Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Pharmaceutical Applications
Introduction to Next-Generation Indole Synthesis
The indole scaffold represents one of the most privileged structures in modern medicinal chemistry, serving as the core backbone for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As illustrated in the structural diversity of known pharmaceuticals, the ability to efficiently functionalize the nitrogen atom of the indole ring is critical for drug discovery and process development. 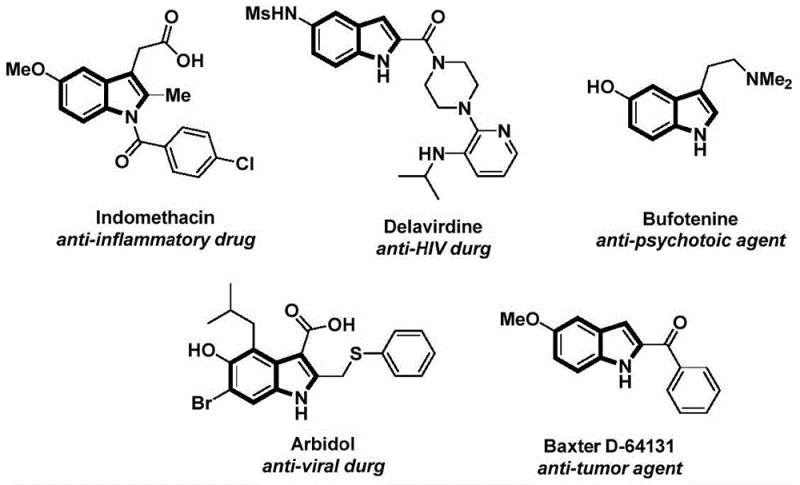 Recent advancements in synthetic methodology have sought to address the limitations of traditional acylation techniques, culminating in the innovative approach detailed in Chinese Patent CN112898192B. This patent discloses a robust preparation method for N-acyl indole compounds that leverages a palladium-catalyzed carbonylation cyclization strategy. By utilizing 2-alkynyl aniline and aryl iodides as readily available starting materials, this technology offers a streamlined, one-pot solution that significantly enhances reaction efficiency and substrate compatibility compared to legacy methods.
Recent advancements in synthetic methodology have sought to address the limitations of traditional acylation techniques, culminating in the innovative approach detailed in Chinese Patent CN112898192B. This patent discloses a robust preparation method for N-acyl indole compounds that leverages a palladium-catalyzed carbonylation cyclization strategy. By utilizing 2-alkynyl aniline and aryl iodides as readily available starting materials, this technology offers a streamlined, one-pot solution that significantly enhances reaction efficiency and substrate compatibility compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has relied heavily on direct acylation using acid chlorides or anhydrides, which often necessitates harsh reaction conditions and generates stoichiometric amounts of corrosive waste. Furthermore, traditional carbonylation routes typically require the use of gaseous carbon monoxide, a highly toxic and flammable reagent that demands specialized high-pressure autoclaves and rigorous safety protocols, thereby inflating capital expenditure and operational complexity. These conventional pathways frequently suffer from poor atom economy and limited functional group tolerance, particularly when sensitive moieties are present on the aromatic rings. The reliance on hazardous gases also introduces significant supply chain vulnerabilities and regulatory hurdles for large-scale manufacturing facilities, making the search for safer, solid-state alternatives a priority for forward-thinking process chemists.
The Novel Approach
The methodology described in patent CN112898192B circumvents these challenges by employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe, solid carbon monoxide surrogate. This innovative substitution allows the carbonylation reaction to proceed under mild atmospheric conditions at a moderate temperature of 60°C, eliminating the need for dangerous high-pressure equipment. The process integrates the carbonylation and cyclization steps into a seamless sequence, first forming an amide intermediate via palladium catalysis and subsequently inducing ring closure through the addition of silver oxide. This dual-catalyst system not only simplifies the operational workflow but also broadens the scope of compatible substrates, enabling the efficient synthesis of complex N-acyl indoles that were previously difficult to access. The result is a versatile platform technology that aligns perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The core of this transformative synthesis lies in its elegant mechanistic pathway, which orchestrates multiple bond-forming events in a single vessel. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide, generating a reactive aryl-palladium intermediate. Crucially, the TFBen additive decomposes under the reaction conditions to release carbon monoxide in situ, which then inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate subsequently undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an N-aryl amide precursor. 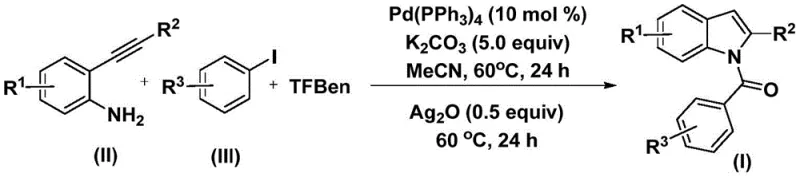 The final stage involves the addition of silver oxide, which acts as both an oxidant and a promoter for the intramolecular cyclization of the alkyne moiety, ultimately forging the indole ring structure. This precise control over the reaction trajectory minimizes side reactions and ensures high regioselectivity.
