Advanced Photocatalytic Synthesis of Benzoxazine Derivatives for Commercial Scale-Up
Advanced Photocatalytic Synthesis of Benzoxazine Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. A significant breakthrough in this domain is detailed in Chinese Patent CN110183394B, which discloses a novel method for preparing 3-nitrile ethyl-2-hydrocarbyl-4H-benzoxazine derivatives. This technology leverages visible light photoredox catalysis to achieve alkenyl bifunctionalization under remarkably mild conditions. For R&D directors and procurement specialists, this patent represents a pivotal shift away from hazardous traditional cyanation methods towards safer, more sustainable synthetic routes. The ability to construct these valuable benzoxazine cores using bromoacetonitrile as a cyanomethyl source, without the need for toxic metal cyanides or stoichiometric oxidants, addresses critical pain points in modern API intermediate manufacturing. This report analyzes the technical merits and commercial viability of this innovation, highlighting its potential for cost reduction and supply chain optimization in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyano-containing compounds has relied heavily on nucleophilic substitution reactions involving alkali metal cyanide salts such as potassium cyanide or sodium cyanide. While effective, these reagents pose severe safety hazards due to their acute toxicity, requiring specialized containment facilities and rigorous waste treatment protocols that drastically inflate operational costs. Furthermore, transition metal-catalyzed cyanation methods, such as those reported by Buchwald involving copper catalysts, often necessitate the use of stoichiometric amounts of oxidants and equivalent copper salts. These traditional approaches not only generate substantial heavy metal waste but also complicate the downstream purification process, as removing trace copper residues to meet stringent pharmaceutical purity specifications can be technically challenging and expensive. The reliance on harsh reaction conditions and hazardous reagents in conventional pathways creates significant bottlenecks for scaling up production safely and economically.
The Novel Approach
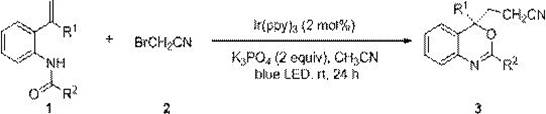
In stark contrast, the methodology described in patent CN110183394B introduces a transformative strategy utilizing visible light photoredox catalysis. By employing bromoacetonitrile as a safe and stable cyanomethyl radical precursor, this method completely circumvents the use of toxic inorganic cyanides. The reaction proceeds under blue LED irradiation at room temperature, utilizing a catalytic amount of tris(2-phenylpyridine)iridium, which eliminates the need for stoichiometric oxidants and copper salts entirely. This paradigm shift simplifies the reaction profile significantly, allowing for the direct construction of 3-nitrile ethyl-2-hydrocarbyl-4H-benzoxazine skeletons from N-[2-isoenylaryl]amides. The operational simplicity, combined with the high atom economy and mild conditions, makes this novel approach exceptionally attractive for industrial applications where safety and environmental compliance are paramount concerns for supply chain continuity.
Mechanistic Insights into Ir(ppy)3-Catalyzed Photoredox Cyclization
The core of this technological advancement lies in the efficient generation of cyanomethyl radicals via a photoredox cycle. Upon irradiation with blue LED light, the iridium catalyst, specifically Ir(ppy)3, enters an excited state capable of engaging in single-electron transfer processes. This excited species facilitates the homolytic cleavage of the carbon-bromine bond in bromoacetonitrile, generating the reactive cyanomethyl radical species essential for the transformation. This radical then undergoes addition to the electron-rich alkene moiety of the N-[2-isoenylaryl]amide substrate. The resulting carbon-centered radical intermediate subsequently participates in an intramolecular cyclization with the amide oxygen, followed by oxidation and deprotonation steps to restore aromaticity and finalize the benzoxazine ring structure. This mechanistic pathway is highly selective, minimizing the formation of side products that typically plague radical reactions, thereby ensuring a cleaner reaction profile.
From an impurity control perspective, the absence of stoichiometric metal oxidants is a critical advantage. Traditional radical cyclizations often suffer from over-oxidation or non-selective radical coupling, leading to complex impurity profiles that are difficult to separate. In this photocatalytic system, the redox potentials are finely tuned by the iridium catalyst, ensuring that the radical intermediates are generated and consumed in a controlled manner. The use of mild inorganic bases like potassium phosphate further buffers the reaction environment, preventing acid-catalyzed degradation of the sensitive nitrile or amide functionalities. For quality assurance teams, this translates to a much simpler impurity spectrum, facilitating easier purification via standard column chromatography or crystallization. The high yields reported, such as the 78% isolated yield for the model substrate, underscore the robustness of this catalytic cycle in maintaining product integrity throughout the synthesis.
