Advanced Catalytic Reduction of Iminodibenzyl Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to synthesize complex amine scaffolds, which serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN110372633B, which discloses a highly efficient and selective method for the catalytic reduction of iminodibenzyl carbonyl derivatives. This technology represents a paradigm shift from traditional transition-metal-catalyzed processes to a more sustainable, non-transition metal system utilizing sodium triethylborohydride (NaBEt3H). For R&D directors and process chemists, this innovation offers a compelling alternative that addresses long-standing challenges regarding catalyst cost, operational safety, and impurity profiles. By leveraging this novel catalytic system, manufacturers can achieve high-purity iminodibenzyl products under relatively mild conditions, thereby enhancing the overall feasibility of large-scale production for drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of inert amide bonds, particularly within the sterically constrained iminodibenzyl framework, has relied heavily on transition metal catalysis. Conventional protocols often employ precious metals such as Ruthenium, Manganese, or Nickel complexes to activate silanes or hydrogen gas for the reduction process. While these methods have achieved varying degrees of success in academic settings, they present substantial hurdles for industrial application. The primary drawback lies in the complexity and exorbitant cost of the catalysts, which often require sophisticated ligand systems that are difficult to synthesize and handle. Furthermore, the presence of transition metals in the final product necessitates extensive and costly purification steps to meet regulatory limits for heavy metal residues in pharmaceuticals. Additionally, many traditional methods require harsh reaction conditions, including high pressures of hydrogen gas or excessive amounts of reducing agents, which pose significant safety risks and environmental burdens in a manufacturing setting.
The Novel Approach
In stark contrast to the legacy technologies, the method described in patent CN110372633B introduces a streamlined catalytic cycle driven by sodium triethylborohydride. This approach utilizes a cheap and easily obtainable organic boron reagent, specifically catecholborane, to effect the reduction. The elimination of transition metals not only drastically reduces the raw material costs but also simplifies the supply chain by removing the dependency on scarce precious metals. The reaction proceeds efficiently in tetrahydrofuran (THF) at a moderate temperature of 80°C, avoiding the need for high-pressure equipment. This transition-metal-free strategy provides a practical new reaction pathway that is inherently safer and more environmentally benign. By adopting this methodology, chemical manufacturers can significantly reduce the complexity of their process workflows while maintaining high selectivity for the desired amine products.
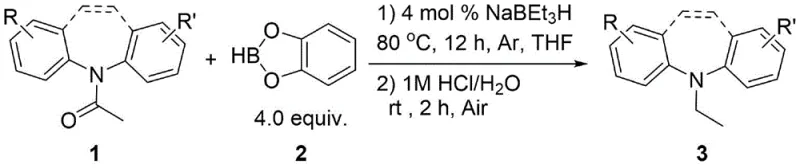
Mechanistic Insights into NaBEt3H-Catalyzed Reduction
The core of this technological advancement lies in the unique reactivity of the sodium triethylborohydride catalyst in conjunction with the organoboron reagent. Unlike traditional hydride sources that might lack the necessary potency to reduce stable amide bonds without aggressive conditions, the NaBEt3H system generates a highly reactive species capable of activating the carbonyl oxygen of the iminodibenzyl derivative. The mechanism likely involves the coordination of the borane reagent to the carbonyl oxygen, increasing its electrophilicity, followed by a hydride transfer from the borohydride catalyst. This synergistic interaction lowers the activation energy barrier for the reduction, allowing the reaction to proceed smoothly at 80°C. The catalytic cycle is sustained by the regeneration of the active borohydride species, ensuring that only a catalytic amount (4 mol %) is required to drive the transformation of multiple equivalents of the substrate. This efficiency is crucial for minimizing waste and maximizing atom economy in large-scale synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-hydride pathways. The specificity of the boron-mediated reduction minimizes side reactions such as over-reduction or the cleavage of sensitive functional groups often present in complex drug molecules. For instance, the method has been shown to tolerate halogen substituents like bromine and iodine, which are prone to dehalogenation under harsher reducing conditions. This chemoselectivity ensures that the final product stream contains fewer by-products, thereby reducing the burden on downstream purification units like crystallization or chromatography. For quality control teams, this translates to a more consistent impurity profile and a higher likelihood of passing stringent regulatory audits for API intermediates, ultimately safeguarding the integrity of the final drug substance.