The final stage involves the addition of silver oxide, which acts as both an oxidant and a promoter for the intramolecular cyclization of the alkyne moiety, ultimately forging the indole ring structure. This precise control over the reaction trajectory minimizes side reactions and ensures high regioselectivity.
From an impurity control perspective, the use of mild conditions and specific reagents like potassium carbonate helps maintain a clean reaction profile. The stepwise addition of silver oxide after the initial 24-hour period allows for the complete consumption of the amide intermediate before triggering the cyclization, thereby preventing the accumulation of partially reacted species. The compatibility of this system with various electron-donating and electron-withdrawing groups suggests that the electronic nature of the substrates does not drastically alter the fundamental catalytic cycle, providing a predictable and robust process for diverse chemical libraries. This mechanistic stability is paramount for ensuring consistent product quality and minimizing the formation of hard-to-remove byproducts during scale-up.
How to Synthesize N-Acyl Indole Compounds Efficiently
Implementing this synthesis requires careful attention to reagent ratios and thermal profiles to maximize yield and purity. The standard protocol involves charging a reaction vessel with the palladium catalyst, base, CO source, and substrates in an organic solvent such as acetonitrile. The mixture is heated to facilitate the initial coupling, followed by a second heating phase after the introduction of the silver oxidant. 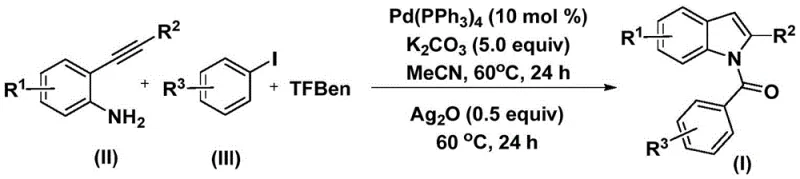 This two-stage thermal treatment is critical for driving both the carbonylation and the subsequent cyclization to completion. For detailed operational parameters and specific molar equivalents optimized for different substrates, please refer to the standardized synthesis guide below.
This two-stage thermal treatment is critical for driving both the carbonylation and the subsequent cyclization to completion. For detailed operational parameters and specific molar equivalents optimized for different substrates, please refer to the standardized synthesis guide below.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide to the mixture and continue heating at 60°C for another 24 hours to promote cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. By replacing hazardous gaseous carbon monoxide with a stable solid powder, the process drastically reduces the safety infrastructure requirements, leading to significant capital cost avoidance in facility design and maintenance. The use of commercially available starting materials, such as simple aryl iodides and 2-alkynyl anilines, ensures a resilient supply chain that is less susceptible to the volatility associated with specialized reagents. Furthermore, the mild reaction temperature of 60°C translates to lower energy consumption compared to high-temperature reflux protocols, contributing to a reduced carbon footprint and lower utility costs per kilogram of product.
- Cost Reduction in Manufacturing: The elimination of high-pressure autoclaves and specialized gas handling systems removes a major barrier to entry for manufacturing this class of intermediates. Additionally, the use of TFBen as a CO source avoids the logistical costs and safety premiums associated with transporting and storing toxic gases. The high atom economy of the reaction means that a larger proportion of the raw material mass is converted into the final product, reducing waste disposal costs and improving overall material efficiency. This streamlined approach allows for a more competitive cost structure, making it an attractive option for large-scale API production where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like potassium carbonate and acetonitrile, alongside widely available palladium catalysts, mitigates the risk of supply disruptions. Unlike processes dependent on custom-synthesized or exotic reagents, this method leverages a supply base that is deep and diversified. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor process deviations. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which involves filtration and standard column chromatography, facilitates easy translation from laboratory benchtop to pilot plant and full commercial scale. The absence of toxic gas emissions simplifies environmental permitting and reduces the burden on scrubber systems. Moreover, the high substrate compatibility allows for the rapid generation of analog libraries without the need for extensive process re-optimization for each new derivative. This flexibility accelerates time-to-market for new drug candidates while ensuring that the manufacturing process remains compliant with increasingly strict environmental regulations regarding volatile organic compounds and hazardous waste.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential licensees and partners.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a safe, solid surrogate for toxic carbon monoxide gas, allowing the reaction to proceed under mild atmospheric pressure without specialized high-pressure equipment.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents including halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Q: How does the two-stage temperature profile affect the yield?
A: Maintaining a consistent temperature of 60°C for both the initial coupling (24 hours) and the subsequent silver-mediated cyclization (24 hours) ensures high conversion rates, with isolated yields ranging from 44% to 82% across diverse substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that efficient synthetic methodologies play in accelerating drug development pipelines. Our team of expert process chemists has thoroughly evaluated the technology disclosed in CN112898192B and is fully equipped to leverage this palladium-catalyzed route for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are designed to handle complex organometallic reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to unlock the full potential of this advanced N-acyl indole synthesis. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss your project specifics,索取 specific COA data for related intermediates, and receive comprehensive route feasibility assessments that will empower your R&D and supply chain strategies.
