Scalable One-Pot Synthesis of Isoindolin-1-One Fused Rings for Pharmaceutical Applications
Scalable One-Pot Synthesis of Isoindolin-1-One Fused Rings for Pharmaceutical Applications
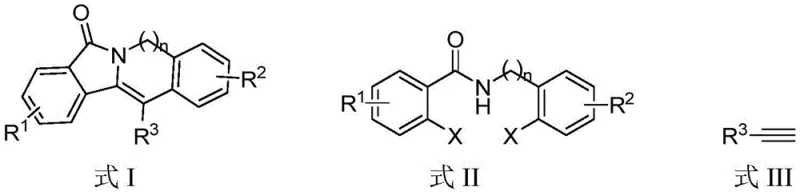
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN114133389B introduces a transformative methodology for the preparation of isoindoline-1-ketone ring derivatives, specifically targeting the structural general formula I. This innovation addresses the longstanding challenges associated with synthesizing these privileged structures, which are known for their potent physiological activities ranging from hNK1 receptor antagonism to anticancer properties. By leveraging a palladium and copper co-catalyzed tandem reaction, this technology enables the direct construction of fused ring systems from readily available dihalogenated benzamide compounds and terminal alkynes. For R&D directors and procurement specialists, this represents a pivotal shift towards more atom-economical and cost-effective manufacturing strategies, eliminating the need for cumbersome multi-step sequences that have historically plagued the production of these high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoindolin-1-one fused ring structures has been fraught with inefficiencies and stringent substrate requirements that hinder large-scale production. Traditional literature methods often rely on two-step processes involving condensation reactions between o-bromoaniline compounds and acetaldehyde, followed by coupling with o-iodobenzoyl chloride and subsequent intramolecular Heck cyclization. These conventional routes suffer from significant drawbacks, including the necessity for expensive and specific halogenated starting materials, such as requiring iodine at the ortho position of benzoyl chloride and bromine at the aniline ortho position. Furthermore, these legacy methods typically demand high loadings of palladium catalysts, often upwards of 10mol% to 20mol%, which drastically inflates the raw material costs and complicates the removal of heavy metal residues from the final active pharmaceutical ingredients. Alternative approaches involving electro-reduction cyclization add another layer of complexity by requiring specialized instrumentation and complex raw material structures, making them unsuitable for robust commercial supply chains.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach disclosed in the patent utilizes a highly efficient one-pot tandem reaction strategy that streamlines the entire synthetic sequence into a single operational step. This method employs dihalogenated benzamide compounds, which are significantly easier to synthesize and procure than the specialized substrates required by older methods, reacting them directly with terminal alkynes in the presence of a palladium catalyst containing phosphine ligands and cuprous iodide. The reaction proceeds smoothly in polar aprotic solvents like DMSO under inert atmospheric conditions, facilitating the formation of multiple chemical bonds simultaneously with high step economy. As illustrated in the reaction scheme below, this process allows for the versatile introduction of various substituents (R1, R2, R3) including aryl, heteroaryl, and electron-withdrawing groups, thereby enabling the rapid generation of diverse chemical libraries for drug discovery efforts without the need for route redesign.
Mechanistic Insights into Pd-Cu Catalyzed Tandem Cyclization
The core of this technological breakthrough lies in the synergistic catalytic cycle involving both palladium and copper species, which orchestrates the sequential bond-forming events required to close the fused ring system. The mechanism likely initiates with a Sonogashira-type cross-coupling between the terminal alkyne and the aryl halide moiety of the benzamide substrate, mediated by the Pd(0) species generated in situ from precursors like Pd(PPh3)2Cl2 and the CuI co-catalyst. Following the initial alkyne insertion, the system undergoes an intramolecular nucleophilic attack or cyclization event, potentially facilitated by the base, to form the isoindolin-1-one core. This tandem sequence is meticulously optimized to prevent side reactions such as homocoupling of the alkyne or dehalogenation, which are common pitfalls in palladium-catalyzed processes. The use of specific phosphine ligands, such as triphenylphosphine or dppf, stabilizes the palladium center and modulates its electronic properties, ensuring high turnover numbers and minimizing catalyst deactivation throughout the extended reaction times of 12 to 36 hours.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over stepwise approaches by reducing the number of isolation points where impurities can accumulate. The reaction conditions, specifically the use of potassium carbonate as a mild base and DMSO as a high-boiling solvent, create an environment that favors the desired cyclization over competing elimination or hydrolysis pathways. The patent data indicates that the reaction is highly selective, often yielding a single major product that can be easily purified via standard silica gel chromatography. This selectivity is crucial for pharmaceutical manufacturing, as it minimizes the formation of difficult-to-remove genotoxic impurities or structurally similar byproducts that could compromise the safety profile of the final drug substance. The ability to tune the electronic nature of the substrates, utilizing electron-withdrawing groups to enhance reactivity, further refines the impurity profile, ensuring that the resulting isoindolinone derivatives meet the stringent purity specifications required for clinical applications.
