Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
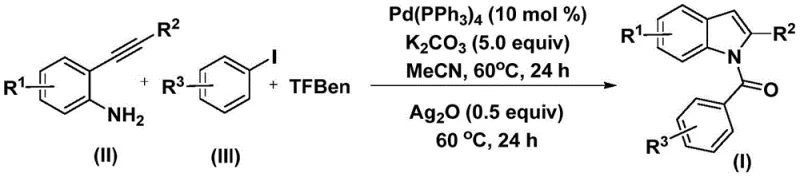
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds, particularly indoles, which serve as critical backbones in numerous bioactive molecules ranging from anti-inflammatory agents to anti-tumor drugs. A significant breakthrough in this domain is detailed in Chinese Patent CN112898192B, which discloses a highly efficient preparation method for N-acyl indole compounds. This novel approach leverages a palladium-catalyzed carbonylation cyclization reaction, utilizing readily available 2-alkynyl anilines and aryl iodides as starting materials. By employing a solid carbon monoxide substitute, the process circumvents the safety hazards associated with traditional high-pressure CO gas methods, offering a safer and more operationally simple pathway for producing these valuable intermediates. The technology represents a substantial advancement for any reliable pharmaceutical intermediate supplier aiming to optimize their production capabilities while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of N-acyl indoles often involves multi-step sequences or the use of hazardous gaseous carbon monoxide under high pressure, which poses significant challenges for industrial scale-up and safety compliance. Conventional carbonylation reactions typically require specialized autoclaves and rigorous safety protocols to handle toxic CO gas, leading to increased capital expenditure and operational complexity. Furthermore, older methodologies may suffer from limited substrate tolerance, where sensitive functional groups on the aromatic rings are incompatible with harsh reaction conditions, resulting in lower yields and difficult purification processes. These limitations create bottlenecks in the supply chain, extending lead times and increasing the overall cost of goods sold for downstream API manufacturers who rely on consistent and high-purity supplies of these complex heterocycles.
The Novel Approach
In stark contrast, the method described in the patent utilizes a mild, one-pot strategy that integrates carbonylation and cyclization into a streamlined sequence. By replacing gaseous CO with 1,3,5-tricarboxylic acid phenol ester (TFBen), the reaction can be conducted in standard glassware at atmospheric pressure, drastically reducing equipment costs and safety risks. The process operates at a moderate temperature of 60°C in acetonitrile, demonstrating exceptional functional group tolerance for substituents like halogens, alkyls, and alkoxy groups. This innovation not only simplifies the workflow but also enhances the overall reaction efficiency, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The ability to synthesize diverse N-acyl indole derivatives in a single vessel without isolating unstable intermediates marks a pivotal shift towards greener and more economical chemical production.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The core of this synthetic breakthrough lies in the intricate palladium catalytic cycle that orchestrates the formation of the indole ring and the acyl group simultaneously. The mechanism initiates with the oxidative addition of the palladium catalyst into the aryl iodide bond, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, which is slowly released in situ from the decomposition of TFBen, inserts into this palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The final crucial step involves the addition of silver oxide, which acts as an oxidant to promote the intramolecular cyclization of the amide, closing the indole ring and regenerating the active catalyst. This mechanistic pathway ensures high atom economy and minimizes the formation of by-products, which is critical for maintaining high purity specifications in fine chemical synthesis.
From an impurity control perspective, the mild reaction conditions play a vital role in suppressing side reactions that often plague high-temperature processes. The use of silver oxide in the second stage specifically targets the cyclization step, preventing premature polymerization or decomposition of the sensitive alkyne moiety. The choice of acetonitrile as the solvent further aids in dissolving both organic substrates and inorganic bases like potassium carbonate, ensuring a homogeneous reaction environment that promotes consistent kinetics. For R&D directors, understanding this mechanism highlights the robustness of the process; the distinct two-stage temperature profile (both at 60°C) allows for precise control over the reaction progress, ensuring that the carbonylation completes before the cyclization begins, thereby maximizing the yield of the desired N-acyl indole product while minimizing difficult-to-remove impurities.
How to Synthesize N-Acyl Indole Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory equipment and commercially available reagents. The protocol involves mixing the palladium catalyst, base, CO source, and substrates in a solvent, followed by a controlled heating sequence. This straightforward procedure eliminates the need for complex gas handling systems, making it accessible for both pilot-scale development and large-scale commercial production. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process chemists in replicating these high-yielding results.
- Combine palladium catalyst, potassium carbonate, TFBen (CO source), 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide to the reaction mixture and continue heating at 60°C for another 24 hours to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical efficiency. The transition from hazardous gas-based carbonylation to a solid-source protocol fundamentally alters the risk profile of the manufacturing process, leading to significant operational improvements. By removing the dependency on high-pressure infrastructure, facilities can reduce their regulatory burden and insurance costs, directly impacting the bottom line. Furthermore, the use of widely available starting materials such as aryl iodides and 2-alkynyl anilines ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO equipment and the associated safety infrastructure leads to substantial capital expenditure savings and lower operational overheads. Additionally, the high conversion rates and simplified one-pot nature of the reaction reduce solvent consumption and waste generation, contributing to a more sustainable and cost-effective production model. The use of inexpensive bases like potassium carbonate and the potential for catalyst recovery further enhance the economic viability of this route, allowing for competitive pricing in the market without compromising on quality.
- Enhanced Supply Chain Reliability: Since the key reagents, including the palladium catalyst and TFBen, are commercially available off-the-shelf products, the risk of supply disruption is minimized. This accessibility allows for flexible inventory management and rapid scaling of production capacity in response to market demand. The robustness of the reaction against various functional groups means that a single platform technology can be used to produce a wide library of derivatives, streamlining the supply chain for diverse project pipelines and reducing the need for multiple specialized manufacturing lines.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions make this process inherently safer and more environmentally friendly, aligning with modern green chemistry principles. The simplified post-treatment, involving filtration and column chromatography, facilitates easier scale-up from gram to kilogram scales without encountering the heat transfer or mixing issues common in high-pressure reactions. This scalability ensures that the method can seamlessly transition from R&D to commercial manufacturing, supporting the long-term growth of the product portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These answers are derived directly from the experimental data and technical disclosures within the patent, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: What is the typical yield range for this N-acyl indole preparation method?
A: According to the patent data, the reaction demonstrates good efficiency with isolated yields ranging generally from moderate to high levels, specifically observed between 44% and 82% across various substrate examples.
Q: Is this method compatible with diverse functional groups on the aromatic rings?
A: Yes, the method exhibits excellent substrate compatibility, tolerating various substituents such as methyl, methoxy, halogens (F, Cl, Br), and trifluoromethyl groups on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly evaluated the technology described in CN112898192B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative method to life. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art manufacturing facilities. Our capability to handle complex palladium-catalyzed reactions ensures that we can consistently supply materials that accelerate your drug discovery and development timelines.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to discuss your specific COA data requirements and to receive comprehensive route feasibility assessments that demonstrate how our optimized processes can drive value and efficiency in your supply chain.
