Advancing Aryl Ketone Production: A Scalable Iron-Catalyzed Radical Coupling Strategy for Fine Chemicals
Advancing Aryl Ketone Production: A Scalable Iron-Catalyzed Radical Coupling Strategy for Fine Chemicals
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for sustainable, cost-effective, and scalable methodologies. Patent CN111056890B introduces a groundbreaking approach to synthesizing aryl ketone derivatives, a structural motif ubiquitous in pharmaceuticals and agrochemicals. This technology leverages an iron-catalyzed radical-radical cross-coupling reaction between aryl-substituted keto acids and aliphatic aldehydes. By utilizing a dual decarboxylation and decarbonylation strategy, this method circumvents the limitations of traditional stoichiometric organometallic routes. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this innovation represents a pivotal shift towards greener chemistry that does not compromise on yield or substrate scope. The ability to operate under air atmosphere with catalytic amounts of inexpensive iron salts marks a substantial departure from the rigorous inert conditions typically required for sensitive coupling reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl ketone scaffolds has relied heavily on Friedel-Crafts acylation or the addition of organometallic reagents to nitriles and amides. While effective in specific contexts, these legacy methods suffer from profound inefficiencies that hinder modern manufacturing. Friedel-Crafts reactions, for instance, demand large quantities of Lewis acid catalysts, generating massive amounts of acidic waste that complicates downstream processing and environmental compliance. Furthermore, these electrophilic aromatic substitutions are notoriously sluggish with electron-deficient arenes, limiting the diversity of accessible chemical space. Alternatively, routes employing Grignard or organolithium reagents require strictly anhydrous and anaerobic conditions, driving up operational costs and safety risks. These stoichiometric methods also produce equimolar amounts of metal salt byproducts, creating significant disposal burdens. For supply chain heads, the reliance on such resource-intensive processes translates to volatile pricing and extended lead times for high-purity intermediates.
The Novel Approach
The methodology disclosed in CN111056890B offers a transformative solution by employing a radical-radical coupling mechanism mediated by iron triacetylacetonate. This novel approach utilizes aryl keto acids and fatty aldehydes as readily available starting materials, bypassing the need for pre-functionalized halides or sensitive organometallic species. The reaction proceeds through a synergistic decarboxylation of the keto acid and decarbonylation of the aldehyde, facilitated by a composite initiator system comprising di-tert-butyl peroxide (DTBP) and potassium persulfate. Crucially, this process is robust enough to proceed under air, eliminating the need for expensive glovebox operations or rigorous nitrogen purging. The broad substrate tolerance allows for the incorporation of diverse functional groups, including halogens, ethers, and trifluoromethyl moieties, which are often incompatible with traditional strong base conditions. This versatility makes it an ideal candidate for cost reduction in fine chemical manufacturing, particularly for complex API intermediates.
Mechanistic Insights into Fe(acac)3-Catalyzed Radical Coupling
Understanding the mechanistic underpinnings of this transformation is essential for R&D teams aiming to optimize process parameters. The reaction initiates with the activation of the aryl keto acid by the composite radical initiator system. Under thermal conditions, the initiator generates radicals that abstract a hydrogen atom or interact with the carboxyl group, leading to the formation of a carboxyl radical intermediate. This unstable species rapidly undergoes decarboxylation to release carbon dioxide and generate a resonance-stabilized acyl radical. Simultaneously, the aliphatic aldehyde participates in a parallel activation pathway where it is converted into an acyl radical, which subsequently loses carbon monoxide to form an alkyl radical. The convergence of these two distinct radical species—the acyl radical derived from the keto acid and the alkyl radical from the aldehyde—results in the formation of the new carbon-carbon bond. This radical-radical coupling is highly exothermic and drives the reaction forward efficiently.
