Advanced Iodine-Promoted Cyclization for High-Purity 3-Trifluoromethyl-1,2,4-Triazoles
Introduction to Next-Generation Triazole Synthesis
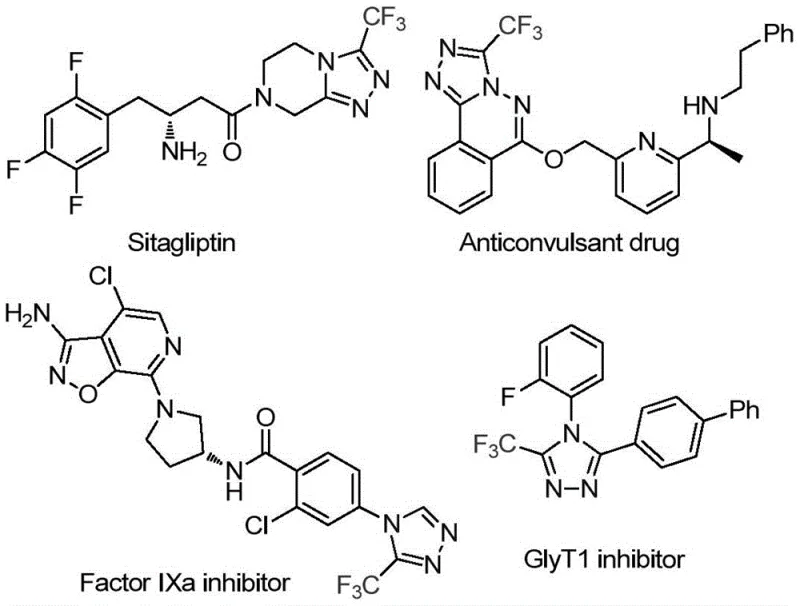
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-rich heterocycles, particularly those bearing fluorinated motifs which are pivotal for enhancing metabolic stability and bioavailability. A groundbreaking advancement in this domain is detailed in Chinese Patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift by leveraging the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source in an iodine-promoted tandem cyclization. The strategic integration of DMF as a C1 synthon allows for the direct assembly of the triazole core from readily available trifluoroethyliminohydrazide precursors. This approach addresses critical pain points in modern process chemistry, specifically the need for atom economy and operational simplicity. By eliminating the requirement for specialized anhydrous or anaerobic conditions, this method offers a compelling value proposition for manufacturers aiming to streamline their production of high-value intermediates used in blockbuster drugs such as Factor IXa inhibitors and GlyT1 inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant logistical and economic challenges that hinder large-scale commercialization. Conventional routes often rely on multi-step sequences involving harsh reaction conditions, such as extreme temperatures or the use of hazardous, moisture-sensitive reagents that demand rigorous exclusion of air and water. These requirements necessitate specialized reactor setups, inert gas purging systems, and extensive safety protocols, all of which inflate capital expenditure (CapEx) and operational expenditure (OpEx). Furthermore, traditional methods frequently utilize stoichiometric amounts of expensive coupling agents or distinct carbon sources that add to the overall material cost and generate substantial chemical waste. The purification of intermediates in these legacy processes can also be cumbersome, often requiring multiple recrystallizations or chromatographic steps to remove metal catalysts or toxic byproducts, thereby reducing the overall throughput and yield of the final active pharmaceutical ingredient (API) intermediate.
The Novel Approach
In stark contrast, the methodology described in patent CN114920707B introduces a streamlined, one-pot strategy that fundamentally simplifies the synthetic landscape. By utilizing molecular iodine as a promoter and DMF as a dual-purpose solvent and reactant, this novel approach bypasses the need for external carbon sources or complex catalytic systems. The reaction proceeds smoothly under an air atmosphere at moderate temperatures ranging from 110°C to 130°C, effectively removing the burden of maintaining strict inert conditions. This operational flexibility translates directly into reduced downtime between batches and lower energy consumption for environmental control systems. Moreover, the broad substrate scope demonstrated in the patent indicates that various functional groups, including halogens, alkyls, and alkoxy groups, are well-tolerated, allowing for the rapid generation of diverse libraries of triazole derivatives without the need for protecting group strategies. This robustness makes the process exceptionally suitable for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
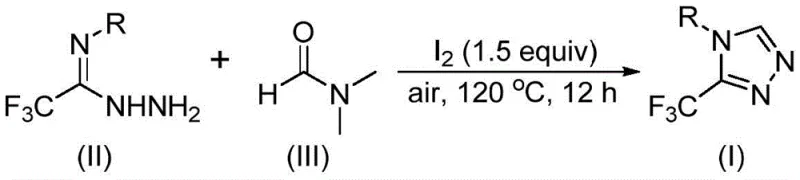
The elegance of this synthesis lies in its mechanistic versatility, where DMF acts as a dynamic carbon donor through two potential pathways, ensuring high reaction efficiency regardless of subtle electronic variations in the substrate. In the first pathway, the formyl group of DMF serves as the carbon source, undergoing a condensation reaction with the trifluoroethyliminohydrazide to generate a hydrazone intermediate. This is followed by an intramolecular cyclization that eliminates a molecule of dimethylamine, directly yielding the desired 3-trifluoromethyl-substituted 1,2,4-triazole product. Alternatively, the N-methyl group of DMF can participate as the carbon synthon. In this scenario, molecular iodine activates the DMF to generate an amine salt, which subsequently eliminates hydrogen iodide. This activated species then undergoes nucleophilic addition with the hydrazide, followed by the elimination of N-methylformamide to form a reactive azadiene intermediate. The final step involves an intramolecular nucleophilic addition coupled with oxidative aromatization to furnish the target heterocycle. This dual-pathway capability ensures that the reaction remains robust even if one pathway is kinetically disfavored by specific substituents on the aromatic ring.
