Advanced Ruthenium Catalysis for Scalable Aryl Ketone Manufacturing
Introduction to Next-Generation Aryl Ketone Synthesis
The synthesis of aryl ketones represents a cornerstone operation in the production of high-value pharmaceutical intermediates and fine chemicals. Historically, this transformation has relied heavily on classical Friedel-Crafts acylation or transition metal-catalyzed couplings using precious metals like Rhodium and Palladium. However, patent CN102153434A introduces a paradigm shift by detailing a highly efficient method utilizing Ruthenium catalysts to couple aldehydes with arylboronic acids. This innovation addresses critical industry pain points regarding catalyst cost, toxicity, and operational complexity. By leveraging relatively inexpensive Ruthenium complexes such as dichloro(p-cymene)ruthenium(II) dimer or tricarbonyl ruthenium dichloride dimer, this technology enables the preparation of diverse aryl ketones under mild conditions (95-100°C) with excellent yields. For procurement and R&D teams, this represents a strategic opportunity to optimize supply chains for key building blocks used in drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for aryl ketones, particularly those employing Rhodium, Palladium, or Platinum catalysts, present substantial economic and environmental burdens. As highlighted in the background art of the patent, methods utilizing Rhodium complexes (e.g., [Rh(CH2CH2)2Cl]2) are plagued by the exorbitant cost of the metal and its high toxicity, which necessitates rigorous and expensive purification steps to meet residual metal specifications for pharmaceutical applications. Furthermore, these legacy systems often exhibit narrow substrate scopes, frequently limited to aromatic aldehydes, and suffer from poor atom economy when using precursors like aryltrifluoroborates. Similarly, Palladium-catalyzed systems, while effective, introduce significant cost volatility due to the fluctuating market price of Pd and often require harsh conditions or specialized ligands that complicate scale-up. The reliance on stoichiometric Lewis acids in Friedel-Crafts reactions generates massive amounts of corrosive waste and suffers from poor regioselectivity, making these methods increasingly untenable in a modern green chemistry context.
The Novel Approach
The methodology disclosed in CN102153434A offers a robust alternative by substituting expensive noble metals with more abundant and cost-effective Ruthenium catalysts. This novel approach operates under inert atmospheres using readily available aldehydes and arylboronic acids as substrates, paired with organic phosphine ligands and potassium phosphate as a base. A key innovation is the use of additives like pinacolone or acetone, which appear to facilitate the catalytic cycle without introducing excessive complexity. The reaction proceeds smoothly in a toluene and water solvent system at moderate temperatures of 95-100°C, eliminating the need for extreme thermal inputs or microwave irradiation often required by older protocols.  This specific transformation exemplifies the protocol's efficiency, converting 4-fluorobenzaldehyde and 4-methoxyphenylboronic acid into the corresponding ketone with high fidelity. By avoiding the use of equivalent amounts of catalyst and enabling simpler workup procedures via silica gel adsorption or recrystallization, this route drastically simplifies the manufacturing process while maintaining high purity standards essential for regulatory compliance.
This specific transformation exemplifies the protocol's efficiency, converting 4-fluorobenzaldehyde and 4-methoxyphenylboronic acid into the corresponding ketone with high fidelity. By avoiding the use of equivalent amounts of catalyst and enabling simpler workup procedures via silica gel adsorption or recrystallization, this route drastically simplifies the manufacturing process while maintaining high purity standards essential for regulatory compliance.
Mechanistic Insights into Ruthenium-Catalyzed Carbonylative Coupling
The core of this technology lies in the unique ability of Ruthenium complexes to activate the C-H bond of the aldehyde and facilitate its coupling with the arylboronic acid. The catalytic cycle likely involves the oxidative addition of the aldehyde C-H bond to the low-valent Ruthenium center, followed by transmetallation with the arylboronic species activated by the phosphate base. The presence of organic phosphine ligands, such as tri-tert-butylphosphine tetrafluoroborate or tricyclohexylphosphine, plays a critical role in stabilizing the active catalytic species and modulating the electronic environment of the metal center to favor reductive elimination of the ketone product. This ligand acceleration effect is crucial for achieving the reported high turnover numbers and broad substrate compatibility. The inclusion of water in the toluene solvent system further aids in the activation of the boronic acid, promoting the formation of the reactive boronate species necessary for efficient transmetallation.
From an impurity control perspective, the mechanism inherently minimizes side reactions common in radical-based or strong acid-mediated processes. The mild basic conditions (using K3PO4) prevent the degradation of acid-sensitive functional groups, while the specific coordination geometry of the Ruthenium-phosphine complex ensures high chemoselectivity. 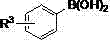 As illustrated by the general structure of the arylboronic acid substrates
As illustrated by the general structure of the arylboronic acid substrates 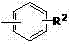 and the aldehyde components, the system tolerates a wide array of substituents including halogens, electron-withdrawing groups like nitro and cyano, and electron-donating groups like methoxy. This tolerance is mechanistically attributed to the non-radical nature of the Ru-catalyzed pathway, which avoids the indiscriminate reactivity often seen with other transition metals. Consequently, the impurity profile is significantly cleaner, reducing the burden on downstream purification units and increasing the overall yield of the desired high-purity aryl ketone.
and the aldehyde components, the system tolerates a wide array of substituents including halogens, electron-withdrawing groups like nitro and cyano, and electron-donating groups like methoxy. This tolerance is mechanistically attributed to the non-radical nature of the Ru-catalyzed pathway, which avoids the indiscriminate reactivity often seen with other transition metals. Consequently, the impurity profile is significantly cleaner, reducing the burden on downstream purification units and increasing the overall yield of the desired high-purity aryl ketone.
