Advanced CuI-Catalyzed Synthesis of Functionalized Pyridine Compounds for Pharmaceutical Applications
Introduction to Novel Pyridine Synthesis Technology
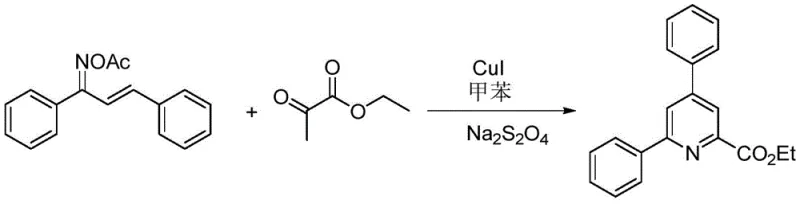
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for efficient routes to nitrogen-containing scaffolds that serve as the backbone of countless active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN112358443A, which discloses a robust and versatile method for preparing functionalized pyridine compounds. This technology leverages a copper-catalyzed cyclization strategy that merges alpha,beta-unsaturated ketoxime acetates with ethyl pyruvate, offering a streamlined alternative to classical heterocycle construction. For R&D directors and process chemists, the appeal lies not just in the novelty of the transformation, but in its operational simplicity and the structural diversity it unlocks. By utilizing inexpensive copper salts and mild reducing agents, this process circumvents the need for precious metal catalysts often associated with cross-coupling reactions, thereby presenting a compelling value proposition for cost-sensitive manufacturing environments.
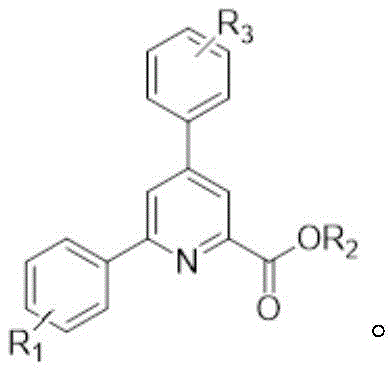
The significance of pyridine derivatives in the pharmaceutical and agrochemical sectors cannot be overstated, as they are pervasive motifs in drugs ranging from antihypertensives to antibiotics. Traditional synthetic approaches often suffer from limitations such as narrow substrate scope, harsh reaction conditions, or the generation of difficult-to-remove impurities. The methodology outlined in this patent addresses these pain points directly by providing a pathway that is both rapid and high-yielding. The ability to construct the pyridine core with precise substitution patterns at the 2, 4, and 6 positions allows for the rapid assembly of complex molecular architectures. This capability is critical for pharmaceutical companies aiming to accelerate their lead optimization phases, where the speed of analog synthesis can determine the success of a drug candidate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyridines has relied on multistep sequences involving condensation reactions, such as the Hantzsch pyridine synthesis or variations thereof. While effective, these classical methods often require stoichiometric amounts of reagents, generate significant waste, and struggle with regioselectivity when unsymmetrical substrates are employed. Furthermore, transition metal-catalyzed approaches using palladium or rhodium, while powerful, introduce substantial cost burdens due to the price of the metals and the rigorous purification steps required to meet residual metal specifications for pharmaceutical use. Iron-catalyzed methods, such as those reported by Yi in 2017 using FeCl3, have offered some relief but can still be limited by the specific nature of the aldehyde partners and the symmetry of the resulting products. These constraints often force process chemists to compromise on yield or purity, leading to extended development timelines and increased overall production costs.
The Novel Approach
In stark contrast, the novel approach described in patent CN112358443A utilizes a copper(I) iodide (CuI) catalytic system in conjunction with sodium hydrosulfite (Na2S2O4) to drive the cyclization of alpha,beta-unsaturated ketoxime acetates with ethyl pyruvate. This method represents a paradigm shift by enabling the direct construction of the pyridine ring under relatively mild thermal conditions, typically between 100°C and 140°C. The use of ethyl pyruvate as a two-carbon synthon introduces an ester functionality directly onto the pyridine ring, which serves as a valuable handle for further derivatization. The reaction is remarkably tolerant of various electronic and steric environments, accommodating substrates with electron-withdrawing halogens or electron-donating methyl and methoxy groups without significant loss in efficiency. This broad applicability makes it an ideal candidate for the late-stage functionalization of complex molecules.
Mechanistic Insights into CuI-Catalyzed Cyclization
The mechanistic pathway of this transformation is a fascinating interplay of radical and organometallic processes that ensures high efficiency and selectivity. The reaction initiates with the activation of the oxime acetate by the copper catalyst, facilitating the homolytic cleavage of the nitrogen-oxygen bond. This step generates a reactive nitrogen-centered radical or a copper-nitrenoid species, which is pivotal for the subsequent cyclization event. The presence of sodium hydrosulfite acts as a crucial reducing agent, maintaining the copper in its active +1 oxidation state and preventing the formation of inactive copper(II) species that could stall the catalytic cycle. This redox management is essential for sustaining the turnover number of the catalyst and ensuring that the reaction proceeds to completion without the need for excessive catalyst loading.