How to Synthesize Iminodibenzyl Amines Efficiently
The implementation of this synthesis route is designed for operational simplicity, making it accessible for both laboratory optimization and pilot plant scale-up. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent moisture sensitivity issues associated with the borane reagents. The standardized protocol involves charging the reactor with the iminodibenzyl amide substrate, the borane reagent, and the solvent, followed by the precise addition of the catalyst. Maintaining the temperature at 80°C for the specified duration is critical to ensure full conversion without thermal degradation. Following the reaction, a straightforward acidic workup quenches any remaining reactive species and facilitates the isolation of the amine product. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Under an inert argon atmosphere, combine the iminodibenzyl amide substrate, catecholborane (4.0 equivalents), and anhydrous THF solvent in a sealed reaction vessel.
- Add sodium triethylborohydride (4 mol %) as the catalyst and heat the mixture to 80°C in an oil bath with stirring for 12 hours to ensure complete reduction.
- Upon completion, quench the reaction with 1M aqueous HCl under air, stir at room temperature for 2 hours, extract the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transition-metal-free reduction technology offers profound strategic benefits that extend beyond simple reaction yields. The shift away from precious metal catalysts directly impacts the cost structure of the manufacturing process. By replacing expensive Ruthenium or Nickel complexes with commodity chemicals like sodium triethylborohydride, the direct material costs are substantially lowered. Furthermore, the elimination of heavy metals from the process stream removes the need for specialized scavenger resins or complex extraction protocols designed to meet ppm-level metal specifications. This simplification of the purification train results in significant operational expenditure savings and reduces the consumption of auxiliary materials. The overall process becomes more lean and cost-effective, providing a competitive edge in the pricing of high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the substitution of high-cost catalytic systems with affordable, commercially available reagents. Traditional methods often require stoichiometric or near-stoichiometric amounts of expensive silanes or hydrogenation catalysts, whereas this method utilizes a catalytic loading of just 4 mol %. Additionally, the avoidance of transition metals eliminates the capital and operational costs associated with metal recovery and waste treatment facilities. The cumulative effect is a drastic reduction in the cost of goods sold (COGS) for iminodibenzyl derivatives, allowing for better margin management in volatile chemical markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by relying on reagents that are widely produced and easy to source. Precious metal catalysts are subject to geopolitical supply risks and price volatility, which can disrupt production schedules. In contrast, sodium triethylborohydride and catecholborane are stable commodities with robust global supply chains. This stability ensures consistent availability of raw materials, reducing the risk of production stoppages due to supplier shortages. Moreover, the simplified logistics of handling non-hazardous solid catalysts compared to pressurized hydrogen gas or pyrophoric metal complexes enhances workplace safety and reduces insurance and compliance overheads.
- Scalability and Environmental Compliance: The scalability of this process is supported by its mild operating conditions and lack of high-pressure requirements. Running the reaction at atmospheric pressure in standard glass-lined or stainless steel reactors facilitates easy scale-up from kilograms to tons without the need for specialized high-pressure autoclaves. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly strict global environmental regulations regarding effluent discharge. The greener profile of this synthesis route supports corporate sustainability goals and simplifies the permitting process for new manufacturing lines, ensuring long-term operational continuity in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reduction technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a reliable foundation for process evaluation. Understanding these nuances is essential for technical teams assessing the feasibility of integrating this method into existing production workflows. The answers highlight the versatility and robustness of the catalytic system across various substrate classes.
Q: What are the primary advantages of using NaBEt3H over transition metal catalysts?
A: The use of sodium triethylborohydride eliminates the need for expensive and toxic transition metals like Ruthenium or Nickel. This significantly simplifies the downstream purification process by removing the requirement for rigorous heavy metal scavenging, which is critical for meeting stringent pharmaceutical purity specifications.
Q: What are the optimal reaction conditions for this reduction protocol?
A: The protocol operates efficiently at 80°C in tetrahydrofuran (THF) under an argon atmosphere. The reaction typically requires 12 hours to reach completion, utilizing 4 mol % of the catalyst and 4.0 equivalents of the borane reagent.
Q: Is this method suitable for substrates with halogen substituents?
A: Yes, the method demonstrates excellent functional group tolerance. Experimental data confirms successful reduction of substrates containing bromine, iodine, and chlorine substituents without dehalogenation side reactions, preserving the integrity of the molecular scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iminodibenzyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the NaBEt3H-mediated reduction described in CN110372633B. As a leading CDMO and supplier of fine chemical intermediates, we possess the technical expertise to translate such innovative laboratory protocols into robust commercial manufacturing processes. Our facility is equipped with state-of-the-art reactors capable of handling sensitive organoboron chemistry under inert atmospheres, ensuring the highest standards of safety and quality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. Our rigorous QC labs enforce stringent purity specifications, ensuring that every batch of iminodibenzyl intermediate meets the exacting requirements of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain and deep process knowledge that can accelerate your time-to-market. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your manufacturing strategy for maximum efficiency and profitability.
