Advanced Stereoselective Synthesis of Ezetimibe Intermediates for Commercial API Production
Advanced Stereoselective Synthesis of Ezetimibe Intermediates for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and highly stereoselective pathways for the production of critical cardiovascular therapeutics like ezetimibe. Patent CN101935309B, published in late 2013, introduces a groundbreaking methodology for preparing ezetimibe and its key intermediates, specifically addressing the formidable challenge of controlling three distinct chiral centers within the molecular architecture. This technology represents a significant leap forward in process chemistry, moving away from traditional, less efficient auxiliaries toward a novel tricyclic indene oxazole system that delivers exceptional optical purity. For R&D directors and procurement strategists, understanding this patented route is essential, as it offers a viable pathway to high-purity pharmaceutical intermediates with improved economic metrics and operational simplicity suitable for industrial-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ezetimibe has relied heavily on the use of (S)-4-benzyl-2-azolactone as a chiral auxiliary to establish the critical stereochemistry of the azetidinone ring. While functional, this conventional approach often suffers from limitations related to steric bulk and stereoselectivity efficiency. The relatively smaller steric profile of the benzyl group can sometimes lead to imperfect facial discrimination during the cyclization and reduction steps, potentially compromising the enantiomeric excess (ee) of the final product. Furthermore, the recovery and recycling of these traditional auxiliaries can be chemically demanding, leading to higher consumption rates of expensive chiral starting materials. These factors collectively contribute to increased production costs and more complex purification workflows, creating bottlenecks for supply chain managers aiming for consistent, high-volume output of active pharmaceutical ingredients.
The Novel Approach
In a decisive break from prior art, the methodology described in CN101935309B employs a sterically demanding tricyclic indene oxazole chiral auxiliary, specifically (3aS, 8aR)-3,3a,8,8a-tetrahydro-2H-indeno[1,2-d]oxazole-2-one. This bulky structural motif exerts a profound influence on the transition state geometry, effectively shielding one face of the reacting molecule and forcing the formation of the desired stereoisomer with remarkable precision. The implementation of this novel auxiliary allows for the efficient generation of two chiral centers in the azetidinone ring simultaneously, while the third chiral center at the 3' position is established via asymmetric reduction. This strategic shift not only simplifies the synthetic sequence but also dramatically enhances the overall stereochemical outcome, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolio.
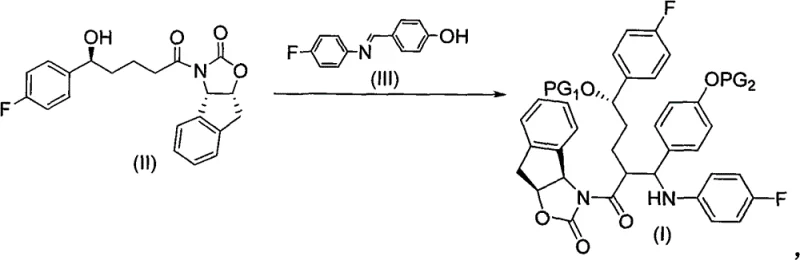
The core of this innovation lies in the coupling reaction depicted above, where the chiral alcohol intermediate reacts with a substituted imine in the presence of titanium tetrachloride and silylating agents. This one-pot transformation efficiently installs the necessary protecting groups and carbon framework, yielding the protected intermediate of general formula I. The ability to perform this complex assembly under relatively mild conditions (-25°C to -30°C) underscores the practical viability of the process for cost reduction in API manufacturing, as it avoids the need for extreme cryogenic temperatures or hazardous high-pressure environments often associated with less optimized routes.
Mechanistic Insights into Tricyclic Indene Oxazole Mediated Stereocontrol
The exceptional performance of this synthesis route is fundamentally rooted in the unique mechanistic behavior of the tricyclic indene oxazole auxiliary. Unlike planar or less constrained auxiliaries, the rigid fused-ring system of the indene moiety locks the oxazole ring into a specific conformation that maximizes steric repulsion with incoming electrophiles or nucleophiles. When the substrate approaches the chiral environment created by this auxiliary, the bulky tricyclic framework effectively blocks the Re-face (or Si-face, depending on the specific step), directing the attack exclusively to the opposite face. This phenomenon, known as diastereofacial differentiation, is the driving force behind the reported enantiomeric excess values reaching as high as 99.97%. Such high fidelity in stereocontrol is paramount for regulatory compliance, as it minimizes the formation of unwanted diastereomers that are difficult and costly to remove downstream.
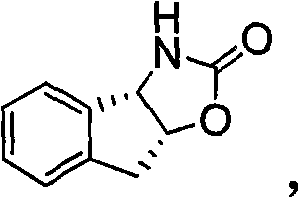
Furthermore, the mechanism facilitates impurity control by ensuring that side reactions leading to racemization are kinetically disfavored. The stability of the intermediate species formed during the amide bond formation and subsequent cyclization is enhanced by the electronic and steric properties of the indene system. This stability translates directly into cleaner reaction profiles, reducing the burden on purification units such as chromatography columns or crystallization tanks. For technical teams, this means a more predictable impurity profile and a reduced risk of batch failure due to out-of-specification optical purity, thereby securing the supply continuity of high-purity ezetimibe intermediates required for final drug substance production.
