Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. Quinazolinones represent a privileged scaffold in medicinal chemistry, widely recognized for their potent biological activities ranging from anti-cancer and anticonvulsant to anti-inflammatory and antifungal properties. The strategic introduction of a trifluoromethyl group into these heterocyclic systems further enhances their pharmacokinetic profiles by improving metabolic stability, lipophilicity, and bioavailability. This patent presents a transformative approach that leverages inexpensive iron catalysis to construct these valuable motifs, addressing critical pain points in traditional synthesis regarding cost and operational complexity.
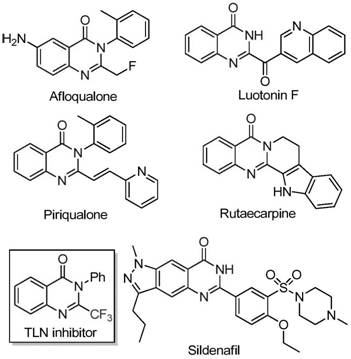
Historically, the construction of trifluoromethyl-substituted quinazolinones has relied heavily on cyclization reactions involving synthons like trifluoroacetic anhydride or ethyl trifluoroacetate with substrates such as anthranilamide or isatoic anhydride. While effective in academic settings, these conventional methodologies often suffer from severe limitations when translated to commercial production. They frequently require harsh reaction conditions, utilize expensive and moisture-sensitive reagents, and exhibit narrow substrate scopes that limit the diversity of accessible analogs. Moreover, the reliance on stoichiometric amounts of activating agents often generates substantial chemical waste, complicating downstream purification and increasing the environmental footprint of the manufacturing process. For procurement managers and supply chain heads, these factors translate into volatile pricing, extended lead times, and significant challenges in securing reliable supplies of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The traditional synthetic pathways for accessing 2-trifluoromethyl quinazolinones are fraught with inefficiencies that hinder their scalability. Primary among these is the dependency on costly fluorinating agents and activated carboxylic acid derivatives, which not only drive up the Bill of Materials (BOM) but also introduce safety hazards associated with their handling and storage. The reaction conditions often necessitate strict anhydrous environments and cryogenic temperatures, demanding specialized equipment and energy-intensive processes that are difficult to maintain on a multi-kilogram scale. Furthermore, the functional group tolerance in these legacy methods is often poor, leading to side reactions and the formation of difficult-to-remove impurities that compromise the overall yield and purity of the final API intermediate. These technical bottlenecks create a fragile supply chain where minor deviations in raw material quality can result in batch failures, posing a significant risk to project timelines.
The Novel Approach
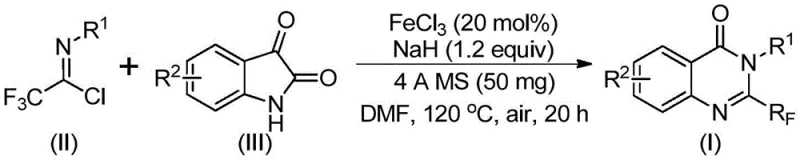
In stark contrast, the methodology outlined in CN111675662B offers a streamlined and robust alternative by utilizing readily available trifluoroethylimidoyl chloride and isatin as starting materials. This innovative route employs ferric chloride (FeCl3) as a cheap and earth-abundant catalyst, coupled with sodium hydride as a base, to facilitate the cyclization under relatively mild conditions. The process operates effectively in common polar aprotic solvents like DMF and tolerates a wide array of functional groups, including halogens, alkyls, and electron-withdrawing nitro groups. By shifting from precious metals or harsh activators to a simple iron salt system, this approach drastically simplifies the operational protocol. The reaction proceeds through a tandem sequence involving carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization, delivering the target quinazolinone scaffolds with high efficiency and minimal byproduct formation.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the dual role of the reaction components in facilitating bond construction and rearrangement. Initially, the base-promoted nucleophilic attack of the isatin nitrogen on the imidoyl chloride generates a key trifluoroacetamidine intermediate. This step is crucial for establishing the carbon-nitrogen framework required for the subsequent ring closure. Following this, the ferric chloride catalyst activates the carbonyl moiety, promoting a decarbonylation event that is essential for aromatization and the formation of the quinazolinone core. This iron-catalyzed isomerization is highly selective, minimizing the formation of regioisomers or polymeric byproducts that often plague similar cyclization reactions. The presence of 4A molecular sieves in the reaction mixture plays a pivotal role in scavenging trace moisture, thereby protecting the sensitive imidoyl chloride and maintaining the activity of the sodium hydride base throughout the prolonged heating period.