How to Synthesize 3-Nitrile Ethyl-2-Hydrocarbyl-4H-Benzoxazine Efficiently
Implementing this synthesis requires careful attention to the photochemical setup and reagent quality to ensure reproducibility. The process begins with the preparation of the reaction vessel, typically a Schlenk tube, which must be thoroughly dried and degassed to maintain an inert nitrogen atmosphere, as oxygen can quench the excited state of the photocatalyst. The precise stoichiometry involves mixing the N-[2-isoenylaryl]amide substrate with two equivalents of bromoacetonitrile and two equivalents of an inorganic base such as potassium phosphate. The catalyst loading is remarkably low at only 2 mol% of Ir(ppy)3, dissolved in acetonitrile as the solvent. Once assembled, the reaction mixture is subjected to continuous stirring under 16 W blue LED irradiation at ambient temperature for approximately 24 hours. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding N-[2-isoenylaryl]amide, bromoacetonitrile, inorganic base (K3PO4), and Ir(ppy)3 catalyst in acetonitrile under nitrogen.
- Irradiate the reaction vessel with 16 W blue LED light at room temperature (25°C) for 24 hours to facilitate the radical cyclization.
- Quench the reaction with saturated brine, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic methodology offers profound strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing hazardous cyanide salts with stable bromoacetonitrile, companies can avoid the stringent regulatory hurdles and specialized logistics associated with transporting and storing highly toxic substances. This shift not only reduces insurance and compliance costs but also mitigates the risk of supply disruptions caused by regulatory crackdowns on dangerous goods. Furthermore, the elimination of stoichiometric copper salts removes a significant cost center related to both reagent purchase and the subsequent expensive processes required for heavy metal scavenging and waste disposal. The overall cost structure for manufacturing these intermediates is therefore significantly optimized through material efficiency and waste reduction.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive and hazardous reagents with commodity chemicals. Traditional methods often require costly transition metal catalysts in stoichiometric quantities, along with oxidants that generate large volumes of chemical waste. In this new protocol, the catalyst loading is reduced to a mere 2 mol%, and the oxidant is effectively replaced by the photon energy from LEDs, which is negligible in cost. Additionally, the simplified workup procedure, which avoids complex metal removal steps, reduces the consumption of solvents and purification media. These factors collectively contribute to a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates without compromising on margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials. Bromoacetonitrile and simple amide precursors are commercially abundant and do not suffer from the supply volatility often seen with specialized organometallic reagents. The reaction conditions are mild, operating at room temperature, which reduces the energy load on manufacturing facilities and minimizes the risk of thermal runaways or equipment failure. This reliability ensures consistent production schedules and shorter lead times for delivering high-purity intermediates to downstream customers. The robustness of the method across a wide range of substrates, including those with electron-donating and electron-withdrawing groups, further guarantees that diverse product portfolios can be maintained without needing to requalify entirely different synthetic routes for each variant.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but advancements in LED technology and flow chemistry have made this increasingly viable. The absence of toxic cyanide waste streams aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. Facilities can operate with reduced environmental liability, as the effluent does not require specialized treatment for heavy metals or cyanide detoxification. This environmental compatibility facilitates smoother regulatory approvals for new manufacturing sites and expansions. The straightforward isolation of products via extraction and chromatography suggests that the process can be adapted for continuous flow manufacturing, offering a clear pathway from gram-scale laboratory optimization to multi-ton commercial production with minimal re-engineering efforts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value for potential partners. Understanding these nuances is essential for making informed decisions about integrating this technology into existing production pipelines.
Q: What are the safety advantages of using bromoacetonitrile over traditional cyanide sources?
A: Traditional cyanation often relies on highly toxic alkali metal cyanides like KCN or NaCN. This patent utilizes bromoacetonitrile as a safer cyanomethyl source, significantly reducing handling risks and environmental hazards associated with free cyanide ions.
Q: Does this method require expensive transition metals like copper?
A: No. Unlike previous methods that required stoichiometric amounts of copper salts and peroxides, this protocol uses a catalytic amount (2 mol%) of an iridium photoredox catalyst, eliminating the need for heavy metal removal steps.
Q: Is this process suitable for large-scale manufacturing?
A: Yes. The reaction operates at room temperature under visible light irradiation, avoiding extreme thermal conditions. The simplicity of the workup and the absence of hazardous reagents make it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitrile Ethyl-2-Hydrocarbyl-4H-Benzoxazine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN110183394B can be realized on an industrial scale. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical clients. We understand that the transition from bench-scale discovery to full-scale manufacturing requires not just chemical expertise but also deep process engineering knowledge, which our team delivers consistently to guarantee supply continuity and product quality.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener, more efficient route can improve your bottom line. Please contact us to request specific COA data for related benzoxazine derivatives and comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates while optimizing your manufacturing costs and environmental footprint.
