Advanced Synthesis Of Ortho-Dimethyl Diphosphine Ligands For Industrial Catalysis And Commercial Scale-Up
Advanced Synthesis Of Ortho-Dimethyl Diphosphine Ligands For Industrial Catalysis And Commercial Scale-Up
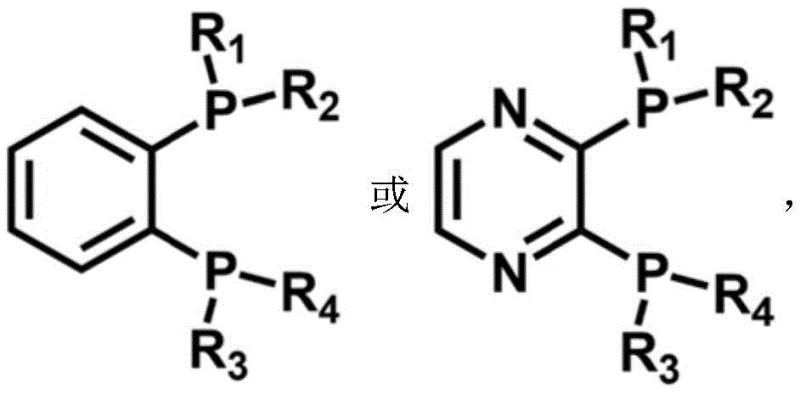
The landscape of organometallic catalysis is continuously evolving, driven by the demand for more efficient and selective ligand systems that can operate under milder conditions while maintaining robust stability. Patent CN113999261B introduces a groundbreaking methodology for the synthesis of ortho-dimethyl aromatic ring type diphosphine ligand compounds, addressing critical bottlenecks in the production of high-performance catalysts. This innovation utilizes a streamlined three-step process starting from ortho-dihalogenated methyl aromatic hydrocarbons, proceeding through phosphonate formation and lithium aluminum hydride reduction, and culminating in the final substitution with diverse organic ligands. The technical significance of this patent lies in its ability to produce a series of bidentate phosphine ligands with exceptional purity exceeding 98% and yields ranging from 77% to 92%, which represents a substantial improvement over legacy synthetic routes that often suffer from low atom economy and difficult purification protocols.
For research and development directors overseeing catalyst formulation, the structural versatility offered by this patent is paramount. The method allows for the precise tuning of steric and electronic properties by varying the R groups attached to the phosphorus atoms, enabling the customization of catalysts for specific transformations such as the alkoxycarbonylation of terminal, internal, and branched alkenes. By establishing a direct and efficient pathway to these valuable intermediates, the technology not only accelerates the discovery of new catalytic systems but also provides a reliable foundation for the commercial scale-up of complex polymer additives and pharmaceutical intermediates. The following analysis details the mechanistic advantages and commercial implications of adopting this synthesis strategy within a modern fine chemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing benzyl and aromatic ring organic phosphine ligands have long been plagued by inherent inefficiencies that hinder their widespread industrial adoption. Conventional routes often rely on multi-step sequences involving harsh reaction conditions, expensive transition metal catalysts, and extensive use of solvents that are difficult to remove during post-treatment. A significant drawback of these legacy processes is the reliance on triphenylphosphine modified systems, which, while effective, frequently lack the necessary steric hindrance to achieve high selectivity in complex coupling reactions. Furthermore, the purification of crude phosphine products from these traditional syntheses often requires chromatographic separation on a large scale, which is economically prohibitive and generates substantial chemical waste, thereby increasing the overall environmental footprint and production costs for manufacturers seeking a reliable agrochemical intermediate supplier or pharma partner.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a direct substitution strategy that dramatically simplifies the synthetic architecture. By initiating the sequence with the reaction of ortho-dihalogenated methyl aromatic hydrocarbons and phosphite esters, the method efficiently constructs the carbon-phosphorus backbone in a single high-yielding step. This is followed by a controlled reduction using lithium aluminum hydride to generate the reactive phosphine hydrogen species, which serves as a versatile platform for further functionalization. The final step involves a one-pot reaction with halogenated, olefin, or alkyne ligands to install the desired organic groups, resulting in the target ortho-dimethyl aromatic ring diphosphine ligand compounds. This streamlined workflow eliminates the need for intermediate isolation of unstable species and reduces the total number of unit operations, directly translating to lower operational expenditures and enhanced process safety.
