Revolutionizing Benzotriazole N2 Alkylation for Commercial Scale Pharmaceutical Manufacturing
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for higher selectivity and more sustainable manufacturing processes. A significant breakthrough in this domain is documented in patent CN111704584B, which introduces a highly selective N2 alkylation method for benzotriazole and its derivatives. Traditionally, achieving regioselective alkylation at the N2 position of the benzotriazole ring has been a formidable challenge for chemists, often resulting in mixtures dominated by the N1 isomer. This new approach leverages scandium trifluoromethanesulfonate as a catalyst to drive the reaction towards the desired N2 product with unprecedented precision. By utilizing cyclohexanone or its derivatives as the alkylating agent, this method operates under remarkably mild conditions, specifically at room temperature and in the presence of air. This represents a paradigm shift from conventional methods that typically demand harsh conditions, inert atmospheres, or expensive noble metal catalysts. For R&D directors and process chemists, this patent offers a robust pathway to access complex molecular backbones that are critical for drug discovery and development, ensuring that the synthetic route is not only feasible but also inherently efficient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of benzotriazole has been plagued by poor regioselectivity, where the reaction predominantly favors the formation of N1 alkylated products or yields a difficult-to-separate mixture of N1 and N2 isomers. Conventional strategies often rely on strong bases or transition metal catalysts that may require rigorous exclusion of moisture and oxygen, necessitating the use of inert gases like argon which adds operational complexity and cost. Furthermore, many existing protocols are limited to specific substrate scopes, such as alkenes or diazonium salts, restricting their utility in diverse synthetic campaigns. The reliance on noble metals in some advanced catalytic systems introduces significant cost pressures and potential contamination risks, requiring additional downstream purification steps to meet stringent pharmaceutical standards. These limitations collectively hinder the rapid scale-up of benzotriazole derivatives, creating bottlenecks in the supply chain for key pharmaceutical intermediates and increasing the overall cost of goods sold for the final active ingredients.
The Novel Approach
In stark contrast, the novel scandium-catalyzed method described in the patent data overcomes these historical barriers by enabling a direct and highly selective transformation. The use of scandium trifluoromethanesulfonate facilitates a condensation reaction followed by a hetero Michael addition, effectively steering the chemistry towards the N2 position with a selectivity ratio exceeding 99:1 in optimal cases. This approach eliminates the need for noble metals and inert gas protection, allowing the reaction to proceed smoothly in air at room temperature, which drastically simplifies the operational requirements. The substrate scope is notably broad, accommodating various electron-withdrawing and electron-donating groups on the benzotriazole ring, as well as different cyclohexanone derivatives. 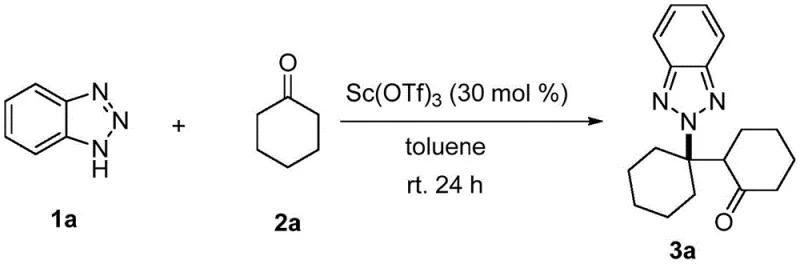
Mechanistic Insights into Scandium-Catalyzed N2 Alkylation
The mechanistic pathway of this transformation is rooted in the unique Lewis acidity of the scandium catalyst, which activates the carbonyl group of the cyclohexanone derivative towards nucleophilic attack by the benzotriazole nitrogen. This activation lowers the energy barrier for the initial condensation step, forming an intermediate that subsequently undergoes an intramolecular hetero Michael addition. The steric and electronic environment created by the scandium complex favors the attack at the N2 nitrogen over the N1 nitrogen, which is the thermodynamic driver for the observed high regioselectivity. Understanding this mechanism is crucial for process optimization, as it highlights the importance of catalyst loading and solvent choice in maintaining the integrity of the catalytic cycle. The reaction tolerates a wide range of functional groups, suggesting that the catalytic species is robust and does not easily decompose in the presence of potential impurities or varying electronic environments on the substrate.
