Advanced Azetidinone Derivative Synthesis for Commercial Ezetimibe Production
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value cholesterol absorption inhibitors, specifically focusing on the efficient production of ezetimibe and its key precursors. Patent CN100564357C introduces a groundbreaking methodology for synthesizing azetidinone derivatives, which serve as critical intermediates in the manufacturing of this vital cardiovascular medication. This intellectual property details a novel chemical route that circumvents the traditional reliance on costly transition metal catalysts, offering a streamlined approach to constructing the complex four-membered azetidinone core. By leveraging a Grignard addition strategy to a Weinreb amide scaffold, the disclosed technology ensures high stereochemical fidelity while drastically simplifying the purification workflow. For global procurement teams and R&D directors, understanding this shift from palladium-catalyzed cross-coupling to nucleophilic addition represents a significant opportunity for optimizing supply chain resilience and reducing overall production expenditures without compromising molecular integrity.
The Limitations of Conventional Methods vs. The Novel Approach
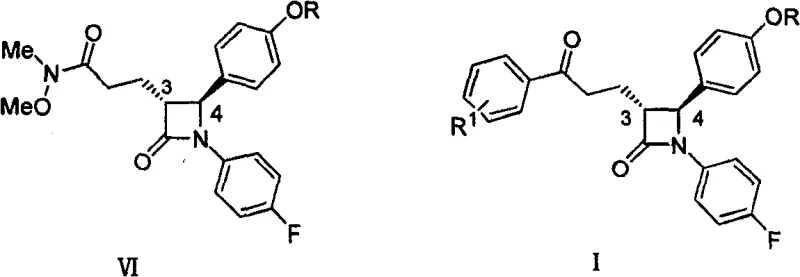
The Limitations of Conventional Methods
Historically, the construction of the side chain on the azetidinone ring has relied heavily on palladium-catalyzed coupling reactions, such as those described in earlier literature like U.S. Pat 5,767,115. These conventional methods typically involve the reaction of a chloride compound with an organic zinc reagent in the presence of catalysts like tetrakis(triphenylphosphine)palladium [Pd(PPh3)4]. A major drawback of this legacy technology is the substantial consumption of precious metal catalysts, often requiring loadings of 10% to 20% by weight relative to the substrate. This high catalyst loading not only inflates the raw material costs significantly but also introduces complex downstream processing challenges related to the removal of residual palladium to meet stringent pharmaceutical purity standards. Furthermore, the sensitivity of palladium catalysts to reaction conditions can lead to batch-to-batch variability, posing risks to supply chain consistency and complicating the regulatory validation process for generic manufacturers seeking to establish reliable production lines.
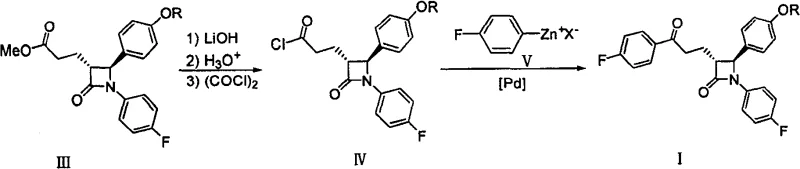
The Novel Approach
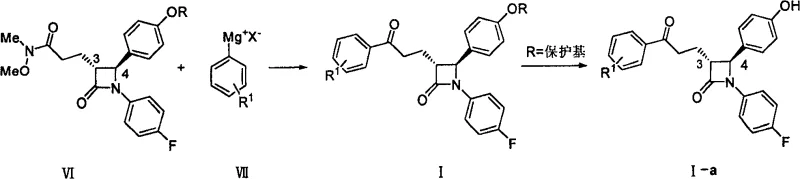
In stark contrast to the metal-dependent legacy routes, the technology disclosed in CN100564357C utilizes a Grignard reagent addition to a Weinreb amide intermediate, effectively bypassing the need for any transition metal catalyst. This innovative pathway involves reacting a protected azetidinone Weinreb amide (Formula VI) with a substituted phenyl magnesium halide (Formula VII) under controlled thermal conditions. The elimination of palladium not only removes the cost burden associated with precious metals but also eradicates the risk of heavy metal contamination in the final active pharmaceutical ingredient. The reaction proceeds smoothly in common ether solvents such as tetrahydrofuran at moderate temperatures ranging from -10°C to 30°C, demonstrating excellent functional group tolerance. This methodological shift transforms the synthesis into a more robust and economically viable process, aligning perfectly with modern green chemistry principles and the industry's drive towards more sustainable and cost-efficient manufacturing protocols for high-volume pharmaceutical intermediates.