How to Synthesize Isoindolin-1-one Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process begins with the precise weighing of dihalogenated benzamide and terminal alkyne substrates, followed by the addition of the catalytic system comprising Pd(PPh3)2Cl2 and CuI in a molar ratio that maximizes efficiency while minimizing metal usage. The reaction mixture is suspended in DMSO and subjected to rigorous degassing cycles to establish an inert nitrogen atmosphere, which is critical for preventing the oxidation of the phosphine ligands and the palladium catalyst. Heating the mixture to 120°C for 24 hours allows the tandem cyclization to proceed to completion, after which the workup involves a straightforward extraction protocol using ethyl acetate and brine. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized guide below.
- Charge dihalogenated benzamide, terminal alkyne, Pd catalyst (e.g., Pd(PPh3)2Cl2), CuI, and base (K2CO3) into a reactor with DMSO solvent.
- Purge the system with nitrogen to create an inert atmosphere and heat the mixture to 120°C for approximately 24 hours.
- Upon completion, perform aqueous workup with ethyl acetate extraction, dry the organic phase, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by replacing expensive, specialty halogenated benzoic acid derivatives with commodity dihalogenated benzamides, manufacturers can secure more stable pricing and reduce dependency on niche suppliers. Furthermore, the reduction in catalyst loading from the industry-standard 10-20mol% down to merely 2.5mol% of palladium represents a massive decrease in the cost of goods sold (COGS), particularly given the volatile market price of precious metals. This efficiency is compounded by the elimination of intermediate isolation steps, which reduces solvent consumption, labor hours, and waste generation, aligning perfectly with modern green chemistry initiatives and environmental compliance standards.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant reduction in catalyst consumption and the use of inexpensive, commercially available solvents like DMSO. By lowering the palladium loading to 2.5mol% and utilizing cuprous iodide as a cheap co-catalyst, the overall material cost per kilogram of product is substantially decreased compared to traditional Heck coupling routes. Additionally, the one-pot nature of the reaction eliminates the need for intermediate purification and drying steps, which reduces energy consumption and equipment occupancy time, leading to lower overhead costs and improved throughput in multipurpose manufacturing facilities.
- Enhanced Supply Chain Reliability: The reliance on robust, widely available starting materials such as dihalogenated benzamides and terminal alkynes mitigates the risk of supply disruptions that often plague projects dependent on custom-synthesized intermediates. The process tolerance to various functional groups means that a single synthetic platform can be used to produce a wide array of analogues, allowing for flexible inventory management and rapid response to changing market demands. This versatility ensures that production schedules remain uninterrupted even if specific substrate variants face temporary availability issues, as the core chemistry remains consistent across the product family.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by the use of standard heating and stirring equipment without the need for specialized electrochemical cells or high-pressure reactors. The workup procedure, involving simple liquid-liquid extraction and silica gel chromatography, is easily adaptable to industrial continuous processing or large-batch operations. Moreover, the high atom economy and reduced waste profile associated with the tandem reaction support sustainability goals, making it easier to obtain regulatory approvals and maintain compliance with increasingly strict environmental regulations regarding heavy metal discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoindolinone synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in patent CN114133389B, providing a reliable foundation for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios or new drug development pipelines.
Q: What are the advantages of this one-pot method over traditional Heck reactions?
A: This method eliminates the need for multi-step synthesis and specialized substrates like ortho-iodobenzoyl chloride. It utilizes cheaper dihalogenated benzamides and reduces palladium catalyst loading from 20mol% to as low as 2.5mol%, significantly lowering production costs.
Q: What is the typical yield and purity profile for these isoindolinone derivatives?
A: Experimental data indicates isolated yields ranging from 40% to 88%, with optimal conditions (120°C, 24h, K2CO3 base) achieving yields above 80%. The single-product nature of the reaction simplifies purification, ensuring high purity suitable for pharmaceutical applications.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the process uses common solvents like DMSO and standard bases like potassium carbonate. The operational simplicity, involving basic extraction and column chromatography, makes it highly adaptable for commercial scale-up without requiring specialized electro-reduction equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolin-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and optimizing production costs. Our team of expert chemists has thoroughly analyzed the potential of this Pd-Cu catalyzed tandem cyclization and is fully prepared to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoindolin-1-one intermediates we deliver meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this one-pot process for your target molecules. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data, and receive comprehensive route feasibility assessments that will empower your organization to achieve superior supply chain performance and cost efficiency.