How to Synthesize Aryl Ketones Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the catalyst and ligands to maximize efficiency. The patent specifies a molar ratio of aldehyde to arylboronic acid preferably at 1:2, ensuring that the boronic acid is in excess to drive the equilibrium towards product formation. Catalyst loading is kept low, typically around 2.5 mol% relative to the aldehyde, while the ligand is used at approximately 10 mol%. The reaction is conducted in a mixture of toluene and water with an additive such as pinacolone (1.8-2.2 equivalents). Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are critical for maintaining reproducibility and safety.
- Charge a reaction vessel with a ruthenium catalyst (e.g., [Ru(cymene)Cl2]2), organic phosphine ligand, aldehyde substrate, arylboronic acid, potassium phosphate base, and an additive like pinacolone in a toluene/water solvent system.
- Maintain the reaction mixture under an inert atmosphere and heat to a temperature range of 95-100°C for a duration of 10 to 24 hours to ensure complete conversion.
- Upon completion, remove the solvent via vacuum spin-drying with silica gel adsorption, followed by purification through simple column chromatography or recrystallization to isolate the high-purity aryl ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Ruthenium-catalyzed protocol offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By replacing Rhodium or Palladium with Ruthenium, manufacturers can insulate their production costs from the volatile pricing of precious metals, leading to significant long-term savings. Furthermore, the lower toxicity profile of Ruthenium compared to Rhodium simplifies environmental compliance and reduces the costs associated with hazardous waste disposal and worker safety protocols. The use of commercially available starting materials like substituted benzaldehydes and phenylboronic acids ensures a stable and reliable supply chain, mitigating the risk of bottlenecks often caused by exotic or custom-synthesized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts like Rhodium directly translates to lower Bill of Materials (BOM) costs. Additionally, the simplified workup procedure, which avoids complex aqueous extractions and allows for purification via simple column chromatography or recrystallization, reduces solvent consumption and labor hours. The ability to use catalytic amounts of Ruthenium rather than stoichiometric reagents further enhances atom economy, minimizing waste generation and disposal fees. These factors combined result in a substantially more cost-effective manufacturing process suitable for high-volume production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, potassium phosphate, and widely available arylboronic acids ensures that the supply chain is robust and resilient. Unlike processes dependent on specialized ligands or air-sensitive organometallic reagents that require cold chain logistics, the reagents for this method are stable and easily sourced from multiple global suppliers. This diversification of supply sources reduces lead times and protects against single-source disruptions, ensuring continuous production capability for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions (95-100°C) are well within the operational limits of standard glass-lined or stainless steel reactors, facilitating easy scale-up from pilot plant to commercial tonnage without the need for specialized high-pressure or cryogenic equipment. The aqueous-organic solvent system is easier to manage and recycle compared to pure organic or hazardous solvent systems. Moreover, the reduced toxicity of the catalyst aligns with increasingly stringent environmental regulations, future-proofing the manufacturing process against tighter emission and effluent standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ruthenium-catalyzed synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for process engineers and quality assurance teams evaluating the technology for adoption.
Q: Why is the Ruthenium-catalyzed method superior to Rhodium or Palladium systems for aryl ketone synthesis?
A: Traditional methods utilizing Rhodium or Palladium catalysts suffer from prohibitively high costs, significant toxicity, and often require harsh reaction conditions. The Ruthenium-based approach described in patent CN102153434A utilizes commercially available, lower-toxicity catalysts that operate under milder conditions, significantly reducing raw material costs and simplifying downstream heavy metal removal processes.
Q: What is the substrate scope of this Ruthenium-catalyzed coupling reaction?
A: This methodology demonstrates exceptional functional group tolerance. It is effective not only for standard aromatic aldehydes but also accommodates aliphatic aldehydes, heterocyclic aldehydes (such as furan and thiophene derivatives), and substrates containing sensitive groups like halogens, nitro, cyano, and trifluoromethyl moieties, making it highly versatile for complex intermediate synthesis.
Q: How does the purification process compare to conventional Friedel-Crafts acylation?
A: Unlike Friedel-Crafts reactions which often generate large amounts of waste and require difficult regioselective separations, this catalytic method produces cleaner reaction profiles. The workup typically involves straightforward silica gel adsorption followed by simple column chromatography or recrystallization, avoiding the complex aqueous workups and hazardous waste streams associated with stoichiometric Lewis acid methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Ruthenium-catalyzed aryl ketone synthesis described in CN102153434A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that consistency and quality are paramount, and our team is dedicated to optimizing this specific catalytic route to deliver high-purity aryl ketones that exceed industry standards.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this Ruthenium-based method can enhance your supply chain efficiency and reduce your overall manufacturing costs. Let us be your partner in driving innovation and value in the fine chemical sector.
