrates the capability to achieve enantiomeric excess (ee) values greater than 99% for a wide range of beta-aryl enamide substrates, ensuring high optical purity for pharmaceutical applications.
Q: Which transition metals are compatible with this ligand system?
A: While Rhodium (Rh) is the preferred metal for optimal performance, the ligand structure is versatile and can form active complexes with other transition metals including Ruthenium (Ru), Iridium (Ir), and Palladium (Pd).
Q: Is this catalytic system suitable for large-scale industrial production?
A: Yes, the system supports extremely high substrate-to-catalyst ratios (s/c) of up to 10,000, which significantly reduces the cost of goods and makes it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug development and commercialization. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral ligand or intermediate meets the highest international standards. Our commitment to quality ensures that your downstream processes remain uninterrupted and compliant with global regulatory requirements.
We invite you to collaborate with us to optimize your synthesis routes and achieve significant operational efficiencies. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your supply chain stability and reduce your overall manufacturing costs. Let us be your partner in delivering high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
