Advanced Photocatalytic Manufacturing of High-Purity Benzoxazine Derivatives for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to construct complex heterocyclic scaffolds, particularly those containing nitrile functionalities which serve as versatile precursors for amines, acids, and amides. A significant breakthrough in this domain is documented in Chinese Patent CN110183394B, which discloses a novel method for preparing 3-nitrile ethyl-2-hydrocarbyl-4H-benzoxazine derivatives. This technology leverages visible light photocatalysis to achieve alkenyl bifunctionalization, offering a transformative approach to synthesizing these valuable intermediates. By utilizing bromoacetonitrile as a cyanomethyl source under blue LED irradiation, the process circumvents the severe safety hazards associated with traditional cyanation reagents. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more cost-effective manufacturing protocols that align with modern green chemistry principles while maintaining high synthetic efficiency and product purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of cyano groups into organic molecules has relied heavily on nucleophilic substitution reactions using alkali metal cyanides such as potassium cyanide or sodium cyanide. While effective, these reagents are acutely toxic, posing severe risks to personnel safety and requiring specialized infrastructure for handling and waste treatment. Furthermore, transition metal-catalyzed cyanation methods, often employing copper salts, typically necessitate stoichiometric amounts of oxidants like peroxides to drive the reaction cycle. These harsh oxidative conditions can lead to poor functional group tolerance, resulting in complex impurity profiles that are difficult and expensive to remove during downstream purification. The reliance on heavy metals also introduces the risk of residual metal contamination in the final API, necessitating costly additional purification steps to meet stringent regulatory limits for pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology described in patent CN110183394B utilizes a photoredox catalytic system that operates under exceptionally mild conditions. The core innovation lies in the use of bromoacetonitrile as a safe, liquid cyanomethyl radical precursor, activated by a tris(2-phenylpyridine)iridium catalyst under blue LED light. This approach completely eliminates the need for toxic cyanide salts and stoichiometric oxidants. The reaction proceeds at room temperature in acetonitrile solvent, demonstrating excellent compatibility with a wide range of functional groups including halogens and electron-donating substituents. This not only simplifies the operational procedure but also significantly enhances the safety profile of the manufacturing process. The general reaction scheme is illustrated below, highlighting the direct conversion of N-[2-isoenylaryl]amides to the target benzoxazine structures.
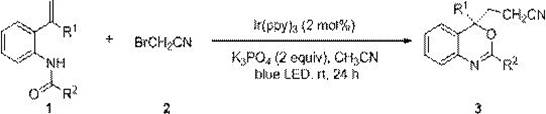
Mechanistic Insights into Ir(ppy)3-Catalyzed Photoredox Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay of single-electron transfer (SET) processes mediated by the excited state of the iridium photocatalyst. Upon irradiation with blue light, the Ir(ppy)3 catalyst reaches an excited state capable of reducing bromoacetonitrile via SET, generating a reactive cyanomethyl radical and a bromide anion. This carbon-centered radical then undergoes a regioselective addition to the electron-rich alkene moiety of the N-[2-isoenylaryl]amide substrate. The resulting benzylic radical intermediate is subsequently oxidized by the oxidized iridium species to form a carbocation, which triggers an intramolecular nucleophilic attack by the amide oxygen atom. This cascade sequence efficiently constructs the benzoxazine ring system with high stereochemical control. The mildness of the radical mechanism ensures that sensitive functional groups on the aromatic rings remain intact, thereby minimizing the formation of side products and simplifying the overall impurity profile of the crude reaction mixture.
From a quality control perspective, the absence of strong oxidants and heavy metal stoichiometric reagents drastically reduces the burden on purification teams. Traditional copper-catalyzed routes often leave behind trace metals that require scavenging resins or complex crystallization protocols to remove. In this photocatalytic system, the catalyst loading is low (2 mol%), and the iridium complex can potentially be recovered or managed more easily than bulk copper salts. Furthermore, the high selectivity of the radical addition step means that fewer regioisomers are formed. This inherent cleanliness of the reaction translates directly into higher isolated yields and reduced solvent consumption during column chromatography or recrystallization, which is a critical factor for both economic efficiency and environmental compliance in large-scale chemical manufacturing.
How to Synthesize 3-Nitrile Ethyl-2-Hydrocarbyl-4H-Benzoxazine Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it highly suitable for both laboratory optimization and pilot plant scaling. The protocol utilizes standard Schlenk techniques to maintain an inert nitrogen atmosphere, ensuring that the radical intermediates are not quenched by oxygen. The reaction parameters are straightforward, requiring only a standard blue LED light source and common laboratory solvents. Detailed standardized operating procedures regarding exact stoichiometry, mixing rates, and specific workup parameters are essential for consistent results. For a comprehensive breakdown of the experimental steps required to achieve the high yields reported in the patent data, please refer to the technical guide below.