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on quality. The mild nature of the iron catalysis prevents the degradation of sensitive functional groups on the aromatic rings, which is a common issue with stronger Lewis acids or high-temperature thermal cyclizations. The reaction conditions—specifically the two-stage heating profile starting at 40°C and ramping to 120°C—allow for controlled kinetics, ensuring complete conversion of the starting materials while suppressing thermal decomposition pathways. Post-reaction workup is straightforward, typically involving filtration to remove the molecular sieves and inorganic salts, followed by standard silica gel column chromatography. This simplicity in purification ensures that the final product meets stringent purity specifications required for pharmaceutical development, with residual metal levels easily managed due to the low loading of the iron catalyst.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the catalyst, base, and drying agent, followed by the addition of the substrates in an organic solvent. The mixture is stirred at a moderate temperature to initiate the coupling, then heated to reflux to drive the cyclization to completion. This standardized procedure minimizes the need for specialized operator training and allows for consistent batch-to-batch reproducibility. For detailed operational parameters, stoichiometry, and specific workup instructions tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- Combine ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF.
- Stir the mixture at 40°C for 8-10 hours to initiate the reaction, then increase the temperature to 120°C.
- Maintain heating at 120°C for 18-20 hours under air, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and supply continuity, this patented process offers compelling economic and logistical benefits. The substitution of expensive reagents with commodity chemicals like isatin and ferric chloride directly impacts the cost structure of the intermediate, making it highly competitive for generic drug manufacturing and large-scale API production. The robustness of the reaction conditions reduces the risk of batch failures, ensuring a steady flow of material to downstream processes. Additionally, the use of non-proprietary, off-the-shelf starting materials mitigates supply chain risks associated with single-source vendors, enhancing the overall resilience of the procurement strategy.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and expensive fluorinating agents results in a drastic reduction in raw material costs. Unlike traditional methods that may require palladium or rhodium complexes, the use of ferric chloride—a bulk industrial chemical—lowers the catalyst cost by orders of magnitude. Furthermore, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials, specifically isatin and various substituted aromatic amines used to prepare the imidoyl chloride, are widely available from multiple global suppliers. This commoditization of inputs ensures that production is not held hostage by the availability of niche reagents. The stability of the reagents also allows for longer shelf-life and easier logistics, reducing the need for cold-chain shipping and specialized storage facilities, which streamlines the supply chain for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction is amenable to scale-up from gram to multi-kilogram quantities without significant re-optimization, facilitating the commercial scale-up of complex pharmaceutical intermediates. The use of iron, a non-toxic metal, aligns with green chemistry principles and simplifies regulatory compliance regarding heavy metal residues in the final drug substance. This environmental compatibility reduces the burden on waste treatment facilities and supports sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts?
A: The use of ferric chloride (FeCl3) significantly reduces raw material costs compared to palladium or rhodium catalysts. Furthermore, it eliminates the need for complex heavy metal removal steps during downstream processing, simplifying purification and ensuring higher purity profiles for pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and provides possibilities for industrial scale application. The use of inexpensive, commercially available starting materials like isatin and the robustness of the iron catalyst support large-scale manufacturing.
Q: What is the functional group tolerance of this reaction?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents on the aryl ring including alkyl, halogen (fluorine, chlorine, bromine, iodine), methoxy, and nitro groups. This versatility allows for the synthesis of a diverse library of quinazolinone derivatives for SAR studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN111675662B and is fully equipped to leverage this iron-catalyzed methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our state-of-the-art facilities are designed to handle sensitive organometallic reactions with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality standards for every batch produced.
We invite you to collaborate with us to optimize your supply chain for 2-trifluoromethyl quinazolinone derivatives. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel route for your specific portfolio. We encourage you to contact us today to request specific COA data for our reference standards and to discuss route feasibility assessments tailored to your unique molecular targets, ensuring a reliable partnership for your long-term manufacturing needs.
