Revolutionizing Nirmatrelvir Intermediate Production via Low-Temperature Cyclization Technology
Revolutionizing Nirmatrelvir Intermediate Production via Low-Temperature Cyclization Technology
The global pharmaceutical landscape has been profoundly reshaped by the urgent demand for effective antiviral therapeutics, specifically targeting the SARS-CoV-2 virus. At the heart of Pfizer's oral antiviral medication, Paxlovid, lies the protease inhibitor Nirmatrelvir (PF-07321332), a complex molecule whose supply chain stability is paramount for global health security. A critical bottleneck in the manufacturing of this life-saving drug is the production of its key chiral building block, 6,6-dimethyl-3-azabicyclo[3.1.0]hexane. Recent intellectual property developments, specifically patent CN114105859B, disclose a groundbreaking synthetic methodology that addresses the historical inefficiencies associated with this intermediate. By shifting away from energy-intensive high-temperature isomerization processes, this new technology offers a streamlined pathway that enhances both economic viability and environmental sustainability. The strategic importance of this innovation cannot be overstated, as it directly impacts the scalability and cost structure of the entire antiviral supply chain.
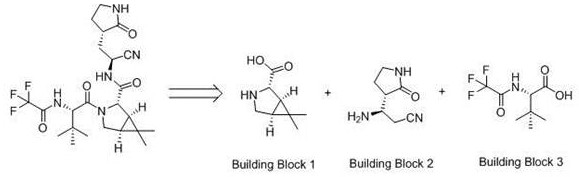
To fully appreciate the technical magnitude of this advancement, one must understand the structural complexity of the target molecule within the context of the final drug substance. As illustrated in the retrosynthetic analysis, Nirmatrelvir is assembled from three distinct fragments, with the bicyclic amine moiety serving as the central scaffold that dictates the stereochemical integrity of the final product. The conventional reliance on scarce precursors has often led to supply constraints and price volatility. However, the methodology outlined in CN114105859B introduces a robust alternative that begins with more accessible lactone or ketone derivatives. This shift not only diversifies the raw material base but also mitigates the risks associated with single-source suppliers. For procurement strategists and R&D leaders alike, this represents a pivotal opportunity to secure a more resilient supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane has been plagued by significant thermodynamic and economic hurdles. The traditional route, as documented in prior art such as WO 2007075790, typically commences with caronic anhydride, a commodity chemical characterized by limited manufacturer availability and a near-monopoly market status. This scarcity inherently drives up costs and creates vulnerability in the supply chain. Furthermore, the preparation of the requisite cis-configured intermediates from standard methyl chrysanthemate derivatives involves a strenuous isomerization step. This process necessitates heating the trans-diacid to extreme temperatures ranging from 190°C to 200°C to achieve the desired cis-configuration before dehydration can occur. Such high-energy requirements not only inflate operational expenditures but also demand specialized reactor equipment capable of withstanding severe thermal stress. Additionally, the harsh conditions often lead to increased formation of by-products, complicating downstream purification and generating substantial waste streams that require costly disposal protocols.
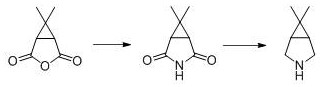
The Novel Approach
In stark contrast to the legacy methodologies, the innovative process described in patent CN114105859B circumvents these thermal bottlenecks through a clever manipulation of stereochemistry at the molecular level. By selecting 6,6-dimethyl-3-oxazolylcyclo[3.1.0]hexane-2-ketone as the starting material, the synthesis inherently produces the critical cis-carboxylic acid intermediate without the need for thermal isomerization. This fundamental change allows the subsequent cyclization and dehydration steps to proceed efficiently at mild temperatures, specifically between 10°C and 50°C. The elimination of the 200°C heating stage translates to a drastic reduction in energy consumption and removes the necessity for high-specification pressure vessels. Moreover, the use of common alkaline reagents and standard oxidants simplifies the reaction matrix, leading to cleaner reaction profiles and easier work-up procedures. This approach effectively decouples the production of this vital intermediate from the volatile caronic anhydride market, offering a more autonomous and controllable manufacturing pathway.
Mechanistic Insights into the Four-Step Cascade Synthesis
The core of this technological breakthrough lies in a meticulously orchestrated four-step cascade that transforms a functionalized lactone into the target bicyclic amine with high fidelity. The initial phase involves the alkaline hydrolysis of the starting ketone-lactone, followed immediately by oxidation of the hydroxymethyl group to a carboxylic acid. Utilizing reagents such as sodium hypochlorite or potassium permanganate in the presence of bases like sodium hydroxide ensures the rapid formation of cis-caronic diacid. Crucially, because the starting material possesses the correct stereochemical alignment, the resulting diacid is already in the thermodynamically favorable cis-configuration required for anhydride formation. This avoids the kinetic trap of the trans-isomer that plagues traditional routes. The subsequent dehydration step employs acetic anhydride as a mild dehydrating agent, facilitating the closure of the anhydride ring at ambient or slightly elevated temperatures. This gentle condition preserves the integrity of the cyclopropane ring, preventing unwanted ring-opening side reactions that are common under acidic or high-thermal stress.
