Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic methodologies to access privileged scaffolds that exhibit potent biological activities. Patent CN112125856A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, a class of compounds renowned for their extensive therapeutic potential ranging from anticancer to antiviral applications. The introduction of the trifluoromethyl group is strategically critical as it significantly enhances metabolic stability and lipophilicity, yet traditional methods often suffer from severe limitations regarding safety and efficiency. This novel approach leverages a transition metal palladium-catalyzed carbonylative tandem reaction, utilizing a solid carbon monoxide substitute to bypass the inherent dangers of toxic gas handling. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this technology represents a paradigm shift towards safer, more sustainable, and economically viable manufacturing processes that align with modern green chemistry principles.
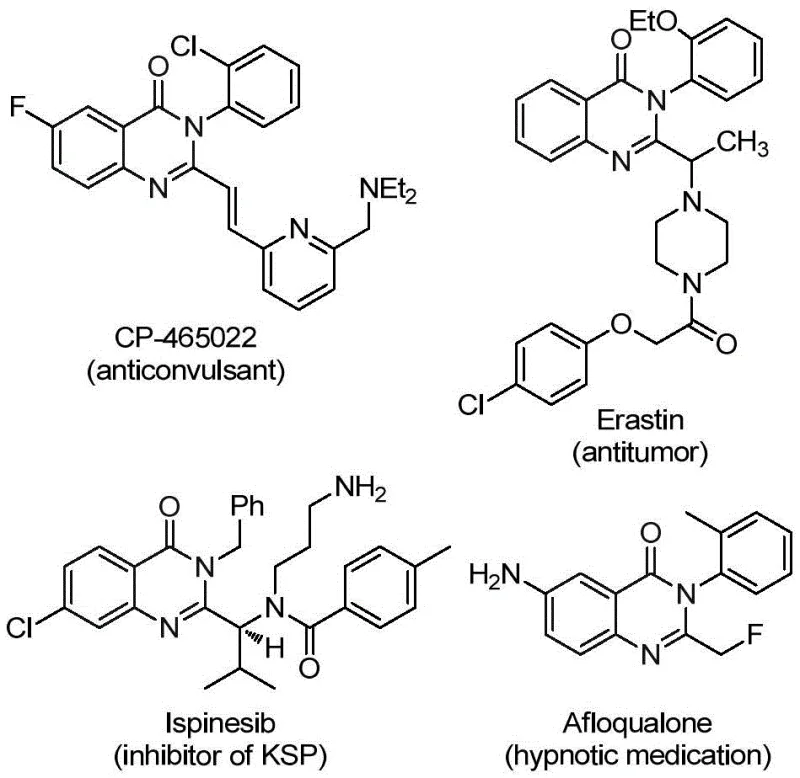
Historically, the construction of the quinazolinone core, particularly with specific substitution patterns at the 2-position, has relied on methodologies that impose significant burdens on both laboratory safety and production costs. Conventional routes frequently necessitate the use of unstable reagents such as trifluoroacetamides or require the direct handling of carbon monoxide gas, which demands specialized high-pressure equipment and rigorous safety protocols to prevent leakage and exposure. Furthermore, existing methods often employ expensive coupling agents like T3P or require pre-activation of substrates, leading to increased raw material costs and complex waste streams that complicate downstream processing. These traditional approaches typically exhibit narrow substrate scopes, failing to accommodate diverse functional groups without significant yield penalties, thereby restricting the chemical space available for medicinal chemistry optimization and limiting the flexibility required for rapid analog synthesis in drug discovery programs.
