Advanced Photocatalytic Manufacturing of High-Purity Benzoxazine Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high efficiency and minimal environmental impact. A significant breakthrough in this domain is documented in Chinese Patent CN110183394B, which discloses a novel method for preparing 3-nitrile ethyl-2-hydrocarbyl-4H-benzoxazine derivatives. This technology leverages visible light photocatalysis to achieve alkenyl bifunctionalization, a critical transformation in the synthesis of bioactive molecules. By utilizing bromoacetonitrile as a cyanomethyl source under blue LED irradiation, this process circumvents the severe safety hazards associated with traditional cyanation reagents. For R&D directors and procurement specialists, this patent represents a paradigm shift towards safer, more sustainable manufacturing of high-purity pharmaceutical intermediates. The methodology not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis, making it an attractive candidate for commercial adoption in the production of advanced drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of cyano groups into organic frameworks has relied heavily on nucleophilic substitution reactions involving alkali metal cyanides such as potassium cyanide or sodium cyanide. While effective, these reagents are notoriously toxic, posing severe risks to personnel safety and requiring specialized infrastructure for waste treatment and disposal. Furthermore, transition metal-catalyzed cyanation methods, although powerful, often necessitate the use of stoichiometric amounts of copper salts or expensive palladium catalysts, which can lead to significant metal contamination in the final product. In the specific context of benzoxazine synthesis, prior art methods frequently require harsh oxidative conditions or the use of equivalent amounts of peroxides to drive the cyclization. These aggressive conditions can degrade sensitive functional groups, limit substrate scope, and generate complex impurity profiles that are difficult to remove during downstream purification, ultimately increasing the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN110183394B introduces a mild, photocatalytic strategy that operates under ambient conditions. The core innovation lies in the use of bromoacetonitrile as a safe, liquid cyanomethyl radical precursor, activated by a visible light-sensitive iridium catalyst. This approach eliminates the need for toxic solid cyanides and avoids the use of stoichiometric oxidants or copper promoters. The reaction proceeds smoothly at room temperature (25°C) using simple blue LED strips, providing a gentle yet effective driving force for the radical addition and subsequent cyclization. This technological advancement allows for the construction of the 3-nitrile ethyl-2-hydrocarbyl-4H-benzoxazine core with high selectivity and yield. By shifting from thermal, hazardous chemistry to photochemical, catalytic processes, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing while adhering to stricter environmental and safety regulations.
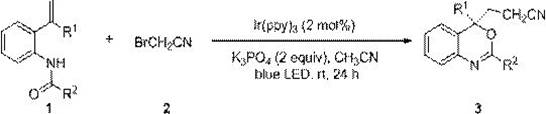
Mechanistic Insights into Ir(ppy)3-Catalyzed Alkenyl Bifunctionalization
The mechanistic pathway of this transformation is rooted in the principles of photoredox catalysis, specifically utilizing tris(2-phenylpyridine)iridium (Ir(ppy)3) as the photosensitizer. Upon irradiation with blue light, the ground state iridium complex absorbs photons to reach an excited state, which possesses sufficient redox potential to interact with the bromoacetonitrile substrate. This interaction facilitates the homolytic cleavage of the carbon-bromine bond, generating a reactive cyanomethyl radical and an iridium species. This radical species then undergoes a regioselective addition to the electron-rich alkene moiety of the N-[2-isoenylaryl]amide substrate. The resulting carbon-centered radical intermediate is subsequently oxidized and undergoes an intramolecular nucleophilic attack by the amide oxygen, closing the ring to form the benzoxazine scaffold. This elegant cascade occurs without the need for external oxidants, as the catalytic cycle is self-sustaining through the regeneration of the photocatalyst, ensuring high turnover numbers and minimal catalyst consumption throughout the reaction duration.
From an impurity control perspective, the mildness of the photocatalytic conditions is paramount. Traditional thermal radical reactions often suffer from non-selective radical propagation, leading to polymerization of the alkene starting material or over-functionalization of the product. However, the controlled generation of radicals via light irradiation ensures that the concentration of reactive species remains low and steady, favoring the desired intermolecular addition over competing side reactions. Furthermore, the absence of strong acids, bases, or heavy metal oxidants prevents the degradation of acid- or base-sensitive functional groups on the aromatic rings. This results in a cleaner crude reaction profile, which significantly simplifies the downstream purification process. For quality control teams, this means fewer unidentified impurities in the final API intermediate, reducing the analytical burden and ensuring consistent batch-to-batch quality that meets stringent regulatory specifications for pharmaceutical applications.