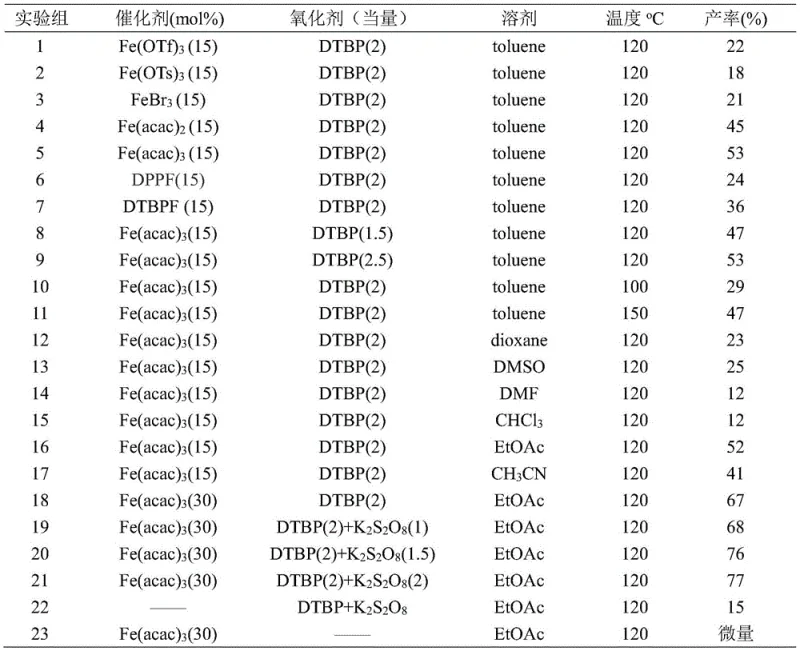
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic additions. Traditional organometallic additions to ketones or esters often suffer from over-addition, leading to tertiary alcohol byproducts that are difficult to separate. In contrast, the radical nature of this coupling ensures high chemoselectivity for the ketone product. The transient nature of the radical intermediates minimizes side reactions such as polymerization or self-coupling, provided the ratio of initiators and substrates is carefully controlled. The patent data indicates that optimizing the ratio of DTBP and potassium persulfate is critical; insufficient initiator leads to incomplete conversion, while excess can promote radical degradation pathways. Furthermore, the use of ethyl acetate as a solvent not only provides a green medium but also helps stabilize the transition states, contributing to the observed high yields across a wide array of substrates. This mechanistic clarity allows for precise tuning of reaction conditions to maximize purity specifications.
How to Synthesize Aryl Ketones Efficiently
Implementing this synthesis route requires attention to specific operational parameters to ensure reproducibility and safety. The standard protocol involves dissolving the aryl keto acid and aliphatic aldehyde in ethyl acetate, followed by the addition of the iron catalyst and the dual oxidant system. The mixture is then heated in a sealed vessel to facilitate the radical generation process. While the reaction is air-tolerant, maintaining a closed system prevents solvent loss and ensures consistent pressure during the thermal cycle. Post-reaction workup is straightforward, involving filtration through silica to remove metal residues followed by standard column chromatography.
- Combine aryl keto acid (0.2 mmol), Fe(acac)3 catalyst (0.06 mmol), and aliphatic aldehyde (1.0 mmol) in a reaction vessel.
- Add radical initiators DTBP (0.4 mmol) and K2S2O8 (0.3 mmol) along with ethyl acetate solvent (1 mL).