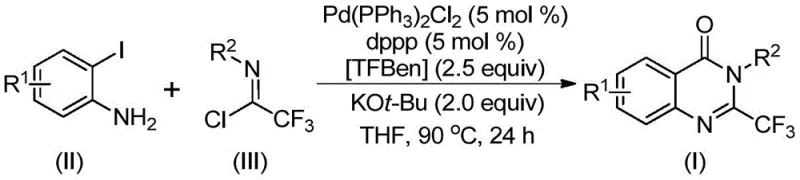
In stark contrast, the novel approach detailed in the patent utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a safe and efficient solid carbon monoxide surrogate, effectively eliminating the need for hazardous gas cylinders and flow reactors. This method employs readily available starting materials, specifically trifluoroethylimidoyl chloride and o-iodoaniline, which are commercially accessible and cost-effective compared to specialized precursors required by older techniques. The reaction proceeds under relatively mild thermal conditions at 90°C, avoiding the extreme temperatures or pressures that often degrade sensitive functional groups or lead to decomposition of the product. By streamlining the synthetic sequence into a one-pot tandem reaction, this methodology drastically simplifies the operational workflow, reduces the number of unit operations, and minimizes solvent consumption, thereby offering substantial cost reduction in pharmaceutical intermediates manufacturing while maintaining high levels of chemical efficiency and product integrity.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps orchestrated by the palladium catalyst system to construct the heterocyclic framework with high precision. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride to generate a trifluoroacetamidine intermediate in situ. Subsequently, the palladium catalyst undergoes oxidative insertion into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium species that is poised for carbonyl insertion. Upon heating, the solid TFBen decomposes to release carbon monoxide directly within the reaction medium, which then inserts into the carbon-palladium bond to generate an acyl-palladium intermediate. This acyl species subsequently undergoes intramolecular nucleophilic attack by the nitrogen atom, facilitated by the base, to form a seven-membered palladacycle before final reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active catalyst.
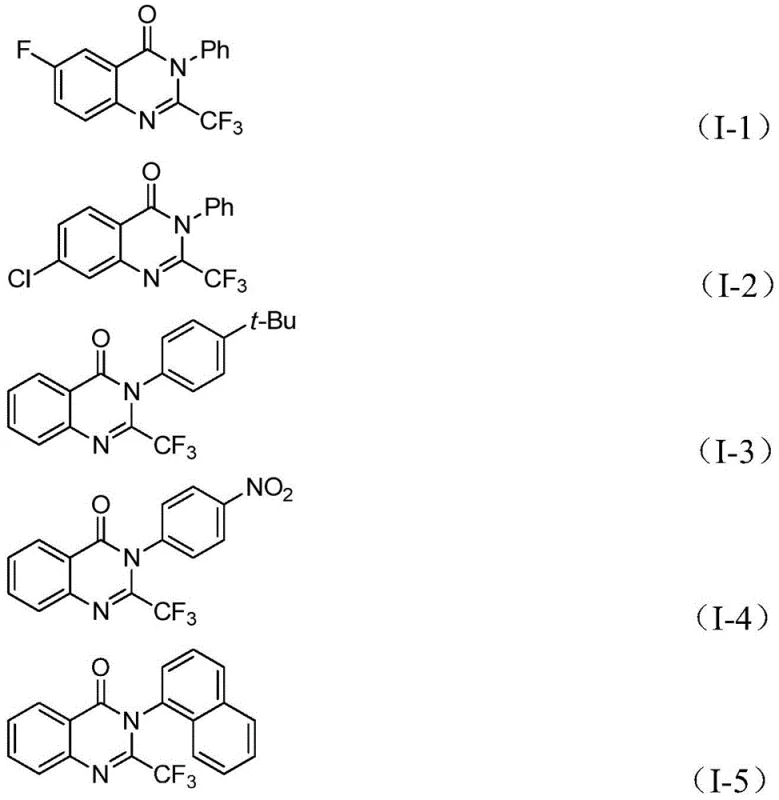
Controlling the impurity profile is paramount for ensuring the quality of high-purity quinazolinone derivatives intended for pharmaceutical applications, and this catalytic system offers inherent advantages in selectivity. The use of specific ligands such as 1,3-bis(diphenylphosphine)propane (dppp) in conjunction with the palladium source helps to stabilize the active catalytic species and suppresses competing side reactions such as homocoupling of the aryl iodide or hydrodehalogenation. The mild reaction conditions prevent the thermal degradation of the trifluoromethyl group, which can be susceptible to defluorination under harsher acidic or basic environments found in classical cyclization methods. Furthermore, the compatibility of the system with various substituents on the aryl ring, including electron-withdrawing and electron-donating groups, ensures that the electronic nature of the substrate does not lead to the formation of complex byproduct mixtures. This high level of chemoselectivity simplifies the purification process, allowing for the isolation of the target molecule with stringent purity specifications through standard chromatographic techniques without the need for extensive recrystallization or specialized separation technologies.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The implementation of this synthetic route requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility across different scales. The process begins with the precise weighing of the palladium catalyst, ligand, base, and the solid carbon monoxide source, which are then combined with the substrates in an aprotic organic solvent such as tetrahydrofuran to ensure homogeneous dissolution. Maintaining the reaction temperature at 90°C for a duration of 16 to 30 hours is critical to drive the carbonylative tandem sequence to completion while allowing sufficient time for the slow release of carbon monoxide from the solid surrogate. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining palladium catalyst, ligand, base, solid CO source, trifluoroethylimidoyl chloride, and o-iodoaniline in an organic solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion via carbonylative tandem reaction.
