Revolutionizing 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Production via Continuous Catalytic Hydrogenation and Amination
The pharmaceutical industry constantly seeks robust, scalable pathways for critical heterocyclic intermediates, particularly those serving as the backbone for antiviral therapeutics. Patent CN115232059A introduces a transformative synthesis method for 6,6-dimethyl-3-azabicyclo[3.1.0]hexane, a pivotal scaffold utilized in the manufacture of advanced protease inhibitors. This molecule is not merely a chemical curiosity; it is the structural core of Boceprevir, a landmark hepatitis C treatment, and PF-07321332, a potent oral SARS-CoV-2 main protease inhibitor. ![Chemical structures of Boceprevir and PF-07321332 containing the 6,6-dimethyl-3-azabicyclo[3.1.0]hexane core](/insights/img/6-6-dimethyl-3-azabicyclo-hexane-catalytic-synthesis-pharma-supplier-20260315065312-02.webp) . The strategic importance of this intermediate cannot be overstated, as global demand for direct-acting antivirals continues to surge. Traditional synthetic routes have long been plagued by safety hazards and inefficiency, relying on stoichiometric reductions that generate massive amounts of toxic waste. The disclosed technology shifts the paradigm towards a continuous, catalytic process that leverages caronic anhydride as a readily available starting material. By employing a dual-catalyst system involving specific supported metals and metal oxides, the invention achieves high selectivity and yield while operating under conditions amenable to large-scale fixed-bed reactor configurations. For R&D directors and supply chain strategists, this represents a critical opportunity to secure a more resilient, cost-effective source of this high-value pharmaceutical intermediate.
. The strategic importance of this intermediate cannot be overstated, as global demand for direct-acting antivirals continues to surge. Traditional synthetic routes have long been plagued by safety hazards and inefficiency, relying on stoichiometric reductions that generate massive amounts of toxic waste. The disclosed technology shifts the paradigm towards a continuous, catalytic process that leverages caronic anhydride as a readily available starting material. By employing a dual-catalyst system involving specific supported metals and metal oxides, the invention achieves high selectivity and yield while operating under conditions amenable to large-scale fixed-bed reactor configurations. For R&D directors and supply chain strategists, this represents a critical opportunity to secure a more resilient, cost-effective source of this high-value pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 6,6-dimethyl-3-azabicyclo[3.1.0]hexane skeleton has relied heavily on the reduction of imide precursors. As illustrated in prior art, this typically involves the use of aggressive, stoichiometric reducing agents such as lithium aluminum hydride (LiAlH4), borane complexes, or sodium borohydride. 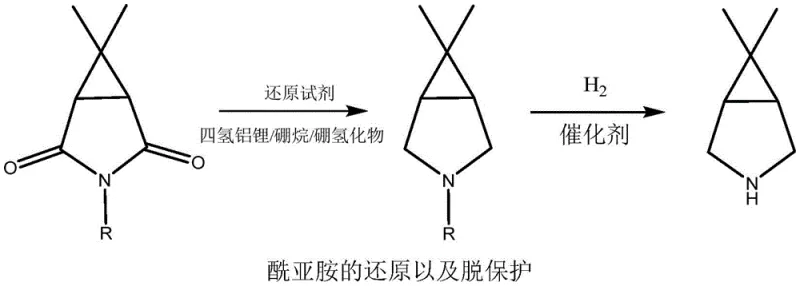 . These reagents pose severe safety risks due to their pyrophoric nature and sensitivity to moisture, necessitating specialized handling equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, the post-reaction workup for these reductions is notoriously complex, often requiring careful quenching steps that generate substantial volumes of aluminum or boron-containing solid waste and wastewater. Alternative routes, such as those starting from dichlorochrysanthemic acid, attempt to mitigate some reduction issues but introduce new bottlenecks. These multi-step sequences involve hydrolysis, ammoniation, halogenation, and Hofmann degradation, resulting in prolonged production cycles and the generation of halogenated organic waste streams that are difficult and expensive to treat. The reliance on batch processing in these legacy methods further limits throughput, creating supply chain vulnerabilities when demand for the final API spikes unexpectedly.
. These reagents pose severe safety risks due to their pyrophoric nature and sensitivity to moisture, necessitating specialized handling equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, the post-reaction workup for these reductions is notoriously complex, often requiring careful quenching steps that generate substantial volumes of aluminum or boron-containing solid waste and wastewater. Alternative routes, such as those starting from dichlorochrysanthemic acid, attempt to mitigate some reduction issues but introduce new bottlenecks. These multi-step sequences involve hydrolysis, ammoniation, halogenation, and Hofmann degradation, resulting in prolonged production cycles and the generation of halogenated organic waste streams that are difficult and expensive to treat. The reliance on batch processing in these legacy methods further limits throughput, creating supply chain vulnerabilities when demand for the final API spikes unexpectedly.