How to Synthesize Protected Ezetimibe Intermediate Efficiently
The synthesis of the key protected intermediate, designated as Compound I in the patent literature, follows a logical and scalable sequence designed for industrial application. The process begins with the activation of a keto-acid precursor, followed by coupling with the chiral auxiliary to set the initial stereochemistry. Subsequent asymmetric reduction establishes the side-chain hydroxyl group, and the final coupling with the fluorophenyl imine completes the carbon skeleton. This modular approach allows for rigorous quality control at each stage, ensuring that the final intermediate meets stringent purity specifications before proceeding to the final cyclization and deprotection steps to yield ezetimibe. The detailed standardized synthesis steps for this high-value intermediate are outlined in the guide below.
- Preparation of Compound IV via amide coupling of 5-(4-fluorophenyl)-5-carbonyl valeric acid with the tricyclic indene oxazole auxiliary using trimethylacetyl chloride activation.
- Asymmetric reduction of Compound IV using a chloroborane reagent to establish the 3' hydroxyl chiral center with high stereoselectivity.
- Titanium-mediated addition reaction with a substituted imine (Compound III) in the presence of silylating agents to form the protected intermediate Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages for procurement managers and supply chain heads focused on long-term sustainability and cost efficiency. The primary driver of value creation in this process is the recoverability of the chiral auxiliary. The patent explicitly notes that the tricyclic indene oxazole auxiliary can be recovered and recycled with a yield of approximately 95%. This high recovery rate effectively amortizes the cost of the chiral reagent over multiple batches, leading to substantial cost savings in raw material expenditure compared to processes where the auxiliary is consumed or discarded. Additionally, the use of common, commercially available reagents such as titanium tetrachloride and trimethylchlorosilane ensures that the supply chain is not dependent on exotic or single-source catalysts, mitigating supply risk.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of complex purification steps often required to separate closely related stereoisomers. Because the reaction achieves such high stereoselectivity (99.97% ee), the need for expensive chiral chromatography or multiple recrystallizations is significantly reduced. This streamlining of the downstream processing directly lowers the cost of goods sold (COGS) and improves the overall margin profile for the manufactured intermediate. Furthermore, the mild reaction conditions reduce energy consumption associated with heating or cooling, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations described ensures high batch-to-batch consistency, a critical factor for maintaining reliable supply lines to downstream API manufacturers. The intermediates generated are stable and can be handled using standard industrial equipment, reducing the likelihood of operational delays caused by specialized hardware requirements. By utilizing a route that relies on well-understood organic transformations rather than fragile biocatalytic systems or unstable organometallics, the supply chain becomes more resilient to disruptions, ensuring that delivery timelines for critical cardiovascular medication components are met without compromise.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, with reaction parameters that translate easily from kilogram to multi-ton scales. The solvent systems employed, primarily dichloromethane and toluene, are standard in the fine chemical industry and can be efficiently recovered and reused, aligning with modern environmental, health, and safety (EHS) standards. The high atom economy of the coupling steps and the recyclability of the chiral auxiliary minimize waste generation, supporting the industry's shift towards greener chemistry practices while maintaining high productivity levels.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ezetimibe synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential licensees or manufacturing partners.
Q: What is the primary advantage of the tricyclic indene oxazole auxiliary over conventional benzyl azolactones?
A: The tricyclic indene oxazole auxiliary possesses significantly larger steric hindrance, which enforces stricter conformational control during the reaction. This results in superior stereoselectivity, achieving ee values up to 99.97% compared to lower selectivity observed with traditional (S)-4-benzyl-2-azolactone auxiliaries.
Q: Is the chiral auxiliary recoverable in this process?
A: Yes, a critical economic advantage of this method is the recyclability of the chiral auxiliary. The patent data indicates that the auxiliary can be recovered and recycled with a yield of approximately 95%, drastically reducing the effective cost of goods for the chiral reagent.
Q: What are the typical reaction conditions for the key coupling step?
A: The key coupling reaction to form the protected intermediate utilizes mild conditions, typically involving titanium tetrachloride (TiCl4) and trimethylchlorosilane at low temperatures ranging from -25°C to -30°C, followed by warming to room temperature, ensuring safety and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of mastering complex chiral synthesis routes to meet the global demand for high-quality cardiovascular therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN101935309B can be translated into reliable, large-volume supply. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the 99.97% ee targets and ensure every batch of ezetimibe intermediate meets the highest international regulatory standards.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how our optimized manufacturing capabilities can enhance your production efficiency and secure your supply of this vital lipid-lowering medication.