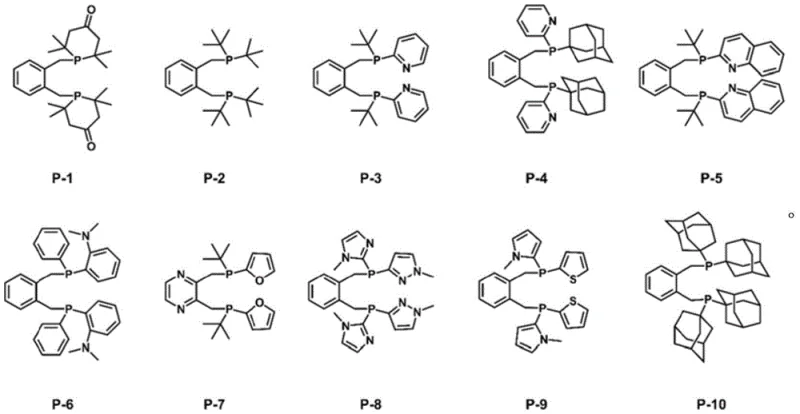
The structural diversity achievable through this method is exemplified by the range of compounds synthesized, including derivatives with bulky adamantyl groups, heterocyclic pyridyl moieties, and various olefinic chains. As illustrated in the provided structural data, the method accommodates a wide array of substituents without compromising the integrity of the central aromatic scaffold. This modularity is crucial for optimizing catalyst performance in specific applications, such as the synthesis of fine chemicals or the production of advanced materials. The ability to access these complex architectures through a unified synthetic platform underscores the robustness of the technology and its potential to serve as a cornerstone for developing next-generation catalytic systems that meet the rigorous demands of modern industrial chemistry.
Mechanistic Insights into LiAlH4-Mediated Phosphine Synthesis
The core of this innovative synthesis lies in the strategic use of lithium aluminum hydride (LiAlH4) combined with trimethylchlorosilane (TMSCl) to effect the reduction of phosphonate esters to phosphine hydrides. This transformation is mechanistically distinct from traditional reduction methods that might utilize silanes alone or require high-pressure hydrogenation. In this protocol, the in situ generation of a reactive silyl-aluminum hydride species facilitates the cleavage of the phosphorus-oxygen bonds in the phosphonate intermediate with high chemoselectivity. The reaction is conducted at low temperatures, typically below -50°C during the addition phase, to prevent side reactions such as over-reduction or decomposition of the sensitive P-H bond. This careful thermal management ensures that the resulting ortho-diphosphine hydrogen methyl aromatic hydrocarbon is obtained in near-quantitative yields, providing a clean substrate for the subsequent substitution steps.
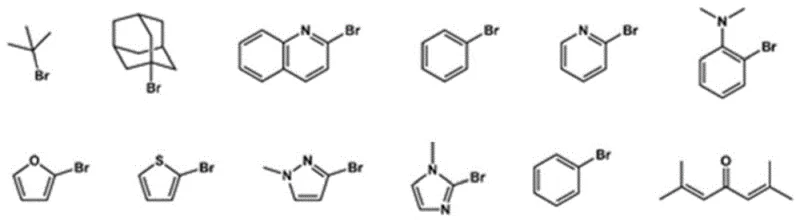
Following the generation of the phosphine hydride intermediate, the mechanism proceeds via a nucleophilic substitution or addition pathway depending on the nature of the incoming ligand. When reacting with halogenated ligands, the phosphorus center acts as a nucleophile, displacing the halide leaving group to form the new P-C bond. In cases where olefin or alkyne ligands are employed, the reaction likely proceeds through an addition mechanism across the unsaturated bond, potentially facilitated by the inherent reactivity of the P-H species or mild thermal activation at 120°C. The versatility of this step allows for the incorporation of diverse functional groups, as shown in the variety of reactants available for this transformation. This mechanistic flexibility is a key driver for the broad applicability of the resulting ligands in catalytic cycles, where the electronic density and steric bulk of the phosphine substituents play a pivotal role in stabilizing metal centers and directing substrate orientation.
Impurity control is another critical aspect managed by this mechanistic design. The use of distillation for the initial phosphonate intermediate and recrystallization for the final product ensures that side products, such as mono-substituted species or oxidized phosphine oxides, are effectively removed. The high purity specifications achieved (>98%) are essential for preventing catalyst poisoning in downstream applications, particularly in the synthesis of active pharmaceutical ingredients where trace metal or organic impurities can compromise drug safety. By understanding the precise reaction conditions that favor the formation of the desired diphosphine structure, manufacturers can implement rigorous quality control measures to guarantee batch-to-batch consistency, a requirement that is non-negotiable for any entity positioning itself as a reliable specialty chemical supplier.
How to Synthesize Ortho-Dimethyl Diphosphine Ligands Efficiently
The practical implementation of this synthesis route requires adherence to strict inert atmosphere conditions and precise temperature control to maximize yield and safety. The process begins with the preparation of the phosphonate precursor, followed by the sensitive reduction step, and concludes with the functionalization of the phosphine core. Each stage has been optimized to balance reaction kinetics with product stability, ensuring that the highly reactive phosphine intermediates are handled safely while maintaining high throughput. The detailed standardized synthesis steps provided below outline the specific reagents, molar ratios, and workup procedures necessary to replicate the high-performance results reported in the patent literature.