- Perform post-treatment including filtration and silica gel mixing, followed by column chromatography purification to isolate the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology addresses several critical pain points that traditionally plague the supply chain for complex heterocyclic intermediates, offering tangible benefits for procurement managers focused on cost and continuity. The elimination of toxic carbon monoxide gas from the process removes the need for specialized gas handling infrastructure and safety monitoring systems, which translates directly into lower capital expenditure and reduced operational overhead for manufacturing facilities. Additionally, the reliance on cheap and easily obtainable starting materials mitigates the risk of supply disruptions caused by the scarcity of exotic reagents, ensuring a more resilient supply chain capable of meeting fluctuating demand without significant price volatility. The simplified workup procedure, involving basic filtration and chromatography, reduces the consumption of processing time and utilities, further enhancing the overall economic efficiency of the production campaign.
- Cost Reduction in Manufacturing: The replacement of hazardous gaseous reagents with a stable solid surrogate significantly lowers the safety compliance costs and insurance premiums associated with chemical manufacturing. By avoiding the use of expensive coupling agents and pre-activated substrates, the raw material bill of materials is optimized, leading to substantial cost savings per kilogram of produced intermediate. The high conversion rates observed with this method minimize the loss of valuable starting materials, ensuring that the input costs are efficiently translated into saleable product output without significant waste generation.
- Enhanced Supply Chain Reliability: Utilizing commercially available o-iodoanilines and trifluoroethylimidoyl chlorides ensures that the raw material base is broad and diversified, reducing dependency on single-source suppliers for niche chemicals. The robustness of the reaction conditions allows for flexible scheduling and batch planning, as the process is less sensitive to minor variations in environmental factors compared to gas-phase reactions. This stability supports reducing lead time for high-purity quinazolinone derivatives, enabling faster response to market demands and tighter integration with downstream drug substance manufacturing timelines.
- Scalability and Environmental Compliance: The absence of toxic gas emissions aligns perfectly with increasingly stringent environmental regulations, facilitating easier permitting and regulatory approval for production sites. The use of common organic solvents and standard purification techniques means that the process can be seamlessly transferred from laboratory scale to commercial scale-up of complex pharmaceutical intermediates without requiring bespoke engineering solutions. The simplified waste profile, devoid of heavy metal residues from exotic catalysts or hazardous gas scrubbing byproducts, streamlines waste disposal logistics and reduces the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios or R&D pipelines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-making processes.
Q: Why is the solid carbon monoxide substitute preferred over gaseous CO?
A: Using 1,3,5-tricarboxylate phenol ester (TFBen) as a solid substitute eliminates the safety hazards and specialized equipment requirements associated with toxic carbon monoxide gas, significantly simplifying operational safety and facility compliance.
Q: What is the substrate compatibility of this synthesis method?
A: The method demonstrates excellent substrate applicability, accommodating various substituents on the aryl ring including halogens, alkyl groups, and nitro groups, allowing for the design of diverse quinazolinone libraries.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes cheap and easily obtainable starting materials and avoids harsh conditions, making it highly practical for commercial scale-up of complex pharmaceutical intermediates with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods like this palladium-catalyzed carbonylation can be successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl quinazolinone derivatives conforms to the highest quality standards required for drug development and manufacturing.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs, unlocking new possibilities for your drug discovery programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your supply chain goals and accelerate your path to market.
