Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Intermediates for Pharmaceutical Applications
Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Intermediates for Pharmaceutical Applications
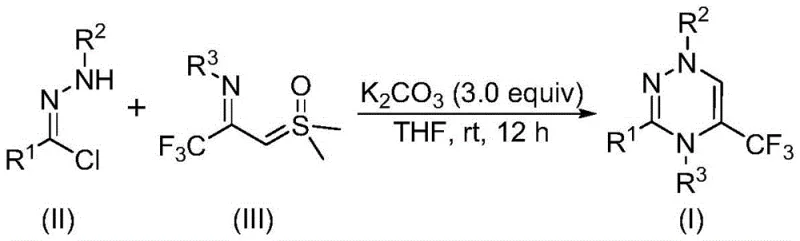
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles due to their profound biological activities. A recent breakthrough detailed in patent CN116253692A introduces a highly efficient preparation method for trifluoromethyl substituted 1,2,4-triazine compounds. This technology addresses critical bottlenecks in heterocyclic chemistry by utilizing a novel [3+3] cycloaddition strategy that operates under exceptionally mild conditions. Unlike traditional methods that often require harsh environments or toxic catalysts, this innovation leverages inexpensive chlorohydrazones and trifluoroacetyl thio ylides in the presence of potassium carbonate. The incorporation of the trifluoromethyl group is particularly strategic, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the resulting drug candidates. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine scaffold has relied on condensation reactions between amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical pathways are well-documented, they suffer from significant operational drawbacks that hinder industrial adoption. Conventional syntheses frequently necessitate the pre-synthesis of complex reaction substrates, which increases both the step count and the overall cost of goods. Furthermore, many existing protocols exhibit low reaction efficiency and poor structural diversity, limiting the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR). The requirement for stringent reaction conditions, such as high temperatures or inert atmospheres, further complicates the engineering controls needed for safe and reliable production at scale.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct cycloaddition between readily available chlorohydrazones and trifluoroacetyl thio ylides. This approach dramatically simplifies the synthetic workflow by eliminating the need for substrate pre-functionalization and avoiding the use of transition metal catalysts entirely. The reaction proceeds smoothly at room temperature (20-40°C) in an open air atmosphere, removing the capital expenditure associated with nitrogen blanketing systems. By employing potassium carbonate as a benign and inexpensive promoter, the process achieves high conversion rates with a broad tolerance for functional groups. This versatility allows for the rapid generation of diverse libraries of trifluoromethyl-substituted triazines, as evidenced by the successful synthesis of various derivatives including those with methoxy, halogen, and alkyl substituents.
Mechanistic Insights into Potassium Carbonate Promoted Cycloaddition
The mechanistic elegance of this transformation lies in the in situ generation of a reactive nitrile imine intermediate. Under the promotion of potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to eliminate one molecule of hydrogen chloride, forming the highly reactive nitrile imine species. This intermediate then engages in a synergistic [3+3] cycloaddition reaction with the trifluoroacetyl sulfur ylide. The process is concerted and efficient, ultimately leading to the formation of the six-membered 1,2,4-triazine ring with the concurrent elimination of one molecule of dimethyl sulfoxide (DMSO). Alternatively, the reaction may proceed through a stepwise pathway involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution. This dual mechanistic possibility ensures robustness across different substrate electronic profiles, providing R&D teams with a reliable tool for constructing complex heterocyclic cores.
From an impurity control perspective, the mild nature of this reaction is a distinct advantage. Traditional high-temperature cyclizations often lead to polymerization or decomposition of sensitive functional groups, resulting in complex impurity profiles that are difficult to purge. By operating at ambient temperatures, this new method minimizes thermal degradation and side reactions. The use of potassium carbonate, a weak inorganic base, prevents the harsh basic conditions that might otherwise hydrolyze esters or amides present on the aromatic rings. Consequently, the crude reaction mixture is cleaner, simplifying the downstream purification process and ensuring that the final high-purity intermediates meet the stringent quality specifications required for clinical trial materials.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
To implement this synthesis effectively, operators should dissolve the chlorohydrazone and trifluoroacetyl sulfur ylide in an aprotic organic solvent such as tetrahydrofuran (THF), which has been identified as the optimal medium for maximizing conversion. Potassium carbonate is then added in a molar ratio of approximately 3.0 equivalents relative to the chlorohydrazone to ensure complete deprotonation and intermediate formation. The detailed standardized synthesis steps, including specific stoichiometric ratios and workup procedures validated across multiple examples, are outlined in the guide below.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran (THF).
- Stir the mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this technology are substantial. The shift away from precious metal catalysis removes a major cost driver and supply chain vulnerability associated with fluctuating prices of palladium or copper. Additionally, the reliance on commodity chemicals like potassium carbonate and common solvents like THF ensures a stable and predictable supply of raw materials. The operational simplicity of running reactions in air at room temperature significantly reduces energy consumption and facility overheads, contributing to a lower overall carbon footprint for the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts directly translates to significant cost savings in raw material procurement. Furthermore, the absence of metal residues simplifies the purification process, potentially reducing the need for specialized scavenging resins or extensive recrystallization steps. This streamlined workflow lowers the cost of goods sold (COGS) and improves the margin profile for commercial production of these valuable intermediates.
- Enhanced Supply Chain Reliability: The starting materials, specifically chlorohydrazones and trifluoroacetyl thio ylides, are derived from widely available precursors such as acyl chlorides, hydrazines, and trifluoroacetic acid derivatives. This reliance on bulk commodity chemicals mitigates the risk of supply disruptions often seen with specialized reagents. The robustness of the reaction conditions also means that production can be easily transferred between different manufacturing sites without requiring highly specialized equipment or rigorous atmospheric controls.
- Scalability and Environmental Compliance: The process has been successfully demonstrated at the gram scale with straightforward post-treatment involving filtration and column chromatography. The use of non-toxic potassium carbonate and the avoidance of hazardous heavy metals align perfectly with modern green chemistry principles and environmental regulations. This compliance facilitates easier permitting for new production lines and reduces the costs associated with waste disposal and environmental remediation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive heavy metal catalysts and inert gas protection, operating efficiently at room temperature in air. It utilizes cheap, non-toxic potassium carbonate as a promoter and offers a broader substrate scope with high yields.
Q: What represents the rate-limiting step in this cycloaddition process?
A: The formation of the nitrile imine intermediate via the elimination of hydrogen chloride from the chlorohydrazone is a critical initiation step, followed by the synergistic [3+3] cycloaddition with the sulfur ylide.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process has been demonstrated to be scalable to gram levels with simple post-treatment procedures like filtration and column chromatography, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl substituted 1,2,4-triazine delivered meets the highest industry standards for safety and efficacy.
We invite you to leverage our technical expertise to optimize your supply chain and accelerate your drug development timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