Mechanistic Insights into Grignard Addition to Weinreb Amides
The core chemical transformation driving this synthetic advancement is the nucleophilic addition of the Grignard reagent to the Weinreb amide functionality present on the azetidinone side chain. Mechanistically, the Grignard reagent attacks the carbonyl carbon of the amide, forming a stable tetrahedral intermediate chelated by the methoxy group of the Weinreb moiety. This chelation effect is crucial as it prevents over-addition of the Grignard reagent, which would otherwise lead to the formation of tertiary alcohols instead of the desired ketone. Upon acidic workup, this stable intermediate collapses to release the N,O-dimethylhydroxylamine and yield the target ketone (Formula I) with high chemoselectivity. The preservation of the azetidinone ring integrity during this strongly nucleophilic process highlights the robustness of the protecting group strategy employed, ensuring that the sensitive beta-lactam structure remains intact throughout the harsh reaction conditions required for carbon-carbon bond formation.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed coupling reactions. The absence of transition metals eliminates the formation of metal-associated byproducts and simplifies the impurity profile significantly. The primary impurities generated are typically derived from unreacted starting materials or hydrolysis products, which are generally easier to separate via standard crystallization or chromatography techniques compared to organometallic residues. Furthermore, the stereochemistry at the 3- and 4-positions of the azetidinone ring is maintained throughout the reaction, as the Grignard addition occurs remotely from the chiral centers on the ring. This stereoretentive characteristic is vital for producing the specific enantiomers required for biological activity, ensuring that the optical purity of the starting material is faithfully transferred to the final intermediate without the need for costly chiral resolution steps post-reaction.
How to Synthesize Azetidinone Derivatives Efficiently
The practical implementation of this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the Weinreb amide precursor, followed by the careful addition of the Grignard reagent at low temperatures to manage exothermicity. Detailed operational procedures, including specific solvent ratios, addition rates, and quenching protocols, are essential for reproducing the high yields reported in the patent examples. Operators must ensure strict anhydrous conditions during the Grignard step to prevent reagent decomposition. The following section outlines the standardized synthesis steps derived directly from the experimental data provided in the patent documentation, serving as a foundational guide for process chemists aiming to replicate this efficient route.
- Prepare the Weinreb amide intermediate (Formula VI) by reacting the corresponding acid chloride with N,O-dimethylhydroxylamine.
- React the Weinreb amide with a substituted phenyl Grignard reagent (Formula VII) in an inert ether solvent at temperatures between -35°C to 50°C.
- Perform deprotection of the phenolic hydroxyl group via hydrogenation or acid hydrolysis to yield the final ketone intermediate (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this palladium-free synthesis route translates into tangible strategic benefits that extend beyond simple chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials, as Grignard reagents are commodity chemicals available from multiple global suppliers, unlike specialized palladium catalysts which are subject to geopolitical supply risks and price volatility. By removing the dependency on precious metals, manufacturers can secure more stable pricing contracts and reduce the inventory costs associated with holding expensive catalysts. Additionally, the simplified workup procedure reduces the consumption of scavenging resins and specialized filtration media typically required for metal removal, further lowering the operational expenditure per kilogram of produced intermediate.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts, which are traditionally used in significant quantities for this transformation, results in substantial direct material cost savings. Without the need for expensive metal scavengers or extensive purification steps to meet residual metal specifications, the overall processing cost is significantly reduced. This economic efficiency allows for more competitive pricing strategies in the generic pharmaceutical market while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on widely available Grignard reagents and standard ether solvents mitigates the risk of supply disruptions often associated with specialized catalytic systems. The robustness of the reaction conditions ensures consistent batch quality, reducing the likelihood of production delays caused by failed batches or out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream API manufacturers and meeting strict delivery schedules.
- Scalability and Environmental Compliance: The process utilizes common solvents and avoids heavy metals, making it inherently safer and more environmentally friendly for large-scale production. The reduced waste burden from metal residues simplifies effluent treatment and compliance with environmental regulations. This scalability ensures that the process can be seamlessly transferred from pilot plant to commercial manufacturing scales without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived from the specific technical disclosures and experimental data found within the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details helps stakeholders make informed decisions regarding technology transfer and vendor qualification.
Q: How does this new method improve upon traditional palladium-catalyzed coupling?
A: The novel method eliminates the need for expensive transition metal catalysts like tetrakis(triphenylphosphine)palladium, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: What is the stereochemical outcome of the Grignard addition?
A: The reaction preserves the trans-configuration at the 3,4-positions of the azetidinone ring, allowing for the production of single enantiomers or specific racemic mixtures depending on the starting material.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of common Grignard reagents and standard ether solvents, combined with mild reaction conditions (-10°C to 30°C), makes this route highly scalable and safer for commercial production compared to sensitive palladium couplings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to leverage advanced synthetic routes like the one described in CN100564357C to deliver superior pharmaceutical intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this palladium-free chemistry are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azetidinone derivative meets the highest global regulatory standards, providing our partners with peace of mind regarding product quality and consistency.
We invite you to collaborate with us to optimize your supply chain for ezetimibe production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term commercial goals.