- Seal the vessel and stir at 120°C for 12 hours under air atmosphere, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this iron-catalyzed technology presents compelling economic and logistical benefits. The shift from stoichiometric precious metals or reactive organometallics to a catalytic iron system fundamentally alters the cost structure of aryl ketone production. Iron is abundant, non-toxic, and significantly cheaper than palladium or rhodium catalysts often used in cross-coupling. Moreover, the elimination of strict inert atmosphere requirements reduces capital expenditure on specialized reactor infrastructure and lowers energy consumption associated with nitrogen blanketing. This simplification of the process workflow directly contributes to substantial cost savings in manufacturing overheads. Additionally, the use of ethyl acetate, a common and environmentally benign solvent, simplifies waste stream management and reduces the regulatory burden associated with hazardous solvent disposal, further enhancing the overall sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency lies in the catalyst system. By utilizing only catalytic amounts of Fe(acac)3 rather than stoichiometric quantities of organolithium or Grignard reagents, the material costs are drastically reduced. Traditional methods generate large volumes of inorganic salt waste that require costly treatment; this radical method produces gaseous byproducts (CO2 and CO) that vent easily, minimizing solid waste disposal fees. The high atom economy of the decarboxylative coupling means less raw material is wasted, improving the overall yield per kilogram of input. Furthermore, the mild reaction conditions reduce energy demands for heating and cooling cycles, contributing to a lower carbon footprint and reduced utility costs for the manufacturing facility.
- Enhanced Supply Chain Reliability: Sourcing high-purity organometallic reagents can be a bottleneck due to their sensitivity and limited shelf life. In contrast, the starting materials for this method—aryl keto acids and aliphatic aldehydes—are commodity chemicals with stable supply chains and long shelf lives. This stability reduces the risk of production delays caused by reagent degradation or shipping restrictions on hazardous materials. The robustness of the reaction under air conditions also means that manufacturing can proceed with greater flexibility, reducing the dependency on specialized personnel and equipment. This reliability ensures consistent delivery schedules for downstream customers, mitigating the risks associated with supply chain disruptions common in the fine chemical sector.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this method, with successful gram-scale reactions maintaining high yields. This proof of concept suggests a smooth path to kilogram and ton-scale production without the need for extensive re-optimization. The environmental profile is equally impressive; the avoidance of heavy metal catalysts simplifies the purification process and ensures the final product meets stringent residual metal specifications required for pharmaceutical applications. The use of green solvents and the generation of minimal hazardous waste align with increasingly strict global environmental regulations, future-proofing the manufacturing process against tightening compliance standards and potential carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed radical coupling technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: What are the advantages of this iron-catalyzed method over traditional Friedel-Crafts acylation?
A: Unlike Friedel-Crafts reactions which require stoichiometric Lewis acids and struggle with electron-deficient aromatics, this method uses catalytic iron, operates under air, and tolerates a wide range of functional groups including halogens and trifluoromethyl groups without generating excessive inorganic waste.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the patent demonstrates successful gram-scale synthesis with maintained yields (e.g., 65% yield on 10 mmol scale), indicating robust scalability. The use of inexpensive iron catalysts and common solvents like ethyl acetate further supports cost-effective commercial production.
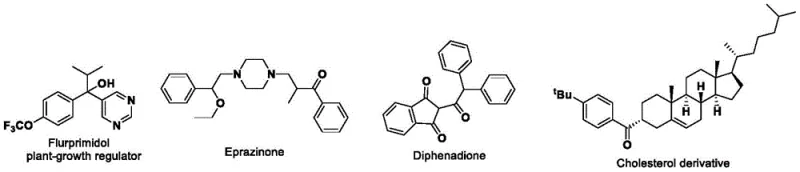
Q: Can this method be applied to synthesize complex agrochemical regulators like Flurprimidol?
A: Absolutely. The patent explicitly details the synthesis of a key intermediate for Flurprimidol, a plant-growth regulator. The method successfully constructs the aryl ketone backbone required for subsequent lithiation and coupling steps, proving its utility in agrochemical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed radical coupling method described in CN111056890B for the production of high-value aryl ketones. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific thermal and pressure requirements of radical reactions, while our rigorous QC labs guarantee that every batch meets stringent purity specifications. We understand that consistency is key for your downstream synthesis, and our commitment to quality assurance ensures that the aryl ketone intermediates we supply are free from problematic impurities that could affect your final API or agrochemical product.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Whether you require custom synthesis of complex aryl ketone derivatives or scale-up of the Flurprimidol intermediate pathway, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes both economic efficiency and scientific excellence, positioning your organization for success in a competitive global market.