- React ortho-dihalogenated methyl aromatic hydrocarbon with phosphite ester at 130°C to form the phosphonate intermediate.
- Reduce the phosphonate using lithium aluminum hydride and trimethylchlorosilane at low temperatures to generate the phosphine hydrogen species.
- Perform nucleophilic substitution with halogenated, olefin, or alkyne ligands at 120°C to finalize the diphosphine ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis methodology offers transformative benefits that extend beyond mere technical performance. The simplification of the synthetic route directly correlates with a reduction in manufacturing complexity, which is a primary driver for cost optimization in the fine chemical sector. By eliminating the need for multiple purification stages and reducing the consumption of exotic reagents, the process lowers the variable cost per kilogram of the final ligand product. This efficiency gain allows suppliers to offer more competitive pricing structures without sacrificing margin, addressing the constant pressure on procurement managers to reduce costs in pharmaceutical manufacturing and other high-value sectors. Furthermore, the use of commodity chemicals such as dibromo-o-xylene and tributyl phosphite as starting materials mitigates supply risk, as these feedstocks are widely available from established global producers.
- Cost Reduction in Manufacturing: The streamlined three-step process significantly reduces the operational burden associated with solvent recovery and waste disposal. Traditional methods often generate large volumes of saline waste from quenching steps or require expensive chromatographic media for purification, both of which add substantial overhead to the production budget. In contrast, this novel approach utilizes distillation and recrystallization, which are scalable and cost-effective unit operations. The high atomic economy of the reaction means that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste generation and aligning with green chemistry principles that are increasingly mandated by regulatory bodies and corporate sustainability goals.
- Enhanced Supply Chain Reliability: The robustness of the synthesis against variations in reaction conditions enhances the reliability of supply. Because the method does not rely on sensitive catalysts that require stringent storage or handling, the risk of production delays due to reagent degradation is minimized. Additionally, the ability to produce a wide range of ligand variants (P-1 to P-10) using a common intermediate platform allows for flexible manufacturing scheduling. Suppliers can respond more agilely to fluctuating market demands for specific ligand types, ensuring that customers receive their orders on time. This flexibility is crucial for maintaining continuity in downstream production lines, particularly in the pharmaceutical industry where interruptions can have cascading effects on drug availability.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations such as round bottom or Schlenk flasks that translate easily to industrial-scale vessels. The absence of high-pressure hydrogenation steps or cryogenic requirements beyond standard cooling capabilities simplifies the engineering requirements for plant expansion. Moreover, the reduced solvent usage and simplified workup procedures contribute to a lower environmental footprint, facilitating compliance with increasingly stringent environmental regulations. This alignment with sustainability standards not only reduces regulatory risk but also enhances the brand value of the supply chain partners, appealing to end-users who prioritize eco-friendly sourcing strategies in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of the ortho-dimethyl diphosphine ligands produced via this patented method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing transparency for potential partners evaluating this technology for integration into their supply chains. Understanding these details is essential for making informed decisions about sourcing strategies and process development.
Q: What are the key advantages of this new synthesis method over traditional routes?
A: The patented method significantly simplifies the process by reducing the number of steps and utilizing readily available raw materials like ortho-dihalogenated methyl aromatic hydrocarbons. It achieves high yields ranging from 77% to 92% and purity exceeding 98%, eliminating the need for complex post-treatment solvents often required in conventional triphenylphosphine modifications.
Q: Can these ligands be used for large-scale industrial applications?
A: Yes, the synthesis is designed for industrial mass production. The operations involve standard unit processes such as distillation and recrystallization, which are easily scalable. The method conforms to green chemistry principles by minimizing waste and avoiding excessive solvent use, making it suitable for commercial scale-up of complex catalysts.
Q: What types of catalytic reactions benefit from these ortho-dimethyl diphosphine ligands?
A: These ligands exhibit excellent catalytic activity and selectivity in hydroformylation, esterification of olefins, and alkoxycarbonylation reactions. They are particularly effective when modifying palladium catalyst systems, offering superior steric hindrance effects compared to monodentate ligands, which enhances performance in synthesizing functional materials and bioactive compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in driving innovation across the pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to manufacture complex organophosphorus compounds positions us as a strategic partner for companies seeking to secure a stable supply of critical catalytic materials.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current catalyst sourcing or require specific COA data and route feasibility assessments for new ligand variants, we are ready to provide the expertise and support you need. By collaborating with us, you gain access to a supply chain that prioritizes quality, reliability, and continuous improvement, empowering your organization to achieve its scientific and commercial objectives with confidence.