- Charge a dehydrated Schlenk tube with N-[2-isoenylaryl]amide, bromoacetonitrile, inorganic base (K3PO4), and Ir(ppy)3 catalyst under nitrogen atmosphere.
- Add acetonitrile solvent and irradiate the mixture with 16 W blue LED light at room temperature for 24 hours with continuous stirring.
- Quench the reaction with saturated brine, extract the organic phase with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible strategic benefits beyond mere chemical novelty. The shift away from hazardous cyanide salts mitigates significant regulatory and liability risks associated with the storage and transport of toxic raw materials. This inherently safer design reduces insurance premiums and simplifies compliance with increasingly strict environmental health and safety (EHS) regulations. Moreover, the elimination of stoichiometric oxidants and copper salts reduces the volume of hazardous waste generated per kilogram of product, leading to substantial cost savings in waste disposal and treatment. The simplicity of the workup procedure, involving basic extraction and chromatography, shortens the production cycle time, allowing for faster turnaround on custom synthesis orders and improved responsiveness to market demands.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of catalytic rather than stoichiometric reagents. By replacing expensive and wasteful copper salts and peroxides with a low-loading iridium catalyst and benign bromoacetonitrile, the raw material cost per unit is significantly optimized. Additionally, the high selectivity of the reaction reduces the loss of valuable starting materials to side reactions, improving the overall atom economy. The mild reaction conditions also imply lower energy consumption for heating or cooling compared to traditional thermal processes, contributing to a lower carbon footprint and reduced utility costs for the manufacturing facility.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including N-[2-isoenylaryl]amides and bromoacetonitrile, are commercially available and stable, ensuring a robust supply chain free from the volatility often associated with specialized toxic reagents. The robustness of the photocatalytic system against varying substrate electronic properties means that a single platform technology can be used to manufacture a diverse library of benzoxazine derivatives. This flexibility allows suppliers to quickly adapt to changing customer requirements without needing to requalify entirely new synthetic routes, thereby ensuring continuity of supply for critical pharmaceutical intermediates even during market fluctuations.
- Scalability and Environmental Compliance: Photocatalytic reactions are uniquely suited for continuous flow processing, a technology that is rapidly becoming the standard for scalable fine chemical manufacturing. The use of LED light sources allows for precise control over photon flux and heat management, facilitating safe scale-up from gram to ton quantities. The absence of toxic cyanide waste streams simplifies the environmental permitting process for new production lines. This alignment with green chemistry principles not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand value of the end-product by appealing to sustainability-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, scalability, and substrate compatibility based on the detailed experimental data provided in the patent literature. These insights are derived directly from the reported examples and comparative studies, offering a realistic view of the technology's capabilities and limitations in a commercial setting.
Q: What are the safety advantages of this photocatalytic cyanation method compared to traditional methods?
A: Unlike traditional methods that rely on highly toxic alkali metal cyanides (like KCN or NaCN), this protocol utilizes bromoacetonitrile as a safer cyanomethyl source. Additionally, it eliminates the need for stoichiometric amounts of copper salts and hazardous peroxides, significantly reducing workplace toxicity risks and waste disposal costs.
Q: Can this reaction be scaled up for industrial production of pharmaceutical intermediates?
A: Yes, the reaction operates under mild conditions (room temperature, atmospheric pressure) using visible light, which is highly amenable to scale-up using flow chemistry reactors. The use of commercially available catalysts and simple workup procedures further supports robust commercial manufacturing.
Q: What is the substrate scope for the N-[2-isoenylaryl]amide starting materials?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituents on the aromatic ring, including electron-donating groups (methyl, methoxy) and electron-withdrawing groups (fluoro, chloro, bromo, trifluoromethyl), as well as heterocyclic and naphthyl variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitrile Ethyl-2-Hydrocarbyl-4H-Benzoxazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in modern drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110183394B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of benzoxazine intermediate meets the highest international standards for pharmaceutical applications. We are committed to delivering high-purity compounds that accelerate your pipeline while adhering to the strictest safety and quality protocols.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your existing supply chain or require specific COA data and route feasibility assessments for new targets, our experts are ready to assist. Contact our technical procurement team today to discuss how we can support your journey from benchtop discovery to commercial success with reliable, high-quality chemical solutions.