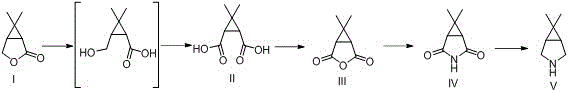
Following the formation of the anhydride, the pathway proceeds through an aminolysis reaction where ammonia or ammonium salts attack the carbonyl centers to open the anhydride and form the corresponding imide or amide intermediate. This step is conducted in polar solvents such as water or alcohols, ensuring high solubility and reaction homogeneity. The final transformation is a selective reduction of the amide functionality to the secondary amine. The patent details the use of powerful reducing systems, including lithium aluminum hydride or sodium borohydride coupled with Lewis acids like zinc chloride or boron trifluoride. This combination allows for the precise reduction of the carbonyl group to a methylene unit while maintaining the stereochemical configuration of the adjacent cyclopropane ring. The result is a high-purity 6,6-dimethyl-3-azabicyclo[3.1.0]hexane product that meets the stringent specifications required for API synthesis. The mechanistic elegance of this route lies in its linearity and the avoidance of protecting group manipulations, which significantly reduces the overall step count and material loss.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
Implementing this synthesis in a commercial setting requires strict adherence to the optimized parameters regarding reagent stoichiometry and temperature control to maximize yield and purity. The process is designed to be operationally simple, utilizing standard chemical engineering unit operations that are readily available in most fine chemical facilities. From the initial hydrolysis to the final distillation, each stage has been refined to minimize solvent usage and maximize throughput. For technical teams looking to adopt this methodology, the following guide outlines the critical operational phases derived from the patent examples. It is essential to note that while the chemistry is robust, precise control over the oxidation potential in the first step and the reduction strength in the final step is key to preventing over-reaction or degradation of the sensitive bicyclic core.
- Hydrolyze and oxidize 6,6-dimethyl-3-oxazolylcyclo[3.1.0]hexane-2-ketone using alkaline reagents and oxidants to form cis-caronic diacid.
- Dehydrate the cis-diacid using acetic anhydride at mild temperatures (10-50°C) to generate caronic anhydride.
- Perform aminolysis on the anhydride with ammonia sources to yield the caronamide intermediate.
- Reduce the caronamide using a borohydride or aluminum hydride system with Lewis acids to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the financial and logistical integrity of the pharmaceutical supply chain, the adoption of this novel synthetic route offers compelling strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the fundamental restructuring of the cost base associated with producing this critical intermediate. By eliminating the dependency on caronic anhydride, manufacturers can bypass a supply node that is historically prone to price spikes and allocation issues due to limited global production capacity. Furthermore, the shift from high-temperature batch processing to mild-temperature reactions significantly lowers the utility burden on the manufacturing plant. The reduction in energy intensity not only aligns with corporate sustainability goals but also provides a buffer against fluctuating energy prices, ensuring more predictable long-term manufacturing costs. This stability is crucial for long-term supply agreements where fixed pricing models are often preferred by large multinational pharmaceutical buyers.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of expensive, monopoly-controlled raw materials and the simplification of the process infrastructure. Traditional methods require specialized reactors capable of sustaining temperatures near 200°C, which entails higher capital expenditure for equipment and increased maintenance costs. In contrast, the new method operates effectively at room temperature or with mild heating, allowing the use of standard glass-lined or stainless steel reactors. Additionally, the avoidance of thermal isomerization reduces the formation of impurities, which in turn lowers the cost of goods sold by improving overall yield and reducing the load on purification units such as chromatography or recrystallization. The cumulative effect is a substantially leaner cost structure that enhances margin potential for contract manufacturing organizations.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the versatility of the starting materials employed in this new route. Unlike caronic anhydride, which has a concentrated supplier base, the precursors for this synthesis, such as substituted lactones or ketones, are derived from broader chemical feedstocks with multiple sourcing options. This diversification mitigates the risk of supply disruption caused by force majeure events at a single vendor site. Moreover, the simplified reaction conditions reduce the likelihood of batch failures due to equipment malfunction or thermal runaway, leading to more consistent production schedules. For supply chain directors, this reliability translates into shorter lead times and the ability to maintain lower safety stock levels, thereby freeing up working capital and improving cash flow efficiency across the organization.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner profile that facilitates easier permitting and scale-up. The reduction in energy consumption directly correlates to a lower carbon footprint, a metric that is increasingly scrutinized by regulators and investors alike. Furthermore, the use of aqueous work-ups and common organic solvents simplifies waste stream management, reducing the volume of hazardous waste requiring specialized treatment. The absence of heavy metal catalysts in the reduction step (when using borohydrides) further eases the burden on wastewater treatment facilities. These factors combined make the technology highly scalable, allowing manufacturers to ramp up production from pilot kilogram quantities to multi-ton commercial volumes without encountering the engineering bottlenecks typical of high-energy processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement officers negotiating supply contracts. The answers reflect the practical realities of scaling this chemistry from the laboratory bench to industrial reactors.
Q: How does this new synthesis method improve upon traditional caronic anhydride routes?
A: Traditional methods require high-temperature isomerization (190-200°C) to convert trans-diacids to cis-forms. This patent utilizes a precursor that naturally yields the cis-configuration, allowing cyclization at room temperature and significantly reducing energy consumption.
Q: What are the primary cost drivers eliminated in this process?
A: The process eliminates the reliance on expensive, monopoly-controlled caronic anhydride starting materials and removes the need for specialized high-temperature equipment, thereby lowering both raw material and operational expenditure.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method features simple post-treatment steps, reduced three-waste pollution, and operates under mild conditions, making it highly adaptable for commercial scale-up and continuous production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
As the global demand for antiviral therapeutics continues to evolve, securing a stable and high-quality supply of key intermediates like 6,6-dimethyl-3-azabicyclo[3.1.0]hexane is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic technologies to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for API synthesis. Our facility is equipped to handle complex chemistries safely and efficiently, making us an ideal partner for long-term strategic collaboration.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. Whether you are in the early stages of process development or looking to optimize an existing supply chain, we can provide a Customized Cost-Saving Analysis tailored to your volume and quality requirements. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the production of life-saving medicines.