How to Synthesize 3-Nitrile Ethyl-2-Hydrocarbyl-4H-Benzoxazine Efficiently
The practical implementation of this synthesis is designed for ease of operation, utilizing standard laboratory glassware and commercially available reagents. The protocol involves charging a Schlenk tube with the amide substrate, bromoacetonitrile, an inorganic base such as potassium phosphate or potassium carbonate, and the iridium catalyst. The system is purged with nitrogen to maintain an inert atmosphere, which is critical for preventing the quenching of the excited photocatalyst by oxygen. Acetonitrile serves as the solvent, providing excellent solubility for all components. The reaction vessel is then placed under 16 W blue LED irradiation and stirred at room temperature for approximately 24 hours. Following the reaction, a simple aqueous workup involving saturation brine quenching and ethyl acetate extraction isolates the organic product. The detailed standardized synthesis steps see the guide below.
- Charge a dehydrated Schlenk tube with N-[2-isoenylaryl]amide, bromoacetonitrile, inorganic base (K3PO4 or K2CO3), and Ir(ppy)3 catalyst under nitrogen atmosphere.
- Add acetonitrile solvent and irradiate the mixture with 16 W blue LED light at room temperature (25°C) while stirring for 24 hours.
- Quench the reaction with saturated brine or ammonium chloride, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers compelling economic and logistical benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain. By replacing hazardous solid cyanides with liquid bromoacetonitrile, companies can eliminate the high costs associated with specialized storage, handling protocols, and hazardous waste disposal fees. Additionally, the catalytic nature of the iridium complex, used at only 2 mol%, contrasts sharply with stoichiometric metal methods, leading to significant savings on precious metal costs. The mild reaction conditions also translate to lower energy consumption, as no heating or cooling beyond ambient temperature is required, further reducing the operational expenditure per kilogram of product manufactured. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of stoichiometric copper salts and toxic cyanide reagents removes the need for expensive metal scavenging resins and complex wastewater treatment processes typically required to meet environmental discharge limits. This streamlining of the post-reaction processing directly lowers the variable costs associated with production. Furthermore, the high yields reported in the patent examples indicate efficient conversion of starting materials, minimizing raw material waste and maximizing the output per batch. The use of common solvents like acetonitrile and bases like potassium phosphate ensures that input costs remain stable and predictable, shielding the manufacturing process from volatility in specialty reagent pricing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including N-[2-isoenylaryl]amides and bromoacetonitrile, are commodity chemicals with robust global supply chains. Unlike specialized organometallic reagents that may have long lead times or single-source dependencies, these inputs are readily available from multiple vendors. This diversity in sourcing options mitigates the risk of supply disruptions, ensuring continuous production schedules. Moreover, the operational simplicity of the reaction—requiring only standard LED lights and nitrogen lines—means that it can be easily transferred between different manufacturing sites or scaled up in existing facilities without the need for capital-intensive reactor modifications or specialized high-pressure equipment.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but modern flow chemistry and large-surface-area LED reactors have effectively solved this bottleneck. The room temperature operation of this process makes it inherently safer for large-scale production, removing the risks associated with thermal runaways common in exothermic cyanation reactions. From an environmental standpoint, the avoidance of heavy metal waste and toxic cyanide byproducts aligns perfectly with green chemistry principles and increasingly strict global environmental regulations. This compliance reduces the regulatory burden on the manufacturing site and enhances the corporate sustainability profile, which is a key metric for many downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic benzoxazine synthesis. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for process chemists and project managers evaluating the feasibility of integrating this route into their current manufacturing portfolios. The answers provided reflect the specific advantages of the blue light-mediated protocol over conventional thermal methods.
Q: How does this photocatalytic method improve safety compared to traditional cyanation?
A: Traditional methods often rely on toxic alkali metal cyanides or stoichiometric copper salts. This patent utilizes bromoacetonitrile as a safer cyanomethyl source under mild visible light conditions, eliminating the need for hazardous cyanide handling and heavy metal waste disposal.
Q: What is the catalyst loading required for this benzoxazine synthesis?
A: The process employs a highly efficient iridium photocatalyst, specifically tris(2-phenylpyridine)iridium (Ir(ppy)3), at a low loading of approximately 2 mol%, which significantly reduces catalyst costs compared to stoichiometric metal promoters.
Q: Is this reaction scalable for industrial pharmaceutical intermediate production?
A: Yes, the reaction operates at room temperature (25°C) using standard blue LED irradiation and common solvents like acetonitrile. The absence of extreme temperatures or pressures makes it highly suitable for commercial scale-up in flow chemistry or large batch reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitrile Ethyl-2-Hydrocarbyl-4H-Benzoxazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modern drug synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110183394B can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that the transition to new synthetic routes requires confidence in both technical execution and supply continuity, and our team is dedicated to providing that assurance through meticulous process optimization and validation.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the viability of this method for your supply chain. Contact us today to discuss how we can support your development of high-purity pharmaceutical intermediates with efficiency, safety, and economic precision.