The Novel Approach
In stark contrast, the methodology disclosed in CN115232059A streamlines the synthesis into a concise, two-step catalytic sequence that bypasses the need for dangerous stoichiometric reductants entirely. The process initiates with the catalytic hydrogenation of caronic anhydride, a commercially accessible feedstock, to form 6,6-dimethyl-3-oxabicyclo[3.1.0]hexane. This intermediate is then subjected to a catalytic amination reaction with ammonia to yield the target azabicyclo amine. 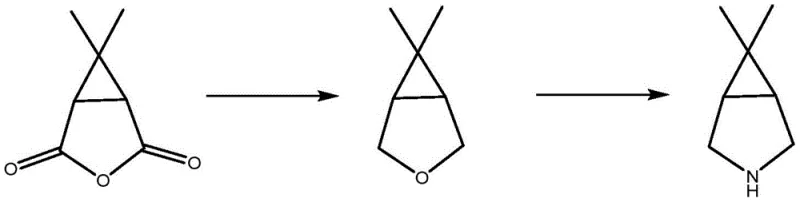 . This approach fundamentally alters the economic and safety profile of the manufacturing process. By utilizing heterogeneous catalysts in a continuous flow setup, the reaction can be run at elevated temperatures and pressures safely within fixed-bed reactors, eliminating the thermal runaway risks associated with batch hydride reductions. The atom economy is significantly improved, as the primary byproduct of the hydrogenation step is water, and the amination step releases minimal waste compared to the salt formation seen in older nucleophilic substitution routes. This shift from batch to continuous processing not only enhances safety but also dramatically improves space-time yields, allowing manufacturers to produce larger quantities of the intermediate in smaller footprints, thereby optimizing capital expenditure and operational efficiency.
. This approach fundamentally alters the economic and safety profile of the manufacturing process. By utilizing heterogeneous catalysts in a continuous flow setup, the reaction can be run at elevated temperatures and pressures safely within fixed-bed reactors, eliminating the thermal runaway risks associated with batch hydride reductions. The atom economy is significantly improved, as the primary byproduct of the hydrogenation step is water, and the amination step releases minimal waste compared to the salt formation seen in older nucleophilic substitution routes. This shift from batch to continuous processing not only enhances safety but also dramatically improves space-time yields, allowing manufacturers to produce larger quantities of the intermediate in smaller footprints, thereby optimizing capital expenditure and operational efficiency.
Mechanistic Insights into Catalytic Hydrogenation and Amination
The success of this novel route hinges on the precise selection and engineering of two distinct catalytic systems tailored for each transformation. In the first step, the hydrogenation of caronic anhydride requires a catalyst capable of selectively reducing the carbonyl groups without opening the strained cyclopropane ring or over-reducing the system. The patent specifies supported metal catalysts where the active component M is selected from Cu, Ni, Co, Pd, Pt, or Ru, dispersed on high-surface-area supports like silica, alumina, or zeolites. The synergy between the metal and the support is critical; for instance, copper-based catalysts on silica (Cu/SiO2) or bimetallic nickel-copper systems have shown exceptional performance. The mechanism likely involves the adsorption of the anhydride onto the metal surface, followed by the dissociative adsorption of hydrogen. The metal facilitates the transfer of hydrogen atoms to the carbonyl oxygen and carbon, sequentially reducing the anhydride to the cyclic ether. The choice of support, such as H-ZSM-5 or gamma-alumina, provides the necessary acidity and pore structure to stabilize the transition states and prevent side reactions like ring-opening hydrolysis, ensuring high selectivity towards the desired oxabicyclo intermediate.
The second step, the conversion of the oxabicyclo ether to the azabicyclo amine, is a challenging C-O bond cleavage and C-N bond formation reaction. This transformation is mediated by a supported metal oxide catalyst, where the active component R consists of rare earth oxides such as lanthanum, cerium, or ytterbium. These Lewis acidic sites on the catalyst surface play a pivotal role in activating the ether oxygen, making the adjacent carbon susceptible to nucleophilic attack by ammonia. The reaction proceeds via an SN2-like mechanism or a surface-mediated substitution where the ammonia molecule displaces the oxygen moiety. The patent highlights that catalysts like La2O3-ZSM-5 or CeO2-SiO2 are particularly effective, likely due to the optimal balance of acidity and basicity provided by the rare earth oxides. This specific catalytic environment suppresses the formation of elimination byproducts, such as the corresponding alkene (6,6-dimethyl-3-azabicyclo[3.1.0]hex-2-ene), which is a common impurity in high-temperature aminations. By fine-tuning the loading of the metal oxide (typically between 0.1wt% and 5wt%) and the activation temperature, the process achieves remarkable selectivity, ensuring that the final product stream requires minimal downstream purification, a key factor in maintaining high overall process efficiency.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
Implementing this synthesis requires careful attention to catalyst preparation and reaction parameters to maximize yield and purity. The process begins with the activation of the hydrogenation catalyst, typically involving heating under a hydrogen stream to reduce the metal precursor to its active metallic state. Following this, the caronic anhydride solution is pumped through the fixed bed at controlled space velocities to ensure sufficient contact time. The effluent is then separated, and the intermediate is fed into the second reactor containing the activated amination catalyst. Precise control of the ammonia-to-substrate ratio and temperature in the second stage is crucial to drive the equilibrium towards the amine while minimizing decomposition.