Following the initial activation, the reactive intermediate undergoes an addition reaction with the electron-deficient double bond of the ethyl pyruvate. This is followed by an elimination step that restores the double bond character, leading to an intermediate capable of undergoing a 6-pi electrocyclization. The final aromatization step is driven by the rapid oxidation facilitated by the copper species, locking in the aromatic pyridine structure. This intricate dance of bond breaking and forming occurs with high fidelity, minimizing the formation of side products such as polymeric tars or isomeric byproducts. For the R&D director, understanding this mechanism provides confidence in the robustness of the process, as the defined pathway suggests that impurity profiles will be consistent and manageable, simplifying the downstream purification protocols required for GMP manufacturing.
How to Synthesize Functionalized Pyridine Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol is designed to be operationally simple, avoiding the need for inert atmosphere techniques in many cases, although standard good laboratory practices are recommended. The choice of solvent plays a critical role, with toluene, acetonitrile, tetrahydrofuran, and 1,2-dichloroethane all proving effective, allowing flexibility based on solubility and boiling point requirements. The molar ratios are optimized to ensure complete conversion of the limiting reagent while minimizing waste, typically employing a slight excess of the oxime acetate relative to the pyruvate. Detailed standardized synthesis steps see the guide below.
- Mix alpha,beta-unsaturated ketoxime acetate, ethyl pyruvate, cuprous iodide (CuI), and sodium hydrosulfite (Na2S2O4) in an organic solvent such as toluene.
- Heat the reaction mixture to a temperature between 100°C and 140°C and maintain stirring for 8 to 12 hours to facilitate the cyclization.
- Filter the reaction solution, concentrate the filtrate, and purify the residue via column chromatography using petroleum ether and ethyl acetate to isolate the target pyridine compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers distinct advantages that translate directly into bottom-line savings and operational resilience. The reliance on commodity chemicals like ethyl pyruvate and copper iodide means that the raw material supply chain is robust and less susceptible to the geopolitical volatility often associated with rare earth or precious metal catalysts. Furthermore, the mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with corporate sustainability goals and potentially lowering utility costs. The simplicity of the work-up procedure, involving basic filtration and chromatography, reduces the demand for specialized equipment and skilled labor, further driving down the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts results in a drastic reduction in raw material costs. Additionally, the use of sodium hydrosulfite as a cheap and effective reducing agent avoids the need for costly phosphines or specialized ligands. The high atom economy of the reaction ensures that a significant portion of the starting mass ends up in the final product, minimizing waste disposal fees. This economic efficiency makes the process highly competitive for the production of high-volume pharmaceutical intermediates where margin pressure is intense.
- Enhanced Supply Chain Reliability: The starting materials, specifically alpha,beta-unsaturated ketoxime acetates and ethyl pyruvate, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The stability of these reagents allows for bulk purchasing and long-term storage, enabling manufacturers to buffer against market fluctuations. Moreover, the reaction's tolerance to various functional groups means that a single platform technology can be used to produce a wide array of derivatives, simplifying inventory management and reducing the need for multiple specialized synthetic routes.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in batch reactors without the need for exotic flow chemistry setups. The absence of toxic heavy metals in the final product simplifies regulatory compliance, particularly regarding ICH Q3D guidelines for elemental impurities. The use of common organic solvents like toluene facilitates solvent recovery and recycling, contributing to a greener manufacturing profile. This ease of scale-up ensures that the transition from gram-scale R&D to multi-ton commercial production can be achieved with minimal technical risk and shortened lead times.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the patent specifications and practical application scenarios. Understanding these details helps stakeholders make informed decisions about adopting this technology for their specific projects. The answers reflect the balance between theoretical potential and practical manufacturability.
Q: What are the key advantages of this CuI-catalyzed method over traditional iron-catalyzed routes?
A: Unlike traditional iron-catalyzed routes which may require harsher conditions or specific aldehyde substrates, this CuI-catalyzed method utilizes readily available alpha,beta-unsaturated ketoxime acetates and ethyl pyruvate. It operates under milder thermal conditions (100-140°C) and demonstrates broader substrate tolerance, allowing for the synthesis of more complex and diversified pyridine structures essential for modern drug discovery.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up. The process uses inexpensive and stable catalysts (CuI) and reducing agents (Na2S2O4), and the work-up involves simple filtration and standard chromatography. The mild reaction conditions reduce energy consumption and safety risks associated with high-pressure or extreme temperature processes, making it economically viable for commercial manufacturing.
Q: What types of substituents are tolerated on the pyridine ring using this protocol?
A: The protocol exhibits excellent functional group tolerance. The R1 and R3 positions on the aromatic rings can accommodate hydrogen, halogens (such as chlorine), nitro groups, methyl, or methoxy groups. This versatility allows medicinal chemists to rapidly generate libraries of substituted pyridine derivatives for structure-activity relationship (SAR) studies without needing to protect sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our team of expert chemists has thoroughly evaluated the technology described in CN112358443A and is fully equipped to leverage this CuI-catalyzed methodology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from the bench to the plant. Our state-of-the-art facilities are designed to handle complex organic transformations with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee product quality.
We invite you to collaborate with us to explore the full potential of this pyridine synthesis route for your pipeline. Whether you require kilogram quantities for preclinical studies or metric tons for commercial launch, our flexible manufacturing capabilities can be tailored to your specific timeline and budget. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development program and optimize your supply chain.