Impurity control is a critical aspect of this mechanism, particularly regarding the suppression of the N1 alkylated byproduct. The high selectivity ratio, often greater than 99:1, implies that the transition state leading to the N2 product is significantly lower in energy than that for the N1 product under these specific catalytic conditions. This inherent selectivity reduces the burden on downstream purification processes, such as column chromatography or crystallization, which are often costly and time-consuming at scale. 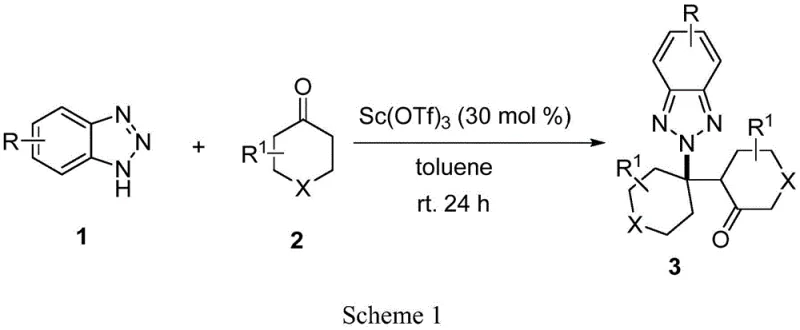
How to Synthesize Benzotriazole Derivatives Efficiently
To implement this synthesis effectively, one must adhere to the optimized parameters identified in the patent data, which balance yield and selectivity. The process involves combining the benzotriazole derivative and the cyclohexanone derivative in a suitable solvent, typically toluene, with a specific molar ratio of catalyst. While the detailed stoichiometry and workup procedures are critical for reproducibility, the general workflow is designed to be straightforward and amenable to standard laboratory equipment. For a comprehensive understanding of the precise reagent quantities, reaction times, and purification techniques required to achieve the reported high yields, please refer to the standardized synthesis guide provided below.
- Prepare the reaction mixture by adding benzotriazole derivatives, cyclohexanone derivatives, and scandium trifluoromethanesulfonate catalyst into a reaction vessel with toluene solvent.
- Stir the reaction mixture at room temperature in air for approximately 24 hours to allow the condensation and hetero Michael addition to proceed.
- Remove solvent and excess reactants under reduced pressure, then purify the crude product via column chromatography to isolate the high-purity N2 alkylated target.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers substantial advantages that directly impact the bottom line and supply chain resilience for procurement managers. The elimination of noble metal catalysts removes a significant cost driver and mitigates the risk of supply disruptions associated with rare earth or precious metal markets. Additionally, the ability to run the reaction at room temperature and in air translates to significant energy savings and reduced safety hazards, as there is no need for specialized high-pressure or cryogenic equipment. These factors collectively contribute to a more sustainable and cost-effective manufacturing process, making the sourcing of these intermediates more reliable and economically viable for long-term production contracts.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts and ligands from the process equation leads to a direct reduction in raw material costs. Furthermore, the simplified operational conditions eliminate the need for inert gas lines and specialized containment, reducing both capital expenditure on equipment and operational expenditure on utilities. The high selectivity of the reaction minimizes waste generation and reduces the solvent and adsorbent consumption required for purification, further driving down the overall cost of production for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like cyclohexanone and common solvents such as toluene, the supply chain for this synthesis is robust and less susceptible to geopolitical or logistical disruptions. The mild reaction conditions also imply a safer manufacturing environment, reducing the risk of plant shutdowns due to safety incidents. This stability ensures a consistent flow of materials to downstream customers, supporting just-in-time manufacturing models and reducing the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: The demonstrated success at the gram scale with high yields suggests a clear path to kilogram and ton-scale production without fundamental changes to the chemistry. The absence of heavy metal contaminants simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. This scalability ensures that the method can meet growing market demand for benzotriazole derivatives while maintaining a low environmental footprint, a key consideration for modern green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this scandium-catalyzed alkylation technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of adopting this route for your specific project requirements.
Q: What is the selectivity ratio of N2 versus N1 alkylation in this method?
A: The patented scandium-catalyzed method achieves an exceptionally high selectivity ratio, with N2 alkylated products to N1 alkylated products exceeding 99:1 in optimized conditions, significantly reducing purification burdens.
Q: Does this synthesis require expensive noble metal catalysts or inert gas protection?
A: No, the process utilizes scandium trifluoromethanesulfonate which is cost-effective compared to noble metals, and the reaction proceeds efficiently in air at room temperature, eliminating the need for argon protection.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method has been validated at the laboratory gram-scale with high yields and uses common solvents like toluene, indicating strong potential for commercial scale-up without complex engineering constraints.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzotriazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this scandium-catalyzed N2 alkylation route for the production of high-value pharmaceutical intermediates. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of benzotriazole derivatives meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthetic technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your project goals with reliability and expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