- Catalytic Hydrogenation: React caronic anhydride with hydrogen over a supported metal catalyst (e.g., Cu/SiO2 or Ni-Cu/SiO2) at 140-350°C and 0.1-8MPa to form 6,6-dimethyl-3-oxabicyclo[3.1.0]hexane.
- Catalytic Amination: React the intermediate oxabicyclo compound with ammonia gas over a supported metal oxide catalyst (e.g., La2O3-ZSM-5) at 180-550°C to yield the final amine product.
- Purification: Condense the reaction effluent, perform gas-liquid separation, and rectify to collect high-purity 6,6-dimethyl-3-azabicyclo[3.1.0]hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers profound strategic benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By eliminating the dependence on lithium aluminum hydride and borane reagents, manufacturers remove a significant logistical bottleneck. These traditional reductants are classified as hazardous materials, requiring specialized transport, storage, and handling procedures that inflate costs and introduce regulatory friction. Replacing them with hydrogen gas and ammonia, which are commodity chemicals with established global infrastructure, significantly de-risks the supply chain and stabilizes input costs. Furthermore, the continuous nature of the process allows for a "just-in-time" production model, reducing the need for large inventory buffers of dangerous intermediates and freeing up working capital.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, driven primarily by the elimination of expensive stoichiometric reagents and the reduction of waste disposal costs. In traditional batch processes, the cost of the reducing agent itself, combined with the extensive labor and energy required for the quenching and filtration of metal salts, constitutes a major portion of the COGS (Cost of Goods Sold). The new catalytic route replaces these consumables with reusable solid catalysts that have long lifespans. Additionally, the continuous flow design inherently offers better heat and mass transfer efficiency, leading to lower energy consumption per kilogram of product. The reduction in three-waste discharge—specifically the avoidance of aluminum/boron sludge and halogenated wastewater—translates directly into lower environmental compliance fees and wastewater treatment operational expenses, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the pharmaceutical sector, where interruptions can halt entire drug production lines. This catalytic process enhances reliability by simplifying the manufacturing workflow. The reduction in the number of unit operations—from multi-step batch sequences to a streamlined two-step continuous flow—minimizes the potential points of failure. Fewer steps mean fewer opportunities for human error, equipment malfunction, or quality deviations. Moreover, the use of robust heterogeneous catalysts allows for extended campaign runs without the need for frequent catalyst changes or reactor cleaning. This stability ensures a consistent, predictable output of high-purity intermediate, enabling downstream API manufacturers to plan their production schedules with greater confidence and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to commercial production is often fraught with challenges, particularly when dealing with exothermic reductions. The fixed-bed reactor configuration described in the patent is inherently scalable; increasing capacity often involves numbering up reactors or increasing bed volume rather than redesigning the entire process chemistry. This modularity facilitates rapid capacity expansion to meet market surges. From an environmental perspective, the process aligns perfectly with modern green chemistry principles. The primary byproduct is water, and the absence of heavy metal waste streams simplifies the path to regulatory approval in jurisdictions with stringent environmental laws. This "green" credential not only mitigates regulatory risk but also enhances the brand value of the final pharmaceutical product, appealing to increasingly eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic synthesis route. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this catalytic route over traditional reduction methods?
A: Unlike traditional methods requiring hazardous stoichiometric reductants like lithium aluminum hydride, this patent utilizes continuous catalytic hydrogenation and amination. This eliminates dangerous waste streams, reduces raw material costs significantly, and allows for safer, scalable industrial production.
Q: Which catalysts are optimal for the hydrogenation step?
A: The patent identifies supported metal catalysts containing Cu, Ni, Co, or Ru on carriers like SiO2, Al2O3, or zeolites as highly effective. Specifically, bimetallic systems like Ni-Cu/SiO2 have demonstrated superior conversion rates and selectivity for the intermediate oxabicyclo compound.
Q: How does this process impact environmental compliance?
A: By replacing batch reductions with continuous flow catalysis, the process drastically reduces three-waste discharge. The main byproduct is water, and the avoidance of halogenated intermediates and heavy metal reducing agents simplifies wastewater treatment and enhances overall environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising catalytic route described in CN115232059A can be seamlessly translated into a robust manufacturing asset. We understand the critical nature of pharmaceutical intermediates, which is why our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications. Whether you require custom catalyst development or full-scale toll manufacturing, our infrastructure is designed to deliver consistency and reliability, safeguarding your supply chain against the volatility of the global chemical market.
We invite you to explore how this advanced synthesis method can optimize your procurement strategy and reduce your overall manufacturing footprint. Our technical procurement team is ready to collaborate with you to evaluate the specific economic benefits for your operation. Please contact us to request a Customized Cost-Saving Analysis, where we will provide specific COA data and route feasibility assessments tailored to your volume requirements. Let us partner with you to secure a sustainable, high-quality supply of this vital antiviral intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