From an impurity control perspective, the use of molecular iodine is particularly advantageous as it facilitates clean oxidative aromatization without leaving behind heavy metal residues that are notoriously difficult to purge from final drug substances. The mechanism inherently drives the reaction towards the thermodynamically stable aromatic triazole system, minimizing the formation of partially cyclized byproducts or oligomers. The patent data indicates that side reactions are minimal, with the primary byproduct being dimethylamine or N-methylformamide, both of which are volatile or easily removed during standard aqueous workups. This clean reaction profile significantly reduces the burden on downstream purification teams, allowing for simpler isolation protocols such as filtration and basic column chromatography. For R&D directors, understanding this mechanism confirms that the process is not only chemically sound but also designed with purity and scalability in mind, mitigating risks associated with genotoxic impurities or residual metals.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The execution of this synthesis is remarkably straightforward, designed to be implemented in standard multipurpose reactors without the need for custom engineering. The process begins by charging the reactor with the trifluoroethyliminohydrazide starting material, molecular iodine, and DMF. The simplicity of the reagent list means that procurement teams can source materials globally with minimal lead time. The reaction is heated to the specified range of 110-130°C and maintained for 10 to 15 hours under ambient air pressure. Upon completion, the workup involves standard filtration to remove any insoluble salts, followed by silica gel treatment and column chromatography to achieve high-purity standards required for pharmaceutical applications.
- Charge a reaction vessel with molecular iodine (1.5 equivalents), trifluoroethyliminohydrazide substrate, and DMF solvent.
- Heat the mixture to 110-130°C under an air atmosphere and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, silica gel mixing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements. The most significant advantage is the drastic reduction in raw material complexity and cost. By utilizing DMF—a commodity chemical produced on a massive global scale—as both the solvent and the carbon building block, the process eliminates the need for purchasing specialized, high-cost C1 reagents. This consolidation of materials simplifies inventory management and reduces the risk of supply chain disruptions caused by the scarcity of niche reagents. Furthermore, the ability to run the reaction under air atmosphere removes the dependency on bulk nitrogen or argon supplies, further lowering utility costs and infrastructure requirements. The operational simplicity also意味着 that the process can be easily transferred between different manufacturing sites or scaled up from pilot plants to commercial production lines with minimal re-validation efforts.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of expensive catalysts and the dual-use nature of the solvent. Traditional methods often require precious metal catalysts or stoichiometric oxidants that contribute significantly to the bill of materials. By replacing these with inexpensive molecular iodine and DMF, the variable cost per kilogram of the intermediate is substantially decreased. Additionally, the simplified workup procedure reduces the consumption of purification media such as silica gel and solvents, leading to lower waste disposal costs. The overall process efficiency means that less raw material is wasted on failed batches or low-yield steps, maximizing the return on investment for every ton of starting material purchased.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical in the post-pandemic era, and this synthesis method bolsters reliability by relying on universally available commodities. DMF and iodine are produced by numerous suppliers worldwide, ensuring that no single point of failure exists in the supply network. The robustness of the reaction conditions—tolerating air and moisture—means that transportation and storage of reagents do not require specialized cold chains or desiccants, reducing logistics costs and the risk of material degradation during transit. This flexibility allows manufacturers to maintain leaner inventories while still guaranteeing continuous production, a key metric for supply chain heads managing just-in-time delivery schedules for global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this method is inherently designed for green chemistry principles. The absence of heavy metals and the use of a common solvent simplify the effluent treatment process, making it easier to meet stringent environmental regulations in major manufacturing hubs. The reaction generates minimal hazardous waste, and the byproducts are relatively benign, facilitating easier permitting for capacity expansion. For operations teams, this means that increasing production volume from hundreds of kilograms to multi-ton scales does not require proportional increases in waste treatment infrastructure, allowing for linear cost scaling and faster time-to-market for new drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity for technical decision-makers evaluating this route for their pipeline.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual function as both the reaction solvent and the critical C1 carbon source, eliminating the need for separate, expensive formylating agents and significantly simplifying the raw material supply chain.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, the patented method operates efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, drastically reducing operational complexity and equipment costs.
Q: What is the typical reaction temperature and time frame?
A: The reaction is typically conducted at elevated temperatures between 110°C and 130°C for a duration of 10 to 15 hours to achieve optimal yields and conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to industrial manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest global regulatory standards. We understand the critical nature of fluorinated intermediates in modern drug design and are committed to delivering consistent quality and supply continuity.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume, demonstrating exactly how this DMF-based route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis requirements. Let us help you accelerate your development timeline with reliable, cost-effective, and high-quality chemical solutions.
